Abstract
Background
Clinical studies have shown similar rapid improvements in body mass and glycemic control after Roux-en-Y gastric bypass (RYGB) and vertical sleeve gastrectomy (VSG). Evidence suggests that adaptive intestinal tissue growth and reprogramming of intestinal glucose disposal play a key role in the beneficial effects on glucose homeostasis after RYGB, but it is not known whether such adaptive changes also occur after sleeve gastrectomy.
Methods
High-fat diet-induced obese rats were subjected to either VSG or RYGB, and intestinal growth and functional adaptations were assessed by using morphometric, immunohistochemical, and immuno-blot techniques, 3 months after surgery or sham surgery.
Results
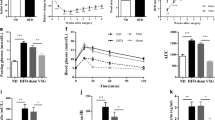
The cross-sectional areas of the Roux and common limbs are significantly increased after RYGB compared with sham surgery (Roux limb: 17.1 ± 4.0 vs. 5.5 ± 0.1 mm2; common limb: 11.7 ± 0.6 vs. 5.1 ± 0.5 mm2; p < 0.01), but the cross-sectional area of the corresponding jejunum is not different from controls after VSG. Similarly, mucosal thickness and the number of GLP-1 cells are not increased after VSG. Protein expression of hexokinase II is increased fourfold (p < 0.01) in the Roux limb after RYGB, but not in the jejunum after VSG.
Conclusions
Adaptive hypertrophy and reprogramming of glucose metabolism in the small intestine are not necessary for VSG to improve body composition and glycemic control. The similar beneficial effects of VSG and RYGB on glucose homeostasis might be mediated by different mechanisms.





Similar content being viewed by others
References
Schauer PR, Bhatt DL, Kirwan JP, et al. Bariatric surgery versus intensive medical therapy for diabetes—3-year outcomes. N Engl J Med. 2014;370:2002–13.
Chambers AP, Smith EP, Begg DP, et al. Regulation of gastric emptying rate and its role in nutrient-induced GLP-1 secretion in rats after vertical sleeve gastrectomy. Am J Physiol Endocrinol Metab. 2014;306:E424–32.
Evrard S, Aprahamian M, Hoeltzel A, et al. Trophic and enzymatic adaptation of the intestine to biliopancreatic bypass in the rat. Int J Obes Relat Metab Disord. 1993;17:541–7.
Borg CM, le Roux CW, Ghatei MA, et al. Biliopancreatic diversion in rats is associated with intestinal hypertrophy and with increased GLP-1, GLP-2 and PYY levels. Obes Surg. 2007;17:1193–8.
le Roux CW, Borg C, Wallis K, et al. Gut hypertrophy after gastric bypass is associated with increased glucagon-like peptide 2 and intestinal crypt cell proliferation. Ann Surg. 2010;252:50–6.
Taqi E, Wallace LE, de Heuvel E, et al. The influence of nutrients, biliary-pancreatic secretions, and systemic trophic hormones on intestinal adaptation in a Roux-en-Y bypass model. J Pediatr Surg. 2010;45:987–95.
Saeidi N, Meoli L, Nestoridi E, et al. Reprogramming of intestinal glucose metabolism and glycemic control in rats after gastric bypass. Science. 2013;341:406–10.
Mumphrey MB, Patterson LM, Zheng H, et al. Roux-en-Y gastric bypass surgery increases number but not density of CCK-, GLP-1-, 5-HT-, and neurotensin-expressing enteroendocrine cells in rats. Neurogastroenterol Motil. 2013;25:e70–9.
Hansen CF, Bueter M, Theis N, et al. Hypertrophy dependent doubling of L-cells in Roux-en-Y gastric bypass operated rats. PLoS ONE. 2013;8:e65696.
Martin GR, Beck PL, Sigalet DL. Gut hormones, and short bowel syndrome: the enigmatic role of glucagon-like peptide-2 in the regulation of intestinal adaptation. World J Gastroenterol. 2006;12:4117–29.
Martin GR, Wallace LE, Sigalet DL. Glucagon-like peptide-2 induces intestinal adaptation in parenterally fed rats with short bowel syndrome. Am J Physiol Gastrointest Liver Physiol. 2004;286:G964–72.
Sigalet DL, Bawazir O, Martin GR, et al. Glucagon-like peptide-2 induces a specific pattern of adaptation in remnant jejunum. Dig Dis Sci. 2006;51:1557–66.
Kaji T, Tanaka H, Redstone H, et al. Temporal changes in the intestinal growth promoting effects of glucagon-like peptide 2 following intestinal resection. J Surg Res. 2009;152:271–80.
Hua Z, Turner JM, Sigalet DL, et al. Role of glucagon-like peptide-2 deficiency in neonatal short-bowel syndrome using neonatal piglets. Pediatr Res. 2013;73:742–9.
Ljungmann K, Hartmann B, Kissmeyer-Nielsen P, et al. Time-dependent intestinal adaptation and GLP-2 alterations after small bowel resection in rats. Am J Physiol Gastrointest Liver Physiol. 2001;281:G779–85.
Romero F, Nicolau J, Flores L, et al. Comparable early changes in gastrointestinal hormones after sleeve gastrectomy and Roux-En-Y gastric bypass surgery for morbidly obese type 2 diabetic subjects. Surg Endosc. 2012;26:2231–9.
Cummings BP, Bettaieb A, Graham JL, et al. Vertical sleeve gastrectomy improves glucose and lipid metabolism and delays diabetes onset in UCD-T2DM rats. Endocrinology. 2012;153:3620–32.
Kunnecke B, Verry P, Benardeau A, et al. Quantitative body composition analysis in awake mice and rats by magnetic resonance relaxometry. Obes Res. 2004;12:1604–15.
Helmio M, Victorzon M, Ovaska J, et al. Comparison of short-term outcome of laparoscopic sleeve gastrectomy and gastric bypass in the treatment of morbid obesity: a prospective randomized controlled multicenter SLEEVEPASS study with 6-month follow-up. Scand J Surg. 2014;103:175–81.
Peterli R, Steinert RE, Woelnerhanssen B, et al. Metabolic and hormonal changes after laparoscopic Roux-en-Y gastric bypass and sleeve gastrectomy: a randomized, prospective trial. Obes Surg. 2012;22:740–8.
Chambers AP, Jessen L, Ryan KK, et al. Weight-independent changes in blood glucose homeostasis after gastric bypass or vertical sleeve gastrectomy in rats. Gastroenterology. 2011;141:950–8.
Acknowledgments
This study was funded and supported by National Institutes of Health grant DK047348 (HRB).
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
It does not apply.
Animal Rights
All applicable institutional and/or national guidelines for the care and use of animals were followed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mumphrey, M.B., Hao, Z., Townsend, R.L. et al. Sleeve Gastrectomy Does Not Cause Hypertrophy and Reprogramming of Intestinal Glucose Metabolism in Rats. OBES SURG 25, 1468–1473 (2015). https://doi.org/10.1007/s11695-014-1547-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-014-1547-9