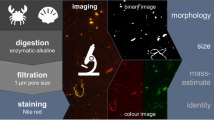
Abstract
As a popular nutraceutical, collagen powder is the target of adulteration like other high-value food products, therefore detection of adulteration in collagen powder is of great practical significance to ensure market order and the health of the people. To achieve accurate detection of adulterants in collagen powder, a modeling method of fluorescence hyperspectral technology combined with machine learning algorithm was proposed. This study employed various preprocessing methods for denoising and spectral correction to enhance the effective spectral data. Principal component analysis was used to visualize the spectral data and initially revealed the spatial distribution of adulterated collagen powder. Genetic algorithm-k nearest neighbor, particle swarm optimization-support vector machine, and gradient boosting decision tree were used to construct classification models to identify adulterated collagen powder, with the best 2-class discriminant model accuracy, 4-class discriminant model accuracy, and 5-class discriminant model accuracy reaching 99%, 94%, and 98%, respectively. In the quantitative detection models of adulteration level, the random forest model performed best with correlation coefficient (R2) of 0.95 to 0.99. These results suggested that fluorescence hyperspectral technology combined with machine learning algorithm can be effectively used to detect adulterated collagen powder.




Similar content being viewed by others
Data availability
Data will be made available on request.
Abbreviations
- FTIR:
-
Fourier transform infrared spectroscopy
- GA:
-
Genetic algorithm
- GA-KNN:
-
Genetic algorithm-k nearest neighbor
- GBDT:
-
Gradient boosting decision tree
- KNN:
-
K-nearest neighbor
- MA:
-
Moving average
- MSC:
-
Multivariate scatter correction
- NIR:
-
Near-infrared spectroscopy
- PCA:
-
Principal component analysis
- PSO:
-
Particle swarm optimization
- PSO-SVM:
-
Particle swarm optimization-support vector machine
- RF:
-
Random forest
- SG:
-
Savitzky-golay
- SVM:
-
Support vector machine
References
S. Ricard-Blum, The collagen family. Cold Spring Harb. Perspect. Biol. 3, a004978 (2011). https://doi.org/10.1101/cshperspect.a004978
L. Dias Campos, A.T.S. de Almeida Pereira, C.B.B. Cazarin, The collagenmarket and knowledge, attitudes, and practices of Brazilian consumers regarding collagen ingestion. Food Res. Int. 170, 112951 (2023). https://doi.org/10.1016/j.foodres.2023.112951
S. Medina, R. Perestrelo, P. Silva, J.A.M. Pereira, J.S. Câmara, Current trends and recent ad-vances on food authenticity technologies and chemometric approaches. Trends Food Sci. Technol. 85, 163–176 (2019). https://doi.org/10.1016/j.tifs.2019.01.017
M.S. Martins, M.H. Nascimento, L.L. Barbosa, L.C.G. Campos, M.N. Singh, F.L. Martin, W. Romão, P.R. Filgueiras, V.G. Barauna, Detection and quantification using ATR-FTIR spectroscopy of whey protein concentrate adulteration with wheat flour. LWT 172, 114161 (2022). https://doi.org/10.1016/j.lwt.2022.114161
J. Andrade, C. Guimarães Pereira, J.C. de Almeida Junior, C.C.R. Viana, L.N. de Oliveira Neves, P.H.F. da Silva, M.J.V. Bell, V. de Carvalhodos Anjos, FTIR-ATR determination of protein content to evaluate whey protein concentrate adulteration. LWT 99, 166–172 (2019). https://doi.org/10.1016/j.lwt.2018.09.079
J. Müller-Maatsch, M. Alewijn, M. Wijtten, Y. Weesepoel, Detecting fraudulent additions in skimmed milk powder using a portble, hyphenated, optical multi-sensor approach in combination with one-class classification. Food Control 121, 107744 (2021). https://doi.org/10.1016/j.foodcont.2020.107744
M. Masci, C. Zoani, T. Nevigato, A. Turrini, R. Jasionowska, R. Caproni, P. Ratini, Authenticity assessment of dairy products by capillary electrophoresis. Electrophoresis 43, 340–354 (2022). https://doi.org/10.1002/elps.202100154
M. Esteki, Z. Shahsavari, J. Simal-Gandara, Food identification by high performance liquid chromatography fingerprinting and mathematical processing. Food Res. Int. 122, 303–317 (2019). https://doi.org/10.1016/j.foodres.2019.04.025
N.A. van Huizen, J.N.M. Ijzermans, P.C. Burgers, T.M. Luider, Collagen analysis with mass spectrometry. Mass Spectrom. Rev. 39, 309–335 (2020). https://doi.org/10.1002/mas.21600
L. Yuan, B. Liu, K. Yin, Z.-L. Xu, Development of an enzyme-linked immunosorbent assay for quantification of estriol in milk. Food Agric. Immunol. 30, 817–828 (2019). https://doi.org/10.1080/09540105.2019.1637824
R. Sun, J.-Y. Zhou, D. Yu, Nondestructive prediction model of internal hardness attribute of fig fruit using NIR spectroscopy and RF. Multimed. Tools Appl. 80, 21579–21594 (2021). https://doi.org/10.1007/s11042-021-10777-4
Y. Wu, X. Li, L. Xu, R. Fan, Y. Lin, C. Zhan, Z. Kang, Counterfeit detection of bulk Baijiu based on fluorescence hyperspectral technology and machine learning. J. Food Meas. Charact. (2024). https://doi.org/10.1007/s11694-024-02384-2
H. Yan, S. Jie, Z. Chunyi, H. Peng, K. Zhiliang, Identification and quantification of adulterated Tieguanyin based on the fluorescence hyperspectral image technique. J. Food Compos. Anal. 120, 105343 (2023). https://doi.org/10.1016/j.jfca.2023.105343
Z. Zou, Q. Wu, T. Long, B. Zou, M. Zhou, Y. Wang, B. Liu, J. Luo, S. Yin, Y. Zhao, L. Xu, Classification and adulteration of mengding mountain green tea varieties based on fluorescence hyperspectral image method. J. Food Compos. Anal. 117, 105141 (2023). https://doi.org/10.1016/j.jfca.2023.105141
J. Hao, F. Dong, Y. Li, S. Wang, J. Cui, S. Liu, Y. Lv, Quantification of polycyclic arom-atic hydrocarbons in roasted Tan lamb using fluorescence hyperspectral imaging technology. J. Food Compos. Anal. 124, 105646 (2023). https://doi.org/10.1016/j.jfca.2023.105646
H. Jiang, X. Jiang, Y. Ru, Q. Chen, J. Wang, L. Xu, H. Zhou, Detection and visualizationof soybean protein powder in ground beef using visible and near-infrared hyperspectral imaging. Infrared Phys. Technol. 127, 104401 (2022). https://doi.org/10.1016/j.infrared.2022.104401
M. De Géa Neves, R.J. Poppi, M.C. Breitkreitz, Authentication of plant-based protein powders and classification of adulterants as whey, soy protein, and wheat using FT-NIR in tandem with OC-PLS and PLS-DA models. Food Control 132, 108489 (2022). https://doi.org/10.1016/j.foodcont.2021.108489
Y. Hu, L. Xu, P. Huang, J. Sun, Y. Wu, J. Geng, R. Fan, Z. Kang, Non-destructive detection of Tieguanyin adulteration based on fluorescence hyperspectral technique. J. Food Meas. Charact. 17, 2614–2622 (2023). https://doi.org/10.1007/s11694-023-01817-8
M. Landauskas, Z. Navickas, A. Vainoras, M. Ragulskis, Weighted moving averaging revisited: an algebraic approach. Comput. Appl. Math. 36, 1545–1558 (2017). https://doi.org/10.1007/s40314-016-0309-9
J. Steinier, Y. Termonia, J. Deltour, Smoothing and differentiation of data by simplified least square procedure. Anal. Chem. 44, 1906–1909 (1972). https://doi.org/10.1021/ac60319a045
J.L. Ilari, H. Martens, T. Isaksson, Determination of particle size in powders by scatter correction in diffuse near-infrared reflectance. Appl. Spectrosc. 42, 722–728 (1988). https://doi.org/10.1366/0003702884429058
S. Marukatat, Tutorial on PCA and approximate PCA and approximate kernel PCA. Artif. Intell. Rev. 56, 5445–5477 (2023). https://doi.org/10.1007/s10462-022-10297-z
Z. Zou, Q. Wu, J. Wang, I. Xu, M. Zhou, Z. Lu, Y. He, Y. Wang, B. Liu, Y. Zhao, Research on non-destructive testing of hotpot oil quality by fluorescence hyperspectral techn-ology combined with machine learning. Spectroc. Acta Pt. A-Molec. Biomolec. Spectr. 284, 121785 (2023). https://doi.org/10.1016/j.saa.2022.121785
M.J. Hasan, J.-M. Kim, Fault detection of a spherical tank using a genetic algorithm-based hybrid feature pool and k-nearest neighbor algorithm. Energies (2019). https://doi.org/10.3390/en12060991
X. Sun, J. Li, Y. Shen, W. Li, Non-destructive detection of insect foreign bodies in finishing tea product based on terahertz spectrum and image. Front. Nutr. (2021). https://doi.org/10.3389/fnut.2021.757491
X. Zhang, J. Sun, P. Li, F. Zeng, H. Wang, Hyperspectral detection of salted sea cucumberadulteration using different spectral preprocessing techniques and SVM method. LWT 152, 112295 (2021). https://doi.org/10.1016/j.lwt.2021.112295
X. Wei, D. Kong, S. Zhu, S. Li, S. Zhou, W. Wu, Rapid identification of soybean varieties by terahertz frequency-domain spectroscopy and grey wolf optimizer-support vector machine. Front. Plant Sci. (2022). https://doi.org/10.3389/fpls.2022.823865
H.F. Jerome, Greedy function approximation: a gradient boosting machine. Ann. Stat. 29, 1189–1232 (2001). https://doi.org/10.1214/aos/1013203451
X.-Z. Wang, H.-L. Wu, T. Wang, A.-Q. Chen, H.-B. Sun, Z.-W. Ding, H.-Y. Chang, R.-Q. Yu, Rapid identification and semi-quantification of adulteration in walnut oil by using excitation–emission matrix fluorescence spectroscopy coupled with chemometrics and ensemble learning. J. Food Compos. Anal. 117, 105094 (2023). https://doi.org/10.1016/j.jfca.2022.105094
A. Amjad, R. Ullah, S. Khan, M. Bilal, A. Khan, Raman spectroscopy based analysis of milk using random forest classification. Vib. Spectrosc. 99, 124–129 (2018). https://doi.org/10.1016/j.vibspec.2018.09.003
J. Fernández-Habas, M. Carriere Cañada, A.M. García Moreno, J.R. Leal-Murillo, M.P. González-Dugo, B. Abellanas Oar, P.J. Gómez-Giráldez, P. Fernández-Rebollo, Estimating pasture quality of Mediterranean grasslands using hyperspectral narrow bands from field spectroscopy by Random Forest and PLS regressions. Comput. Electron. Agric. (2022). https://doi.org/10.1016/j.compag.2021.106614
A.D. Vibhute, K.V. Kale, S.C. Mehrotra, R.K. Dhumal, A.D. Nagne, Determination of soil physicochemical attributes in farming sites through visible, near-infrared diffuse reflectance spectroscopy and PLSR modeling. Ecol. Process. 7, 26 (2018). https://doi.org/10.1186/s13717-018-0138-4
V.G. Kelis Cardoso, R.J. Poppi, Cleaner and faster method to detect adulteration in cassavastarch using Raman spectroscopy and one-class support vector machine. Food Control 125, 107917 (2021). https://doi.org/10.1016/j.foodcont.2021.107917
Ł Saletnik, W. Szczęsny, J. Szmytkowski, J.J. Fisz, On the nature of stationary and time-resolved fluorescence spectroscopy of collagen powder from bovine achilles tendon. Int. J. Mol. Sci. (2023). https://doi.org/10.3390/ijms24087631
Funding
This work received support from the subject double support program of Sichuan Agricultural University (Grant No. 035-1921993093).
Author information
Authors and Affiliations
Contributions
Yi Lin: Conceptualization, Resources, Formal analysis, Roles/Writing–original draft, Data curation, Project administration, Investigation, Methodology. Youli Wu: Software, Data analysis, Equipment commissioning, Supervision, Validation. Rongsheng Fan: Methodology, Supervision, Validation. Chunyi Zhan: Supervision, Validation. Zhiliang Kang: Funding acquisition, Review, Editing, Supervision, Validation.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, Y., Wu, Y., Fan, R. et al. Identification and quantification of adulterated collagen powder by fluorescence hyperspectral technology. Food Measure (2024). https://doi.org/10.1007/s11694-024-02577-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11694-024-02577-9