Abstract
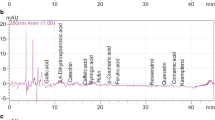
Antioxidant activity (AA), total polyphenols content (TPC) and polyphenols profile (PP) were investigated during 10 days of postharvest ripening, in peels and pulps from fruits from three Carica papaya genotypes of different origin. Wild-genotype (WG) collected in Yucatan, Mexico (part of its center of origin) showed higher AA and TPC values than the commercial-genotype (CG). Likewise, PP analysis resulted in the identification and quantification of 4 families: phenolic acids, flavanols, dihydrochalcones and flavonols in both peels and pulps, as well as lignans, that were only found in fruit peels. Interestingly, fruits from WG also had higher contents than CG, in the following specific polyphenols: in peels; 5-O-Caffeoylquinic acid (chlorogenic acid) and kaempferol (both absent in CG), 4-Hydroxybenzoic acid 4-O-glucoside (3.8 times), quercetin 3-O-hexoside (3.5 times), 4-O-Caffeoylquinic acid (2.8 times), 5-O-Galloylquinic acid (2.7 times) and matairesinol (2.6 times). In pulps; 4-O-Caffeoylquinic acid (9.4 times), quercetin-3-O-rutinoside (rutin; 6.8 times), p-Coumaric acid 4-O-glucoside (p-coumaroyl hexoside; 2.5 times), p-Coumaroyl glycolic acid (2.5 times) and phloridzin (1.7 times). The superior capacity of wild native genotypes to accumulate more total polyphenols, to show higher antioxidant activity and to show even higher contents of some specific polyphenols known to have beneficial health effects (against chronic degenerative diseases such as cancer and cardiovascular diseases), might represent a good base for breeding programs aiming to obtain new varieties with superior nutraceutical properties.





Similar content being viewed by others
Abbreviations
- AA:
-
Antioxidant activity
- ABTS:
-
2,2′-Azinobis-3-ethylbenzotiazoline-6-sulphonic-acid
- C. papaya :
-
Carica papaya
- CG:
-
Commercial-genotype
- CrG:
-
Creole-genotype
- DPPH:
-
2,2′-Diphenyl-1-116 picrylhydrazyl
- DW:
-
Dry weight
- FW:
-
Fresh weight
- G:
-
Green stage
- GA:
-
Gallic acid
- GAE:
-
Gallic acid equivalents
- PP:
-
Polyphenols profile
- RP:
-
Reducing power
- TE:
-
Trolox equivalents
- TPC:
-
Total polyphenol content
- UPLC:
-
Ultra-performance liquid chromatography
- WG:
-
Wild-genotype
References
C. Santos-Buelga, A. Scalbert, Proanthocyanidins and tannin-like compounds—nature, occurrence, dietary intake and effects on nutrition and health. J. Sci. Food Agric. 80, 1094–1117 (2000). https://doi.org/10.1002/(SICI)1097-0010(20000515)80:7%3c1094::AID-JSFA569%3e3.0.CO;2-1
D. Del Rio, A. Rodriguez-Mateos, J.P. Spencer, M. Tognolini, G. Borges, A. Crozier, Dietary (Poly) phenolics in human health: structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal 18(14), 1818–1892 (2012). https://doi.org/10.1089/ars.2012.4581
I. Urquiaga, F. Leighton, Plant polyphenol antioxidants and oxidative stress. Biol. Res. 33, 55–64 (2000). https://doi.org/10.4067/s0716-97602000000200004
M. La Marca, P. Beffy, A. Pugliese, V. Longo, Fermented wheat powder induces the antioxidant and detoxifying system in primary rat hepatocytes. PLoS ONE 8(12), e83538 (2013). https://doi.org/10.1371/journal.pone.0083538
A. Scalbert, C. Manach, C. Morand, C. Rémésy, L. Jiménez, Dietary polyphenols and the prevention of diseases. Crit. Rev. Food Sci. Nutr. 45(4), 287–306 (2005). https://doi.org/10.1080/1040869059096
O.R. Alara, N.H. Abdurahman, J.A. Alara, Carica papaya: comprehensive overview of the nutritional values, phytochemicals and pharmacological activities. Adv. Tradit. Med. (2020). https://doi.org/10.1007/s13596-020-00481-3
E.H.K. Ikram, R. Stanley, M. Netzel, K. Fanning, Phytochemicals of papaya and its traditional health and culinary uses—a review. J. Food Compos. Anal. 41, 201–211 (2015). https://doi.org/10.1016/j.jfca.2015.02.010
Q.V. Vuong, S. Hirun, P.D. Roach, M.C. Bowyer, P.A. Phillips, C.J. Scarlett, Effect of extraction conditions on total phenolic compounds and antioxidant activities of Carica papaya leaf aqueous extracts. J. Herb. Med. 3(3), 104–111 (2013). https://doi.org/10.1016/j.hermed.2013.04.004
K. Zhou, H. Wang, W. Mei, X. Li, Y. Luo, H. Dai, Antioxidant activity of papaya seed extracts. Molecules 16(8), 6179–6192 (2011). https://doi.org/10.3390/molecules16086179
L.E. Gayosso-García Sancho, E.M. Yahia, G.A. González-Aguilar, Identification and quantification of phenols, carotenoids, and vitamin C from papaya (Carica papaya L., cv. Maradol) fruit determined by HPLC-DAD-MS/MS-ESI. Food Res. Int. 44(5), 1284–1291 (2011). https://doi.org/10.1016/j.foodres.2010.12.001
D.M. Rivera-Pastrana, E.M. Yahia, G.A. González-Aguilar, Phenolic and carotenoid profiles of papaya fruit (Carica papaya L.) and their contents under low temperature storage. J. Sci. Food Agric. 90(14), 2358–2365 (2010). https://doi.org/10.1002/jsfa.4092
T.J. O’Hare, D.J. Williams, Papaya as a medicinal plant, in Genetics and Genomics of Papaya: Crops and Models. ed. by R. Ming, P.H. Moore (Springer, New York, 2014), pp. 391–407
A. Hee-Young, C. Hyun-Dong, C. Young-Su, Comparison of antioxidant effect and phenolic compounds in tropical fruits. SN Appl. Sci. 2, 1120 (2020). https://doi.org/10.1007/s42452-020-2927-5
G. Fuentes, J. Santamaría, Papaya (Carica papaya L.): origin, domestication, and production, in Genetics and Genomics of Papaya: Crops and Models. ed. by R. Ming, P.H. Moore (Springer, New York, 2014), pp. 3–15
U. Tiwari, E. Cummins, Factors influencing levels of phytochemicals in selected fruit and vegetables during pre- and post-harvest food processing operations. Food Res. Int. 50(2), 497–506 (2013). https://doi.org/10.1016/j.foodres.2011.09.007
W. Brand-Williams, M.E. Cuvelier, C. Berset, Use of a free radical method to evaluate antioxidant activity. Lebensm-Wiss Technol. 28(1), 25–30 (1995). https://doi.org/10.1016/S0023-6438(95)80008-5
N.J. Miller, C. Rice-Evans, M.J. Davies, V. Gopinathan, A. Milner, A novel method for measuring antioxidant capacity and its application to monitoring the antioxidant status in premature neonates. Clin. Sci. 84(4), 407–407 (1993). https://doi.org/10.1042/cs0840407
M. Oyaizu, Studies on products of browning reaction: antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. J. Nutr. Diet. 44(6), 307–315 (1986)
L.E. Gayosso-García Sancho, E.M. Yahia, M.A. Martínez-Téllez, G.A. González-Aguilar, Effect of maturity stage of papaya maradol on physiological and biochemical parameters. Am. J. Agric. Biol. Sci. 5(2), 194–203 (2010). https://doi.org/10.3844/ajabssp.2010.194.203
L. Fu, B.-T. Xu, X.-R. Xu, R.-Y. Gan, Y. Zhang, E.-Q. Xia, H.-B. Li, Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 129(2), 345–350 (2013). https://doi.org/10.1016/j.foodchem.2011.04.079
R. Rajamurugan, N. Selvaganabathy, S. Kumaravel, C.H. Ramamurthy, V. Sujatha, C. Thirunavukkarasu, Polyphenol contents and antioxidant activity of Brassica nigra (L.) Koch. leaf extract. Nat. Prod. Res. 26(23), 2208–2210 (2012). https://doi.org/10.1080/14786419.2011.637215
H.V. Annegowda, R. Bhat, K.J. Yeong, M.T. Liong, A.A. Karim, S.M. Mansor, Influence of drying treatments on polyphenolic contents and antioxidant properties of raw and ripe papaya (Carica papaya L.). Int. J. Food Prop. 17(2), 283–292 (2014). https://doi.org/10.1080/10942912.2011.631248
M.M.B. Almeida, P.E.M. de Sousa, A.M.C. Arriaga, G.M. do Prado, C.E.C. Magalhães, G.A. Maia, T.L.G. de Lemos, Bioactive compounds and antioxidant activity of fresh exotic fruits from northeastern Brazil. Food Res. Int. 44(7), 2155–2159 (2011). https://doi.org/10.1016/j.foodres.2011.03.051
J. Gruz, F.A. Ayaz, H. Torun, M. Strnad, Phenolic acid content and radical scavenging activity of extracts from medlar (Mespilus germanica L.) fruit at different stages of ripening. Food Chem. 124(1), 271–277 (2011). https://doi.org/10.1016/j.foodchem.2010.06.030
C.M.B. Omena, I.B. Valentim, G.D.S. Guedes, L.A. Rabelo, C.M. Mano, E.J.H. Bechara, A.C.H.F. Sawaya, M.T.S. Trevisan, J.G. da Costa, R.C.S. Ferreira, A.E.G. SantˈAna, M.O.F. Goulart, Antioxidant, anti-acetylcholinesterase and cytotoxic activities of ethanol extracts of peel, pulp and seeds of exotic Brazilian fruits: antioxidant, anti-acetylcholinesterase and cytotoxic activities in fruits. Food Res. Int. 49(1), 334–344 (2012). https://doi.org/10.1016/j.foodres.2012.07.010
E.M. Kuskoski, A.G. Asuero, M.T. Morales, R. Fett, Frutos tropicais silvestres e polpas de frutas congeladas: atividade antioxidante, polifenóis e antocianinas. Ciênc. Rural 36(4), 1283–1287 (2006). https://doi.org/10.1590/S0103-84782006000400037
F. Saidani, R. Giménez, C. Aubert, G. Chalot, J.A. Betrán, Y. Gogorcena, Phenolic, sugar and acid profiles and the antioxidant composition in the peel and pulp of peach fruits. J. Food Compos. Anal. 62(3), 126–133 (2017). https://doi.org/10.3390/ijms16035762
D.R. Morais, E.M. Rotta, S.C. Sargi, E.M. Schmidt, E.G. Bonafe, M.N. Eberlin, A.C. Sawaya, J.V. Visentainer, Antioxidant activity, phenolics and UPLC-ESI(-)-MS of extracts from different tropical fruits parts and processed peels. Food Res. Int. 77(3), 392–399 (2015). https://doi.org/10.1016/j.foodres.2015.08.036
A.Y. Coulibaly, R. Hashim, S.F. Sulaiman, O. Sulaiman, L.Z.P. Ang, K.L. Ooi, Bioprospecting medicinal plants for antioxidant components. Asian Pac. J. Trop. Med. 7, S553–S559 (2014). https://doi.org/10.1016/S1995-7645(14)60289-3
C.J. Nieto, M. Cueto, A. Farroni, M. Pla, L.N. Gerschenson, Antioxidant characterization of new dietary fiber concentrates from papaya pulp and peel (Carica papaya L.). J. Funct. Foods 27, 319–328 (2016). https://doi.org/10.1016/j.jff.2016.09.012
V. Zunjar, D. Mammen, B.M. Trivedi, Antioxidant activities and phenolics profiling of different parts of Carica papaya by LCMS-MS. Nat. Prod. Res. 29(22), 2097–2099 (2015). https://doi.org/10.1080/14786419.2014.986658
D. Kitts, Y. Yuan, A. Wijewickreme, L. Thompson, Antioxidative activity of the flaxseed lignan secosiolariciresinol diglycoside and its mammalian lignan metabolites enterodiol and enterolactone. Mol. Cell. Biochem. 202(1–2), 91–100 (1999). https://doi.org/10.1023/a:1007022329660
U.A. Fischer, A.V. Jaksch, R. Carle, D.R. Kammerer, Determination of lignans in edible and nonedible parts of pomegranate (Punica granatum L.) and products derived therefrom, particularly focusing on the quantitation of isolariciresinol using HPLC-DAD-ESI/MSn. J. Agric. Food Chem. 60(1), 283–292 (2012). https://doi.org/10.1021/jf203598m
P. Johnsson, N. Peerlkamp, A. Kamal-Eldin, R.E. Andersson, R. Andersson, L.N. Lundgren, P. Aman, Polymeric fractions containing phenol glucosides in flaxseed. Food Chem. 76(2), 207–212 (2002). https://doi.org/10.1016/S0308-8146(01)00269-2
J.M. Harnly, R.F. Doherty, G.R. Beecher, J.M. Holden, D.B. Haytowitz, S. Bhagwat, S. Gebhardt, Flavonoid content of U.S. fruits, vegetables, and nuts. J. Agric. Food Chem. 54(26), 9966–9977 (2006). https://doi.org/10.1021/jf061478a
I.C. Arts, B. Van de Putte, P.C. Hollman, Catechin contents of foods commonly consumed in The Netherlands. 1. Fruits, vegetables, staple foods, and processed foods. J. Agric. Food Chem. 48(5), 1746–1751 (2000). https://doi.org/10.1021/jf000025h
M.P. Marinovic, A.C. Morandi, R. Otton, Green tea catechins alone or in combination alter functional parameters of human neutrophils via suppressing the activation of TLR-4/NFκB p65 signal pathway. Toxicol. In Vitro 29(7), 1766–1778 (2015). https://doi.org/10.1016/j.tiv.2015.07.014
L. Bai, S. Guo, Q. Liu, X. Cui, X. Zhang, L. Zhang, X. Yang, M. Hou, H. Chi-Tang, N. Bai, Characterization of nine polyphenols in fruits of Malus pumila mill by high-performance liquid chromatography. J. Food Drug Anal. 24(2), 293–298 (2016). https://doi.org/10.1016/j.jfda.2015.10.002
S. Kandakumar, V. Manju, Pharmacological applications of isorhamnetin: a short review. IJTSRD 1(4), 672–678 (2017)
J. Lako, C.V. Trenerry, M. Wahlqvist, N. Wattanapenpaiboon, S. Sotheeswaran, R. Premier, Phytochemical flavonols, carotenoids and the antioxidant properties of a wide selection of Fijian fruit, vegetables and other readily available foods. Food Chem. 101(4), 1727–1741 (2007). https://doi.org/10.1016/j.foodchem.2006.01.031
Acknowledgements
To CONACYT for the Grant No. CB221208 given to JSF and the PhD Scholarship No. 362319 given to ACL. To Mr. José Arjona for donating the commercial fruits used in the present study. To Canadian Bureau for International Education (CBIE) as well as Foreign Affairs and International Trade Canada (DFAIT) for having awarded ACL the Emerging Leaders in the Americas Program (ELAP) scholarship. We also acknowledge the use of the lab facilities at INAF, Quebec Canada.
Author information
Authors and Affiliations
Contributions
AC-L, performed the experiments, wrote the first draft of the manuscript. HE-M, GFO, VM-H, CC-C, ES-D, conceived, designed the experiments, analyzed data and reviewed the manuscript. PD, YD, designed the PP experiments and contribute to the writing of the manuscript. JMS, corresponding author, general conception of the project and responsible for writing the final manuscript. All authors have read the final manuscript and approved its submission.
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest among the author and coauthors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chan-León, A., Estrella-Maldonado, H., Dubé, P. et al. Determination of total phenolic contents and antioxidant activities of fruits from wild and creole Carica papaya genotypes in comparison to commercial papaya cultivars. Food Measure 15, 5669–5682 (2021). https://doi.org/10.1007/s11694-021-01121-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-021-01121-3