Abstract
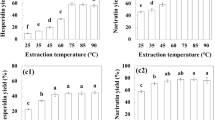
This study aimed to evaluate bioactivity related properties of wild mustard parts (WMP) including the leaves, flowers, stems, fruits, and seeds. The biomass and ion composition of WMP were analyzed by elemental analyzer and ion chromatography, respectively. Extraction conditions of phenolics were optimized with Response Surface Methodology by taking into consideration 3 factors such as the ratio of ethanol to water, temperature, and time. LC–ESI–MS/MS was used for the identification of phenolics in WMP. The principal phenolic compounds in the leaves, flowers and seeds of wild mustard were kaempferol, hydroxycinnamic acid, and catechin, respectively, whereas vanillic acid was dominant in the stems and fruits. The antioxidant capacities of WMP were 1.88–15.56, 309.61–775.63, 25.46–87.24, and 193.58–617.08 µmol TE/g for DPPH, ABTS, FRAP, and CUPRAC, respectively. Antioxidant capacity of the flowers was in all cases superior to the other parts irrespectively of the antioxidant capacity method being used. Therefore, the flower extract was transformed into powder in order to determine the potentiality of it as an ingredient for the enrichment of food formulations. Powdered flower extract (PFE) was incorporated into ice cream at the levels of 0.3–0.9%. The sensory scores indicated that PFE had almost no effects on texture related properties of ice cream, whereas overall acceptability of PFE enriched ice creams decreased as the amount of PFE increased. PFE also showed antimicrobial activity on selected food-borne pathogens, most notably on Staphylococcus haemolyticus. Wild mustard can be evaluated as a source of natural phenolic antioxidant compounds in different industries.


Similar content being viewed by others
References
J. Xiang, F.B. Apea-Bah, V.U. Ndolo, M.C. Katundu, T. Beta, Food Chem. 275, 361–368 (2019)
C.G. Pereira, L. Barreira, N. da Rosa Neng, J.M.F. Nogueira, C. Marques, T.F. Santos, J. Varela, L. Custódio, Food Chem. Toxicol. 107, 581–589 (2017)
K. Muniyandi, E. George, S. Sathyanarayanan, B.P. George, H. Abrahamse, S. Thamburaj, P. Thangaraj, Food Sci. Hum. Wellness 8, 73–81 (2019)
Z. Derakhshan, M. Ferrante, M. Tadi, F. Ansari, A. Heydari, M.S. Hosseini, G.O. Conti, E.K. Sadrabad, Food Chem. Toxicol. 114, 108–111 (2018)
I. Karaman, F. Şahin, M. Güllüce, H. Öǧütçü, M. Şengül, A. Adıgüzel, J. Ethnopharmacol. 85, 231–235 (2003)
N. Al-Zoreky, Int. J. Food Microbiol. 134, 244–248 (2009)
F. Shahbazi, J. Sci. Food Agric. 93, 1466–1470 (2013)
K. Dhima, I. Eleftherohorinos, J. Agron. Crop Sci. 191, 241–248 (2005)
Q. Mushtaq, M. Irfan, F. Tabssum, J. Iqbal Qazi, J. Food Process Eng. 40, e12512 (2017)
S.K. Panda, S.S. Mishra, E. Kayitesi, R.C. Ray, Environ. Res. 146, 161–172 (2016)
N.A. Sagar, S. Pareek, S. Sharma, E.M. Yahia, M.G. Lobo, Compr. Rev. Food Sci. F. 17, 512–531 (2018)
S.-H. Choi, N. Kozukue, H.-J. Kim, M. Friedman, J. Food Compos. Anal. 50, 77–87 (2016)
G.X. Paula Filho, T.F. Barreira, R.H. Santos, S.E. Priore, C.M. Della Lucia, H.M. Pinheiro-Sant’ Ana, Hortic. Bras. 36, 59–65 (2018)
V.L. Singleton, J.A. Rossi, Am. J. Enol. Vitic. 16, 144–158 (1965)
J. Zhishen, T. Mengcheng, W. Jianming, Food Chem. 64, 555–559 (1999)
R. Willis, Analyst 123, 435–439 (1998)
M. Çam, Y. Hışıl, G. Durmaz, Food Chem. 112, 721–726 (2009)
R. Apak, K. Güclü, M. Özyürek, S.E. Celik, Microchim. Acta. 160, 413–419 (2008)
I.F. Benzie, J.J. Strain, Anal. Biochem. 239, 70–76 (1996)
G.J. McDougall, F. Shpiro, P. Dobson, P. Smith, A. Blake, D. Stewart, J. Agric, Food Chem. 53, 2760–2766 (2005)
M. Çam, M.D. Işıklı, E. Yüksel, H. Alaşalvar, B. Başyiğit, J. Food Meas. Charact. 12, 1927–1934 (2018)
M. Çam, F. Erdoğan, D. Aslan, M. Dinç, J. Food Sci. 78, C1543–C1550 (2013)
F. Munalula, M. Meincken, Biomass Bioenergy. 33, 415–420 (2009)
C. Yılmaz, V. Gökmen, Ind. Crop Prod. 49, 130–135 (2013)
N.S. Reddy, S. Navanesan, S.K. Sinniah, N.A. Wahab, K.S. Sim, BMC Complement Altern. Med. 12, 128 (2012)
K. Reilly, J. Valverde, L. Finn, D.K. Rai, N. Brunton, J.C. Sorensen, H. Sorensen, M. Gaffney, J. Sci. Food Agric. 94, 322–330 (2014)
L. Giorgetti, G. Giorgi, E. Cherubini, P.G. Gervasi, C.M. Della Croce, V. Longo, L. Bellani, Nat. Prod. Res. 32, 1617–1626 (2018)
T. Mosha, H. Gaga, R. Pace, H. Laswai, K. Mtebe, Plant Food Hum. Nutr. 47, 361–367 (1995)
M.R. Loizzo, R. Tundis, M. Bonesi, F. Menichini, V. Mastellone, L. Avallone, F. Menichini, J. Food Compos. Anal. 25, 179–184 (2012)
G. Luisi, A. Stefanucci, G. Zengin, M. Dimmito, A. Mollica, Antioxidants 8, 7 (2019)
T. Sotelo, M.E. Cartea, P. Velasco, P. Soengas, PLoS ONE 9, e107290 (2014)
Q. Shen, J. Jiang, M. Wang, J. Chen, D. Liu, X. Ye, Y. Hu, J. Food Process. Preserv. 43, e13918 (2019)
S.H. Jo, C.Y. Cho, K.S. Ha, J.Y. Lee, H.Y. Choi, Y.I. Kwon, E. Apostolidis, J. Food Biochem. 42, e12583 (2018)
I. Koss-Mikołajczyk, B. Kusznierewicz, W. Wiczkowski, N. Płatosz, A. Bartoszek, J. Sci. Food Agric. 99, 5499–5507 (2019)
A. Bouhafsoun, M.A. Yilmaz, A. Boukeloua, H. Temel, M.K. Harche, Food. Sci. Technol. 38, 242–247 (2018)
J.P. Spencer, A. Crozier, Flavonoids and related compounds: bioavailability and function (CRC Press, Boca Raton, 2012)
R.K. Goraya, U. Bajwa, J. Food Sci. Technol. 52, 7861–7871 (2015)
S. Karaman, A. Kayacier, Food Bioprocess Technol. 5, 3159–3169 (2012)
Acknowledgements
This work was financially supported by Erciyes University Scientific Research Projects Unit (Project No: FBA-2017-6826).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Başyiğit, B., Alaşalvar, H., Doğan, N. et al. Wild mustard (Sinapis arvensis) parts: compositional analysis, antioxidant capacity and determination of individual phenolic fractions by LC–ESI–MS/MS. Food Measure 14, 1671–1681 (2020). https://doi.org/10.1007/s11694-020-00415-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-020-00415-2