Abstract
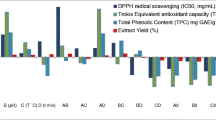
In this study, by applying response surface methodology (RSM) maceration conditions including enzyme concentration (A), incubation time (B), temperature (C) and pH were optimized for the liberation of Cassia fistula pod phenolics (CFPP) along with their retained antioxidant character. Briefly, mature Cassia fistula pods (CFP) were macerated under various conditions: A: 3–5%, B: 60–90 min, C: 40–70 °C and D: 5.5–8.5 and extract obtained thus were simultaneously evaluated for their total phenolic content (TPC), free radical scavenging capacity (FRSC), Trolox equivalent antioxidant capacity (TEAC), inhibition of linoleic acid peroxidation (ILP) and ferric reducing power (FRP). The solution having highest desirability endorsed treatment of CFP with 3.40% acid cellulose enzyme at 55.11 °C and 6.1 pH for 75 min, which offered an extract yield 31.66 g/100 g containing 165.63 milligram gallic acid equivalents TPC/g of CFP with appreciable level of FRSC (DPPH IC50 6.50 mg/mL), TEAC (282.64 mg TE/g of extract), ILP (55.61%), and FRP of 0.68.. Exclusively it could be acclaimed that a carefully optimized enzyme pre-treatment may furnish good quantity and quality of polyphenols.

Similar content being viewed by others
References
H.A. Deshpande, S.R. Bhalsing, Recent advances in the phytochemistry of some medicinally important Cassia species: a review. Int. J. Pharm. Med. Biol. 2, 60–78 (2013)
R. Gupta, Medicinal and Aromatic Plants (CBS Publisher and Distribution, Daryaganj, 2010), pp. 234–267
C.P. Khare, Indian Medicinal Plants: An Illustrated Dictionary. (Springer, New York, 2008), pp. 220–240
K. Kirtikar, B. Basu, Indian Medicinal Plant, 4th edn. (Inter Book Distribution, Dehra Dun, 2006), pp. 1466–1467
R. Ros-Zamora, M. Rabassa, A. Cherubini, M. Urpi-Sarda, S. Bandinelli, L. Ferrucci, C.A. Lacueva, High concentrations of a urinary biomarker of polyphenol intake are associated with decreased mortality in older adults. J. Nutr. 143, 1445–1450 (2013)
N. Azwanida, A review on the extraction methods use in medicinal plants, principle, strength, and limitation. Med. Aromat. Plants. (2015). https://doi.org/10.1007/s11694-017-9685-0
M. Contini, S. Baccelloni, R. Massantini, G. Anelli, A review on the extraction methods use in medicinal plants, principle, strength and limitation. Food Chem. 110, 659–669 (2008)
J. Wang, B. Sun, Y. Cao, Y. Tian, X. Li, Optimization of ultrasound-assisted extraction of phenolic compounds from wheat bran. Food Chem. 106, 804–810 (2008)
M. Mushtaq, B. Sultana, S. Akram, F. Anwar, A. Adnan, S.S. Rizvi, Enzyme-assisted supercritical fluid extraction: an alternative and green technology for non-extractable polyphenols. Anal. Bioanal. Chem. 409, 3645–3655 (2017)
H. Abbasi, K. Rezaei, Z. Emamdjomeh, S.M.E. Mousavi, Effect of various extraction conditions on the phenolic contents of pomegranate seed oil. Eur. J. Lipid Sci. Technol. 110, 435–440 (2008)
M. Mushtaq, B. Sultana, H.N. Bhatti, M. Asghar, RSM based optimized enzyme-assisted extraction of antioxidant phenolics from underutilized watermelon (Citrullus lanatus Thunb.). J. Food Sci. Technol. 52, 5048–5056 (2014)
P.W. Araujo, R.G. Brereton, Experimental design 3 quantitation. Trends Anal. Chem. 15, 156–163 (1996)
T. Lundstedt, E. Seifert, L. Abramo, B. Thelin, A. Nystrom, J. Pettersen, R. Bergman, Experimental design and optimization. Chemom. Intell. Lab. Syst. 42, 3–40 (1998)
M.A. Bezerra, R.E. Santelli, E.P. Oliveira, L.S. Villar, L.A. Escaleira, Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 76, 965–977 (2008)
T.E. Murphy, K.L. Tsui, J.K. Allen, A review of robust design methods for multiple responses. Res. Eng. Des. 15, 201–215 (2005)
S.F. Sulaiman, N.A.M. Yusoff, I.M. Eldeen, E.M. Seow, A.A.B. Sajak, K.L. Ooi, Correlation between total phenolic and mineral contents with antioxidant activity of eight Malaysian bananas (Musa sp.). J. Food Compos. Anal. 24, 1–10 (2011)
Z. Chen, R. Bertin, G. Froldi, EC 50 estimation of antioxidant activity in DPPH assay using several statistical programs. Food Chem. 138, 414–420 (2013)
M.J. Arts, G.R. Haenen, H.P. Voss, A. Bas, Antioxidant capacity of reaction products limits the applicability of the trolox equivalent antioxidant capacity (TEAC) assay. Food Chem. Toxicol. 42, 45–49 (2004)
M. Oyaizu, Studies on products of browning reaction. Jpn. J. Nutr. Diet. 44, 307–315 (1986)
A.M. Alashi, C.L. Blanchard, R.J. Mailer, S.O. Agboola, A.J. Mawson, R. He, A. Girgih, R.E. Aluko, Antioxidant properties of Australian canola meal protein hydrolysates. Food Chem. 146, 500–506 (2014)
G. Derringer, R. Suich, Simultaneous optimization of several response variables. J. Qual. Technol. 12, 214–219 (1980)
K. Zhong, Q. Wang, Optimization of ultrasonic extraction of polysaccharides from dried longan pulp using response surface methodology. Carbohydr. Polym. 80, 19–25 (2010)
J.P. Maran, V. Mekala, S. Manikandan, Modeling and optimization of ultrasound-assisted extraction of polysaccharide from Cucurbita moschata. Polymers. 92, 2018–2026 (2013)
P. Siddhuraju, P.S. Mohan, K. Becker, Studies on the antioxidant activity of Indian Laburnum (Cassia fistula L.): a preliminary assessment of crude extracts from stem bark, leaves, flowers and fruit pulp. Food Chem. 79, 61–67 (2002)
M. Puri, D. Sharma, C.J. Barrow, Enzyme-assisted extraction of bioactives from plants. Trend Biotechnol. 30, 37–44 (2012)
T. Bahorun, V.S. Neergheen, O.I. Aruoma, Phytochemical constituents of Cassia fistula. Afr. J. Food Agric. Nutr. Dev. 4, 1530–1540 (2004)
E. Karacabey, G. Mazza, Optimisation of antioxidant activity of grape cane extracts using response surface methodology. Food Chem. 119, 343–348 (2010)
M. Serafini, G. Morabito, The Role of polyphenols in the modulation of plasma non-enzymatic antioxidant capacity (NEAC). Int. J. Vita. Nutr. Res. 82, 228–232 (2012)
W. Chanput, C. Theerakulkait, S. Nakai, Antioxidative properties of partially purified barley hordein, rice bran protein fractions and their hydrolysates. J. Cereal Sci. 49, 422–428 (2009)
Y. Zou, Y. Lu, D. Wei, Antioxidant activity of a flavonoid-rich extract of Hypericum perforatum L. in vitro. J. Agri. Food Chem. 52, 5032–5039 (2004)
A. Yıldırım, A. Mavi, M. Oktay, A.A. Kara, O.F. Algur, V. Bilaloglu, Comparison of antioxidant and antimicrobial activities of Tilia (Tilia argentea Desf ex DC), sage (Salvia triloba L.), and Black tea (Camellia sinensis) extracts. J. Agric. Food Chem. 48, 5030–5034 (2000)
M.A. Ebrahimzadeh, S.M. Nabav, S.F. Nabavi, F. Bahramian, A.R. Bekhradnia, Antioxidant and free radical scavenging activity of H. officinalis L. var. angustifolius, V. odorata, B. hyrcana. and C. speciosum. Pak. J. Pharm. Sci. 23, 29–34 (2010)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haider, W., Sultana, B., Mushtaq, M. et al. Multi-response optimization of enzyme-assisted maceration to enhance the yield and antioxidant activity of Cassia fistula pods extracts. Food Measure 12, 2685–2694 (2018). https://doi.org/10.1007/s11694-018-9886-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-018-9886-1