Abstract
Introduction
Cystic Echinococcosis (CE) is a chronic parasitic disease caused by the metacestodes of Echinococcus granulosus senso lato (E. granulosus s.l.). The larval stages of this parasite, hydatid cyst, are usually diagnosed using imaging modalities and serological testing; however, several studies have recently suggested using the parasite-derived microRNAs (miRNAs) as novel diagnostic biomarkers.
Materials and Methods
The present study included 31 CE patients who were older than 5 years and were admitted to the hospitals of Ahvaz, Iran for hydatid cyst removal surgery during 2019–2021. The egr-miR-125-5p and egr-miR-2a-3p levels were evaluated in the sera of the CE patients before and 6 months after the surgery using Quantitative Real-Time PCR (qRT-PCR), and the results were compared with the serum samples from 15 healthy volunteers. Then, the intergroup comparisons were performed using the t test.
Results
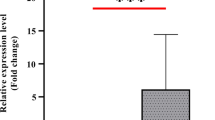
The patients’ age range was 6–72 years, with a mean age of 34.6 years. Moreover, based on the classification by the WHO-IWGE, one patient (3.2%) had CE1, 14 patients (45.2%) had CE2, 5 patients (16.1%) had CE3, 2 patients (6.5%) had CE4, and 9 patients (29%) had CE5. Also, 21 patients (67.74%) had a positive antigen test using the ELISA method, while 10 patients (32.26%) had a negative ELISA. The pre-operative expression level of egr-miR-2a-3p was 10.36 folds higher compared to 6 months after the surgery, with an AUC value of 0.8176. However, the expression levels of egr-miR-125-5p did not change significantly 6 months after the surgery compared to pre-operative levels.
Conclusions
According to the present study results, the serum levels of egr-miR-2a-3p can be a promising non-invasive biomarker for diagnosing CE and monitoring its potential recurrence after cystectomy.








Similar content being viewed by others
Data availability statement
All data generated or analyzed during the current study are included in the paper.
References
Mariconti M, Vola A, Manciulli T, Genco F, Lissandrin R, Meroni V et al (2019) Role of microRNAs in host defense against Echinococcus granulosus infection: a preliminary assessment. Immunol Res 67:93–97. https://doi.org/10.1007/s12026-018-9041-4
WHO Informal Working Group (2003) International classification of ultrasound images in cystic echinococcosis for application in clinical and field epidemiological settings. Acta Trop 85:253–261. https://doi.org/10.1016/s0001-706x(02)00223-1
Brunetti E, Kern P, Vuitton DA (2010) Expert consensus for the diagnosis and treatment of cystic and alveolar echinococcosis in humans. Acta Trop 114:1–16. https://doi.org/10.1016/j.actatropica.2009.11.001
Sharma K, Sharma R, Tiwari T, Goyal S (2022) Contrast CT evaluation of an isolated renal hydatid cyst. BMJ Case Reports 15:e248006. https://doi.org/10.1136/bcr-2021-248006
Zhang W, Wen H, Li J, Lin R, McManus DP (2012) Immunology and immunodiagnosis of cystic echinococcosis: an update. Clin Dev Immunol 2012:101895. https://doi.org/10.1155/2012/101895
Hernández-González A, Santivañez S, García HH, Rodríguez S, Muñoz S, Ramos G et al (2012) Improved serodiagnosis of cystic echinococcosis using the new recombinant 2B2t antigen. PLoS Negl Trop Dis 6:e1714-e. https://doi.org/10.1371/journal.pntd.0001714
Díaz Á (2017) Immunology of cystic echinococcosis (hydatid disease). Br Med Bull 124:121–133. https://doi.org/10.1093/bmb/ldx033
Alizadeh Z, Mahami-Oskouei M, Spotin A, Kazemi T, Ahmadpour E, Cai P et al (2020) Parasite-derived microRNAs in plasma as novel promising biomarkers for the early detection of hydatid cyst infection and post-surgery follow-up. Acta trop 202:105255. https://doi.org/10.1016/j.actatropica.2019.105255
Örsten S, Baysal İ, Yabanoglu-Ciftci S, Ciftci T, Ünal E, Akıncı D et al (2022) Can parasite-derived microRNAs differentiate active and inactive cystic echinococcosis patients? Parasitol Res 121:191–196. https://doi.org/10.1007/s00436-021-07382-7
Cabral BCA, Hoffmann L, Bottaro T, Costa PF, Ramos ALA, Coelho HSM et al (2020) Circulating microRNAs associated with liver fibrosis in chronic hepatitis C patients. Biochem Biophys Rep 24:100814. https://doi.org/10.1016/j.bbrep.2020.100814
Silakit R, Loilome W, Yongvanit P, Chusorn P, Techasen A, Boonmars T et al (2014) Circulating miR-192 in liver fluke-associated cholangiocarcinoma patients: a prospective prognostic indicator. J Hepatobiliary Pancreat Sci 21:864–872. https://doi.org/10.1002/jhbp.145
He Z, Yan T, Yuan Y, Yang D, Yang G (2020) miRNAs and lncRNAs in Echinococcus and echinococcosis. Int J Mol Sci 21:730. https://doi.org/10.3390/ijms21030730
Cucher M, Prada L, Mourglia-Ettlin G, Dematteis S, Camicia F, Asurmendi S et al (2011) Identification of Echinococcus granulosus microRNAs and their expression in different life cycle stages and parasite genotypes. Parasitol Int 41:439–448. https://doi.org/10.1016/j.ijpara.2010.11.010
Velasco-Tirado V, Romero-Alegría Á, Belhassen-García M, Alonso-Sardón M, Esteban-Velasco C, López-Bernús A et al (2017) Recurrence of cystic echinococcosis in an endemic area: a retrospective study. BMC Infect Dis 17:455. https://doi.org/10.1186/s12879-017-2556-9
Trakunram K, Champoochana N, Chaniad P, Thongsuksai P, Raungrut P (2019) MicroRNA isolation by trizol-based method and its stability in stored serum and cDNA derivatives. Asian Pac J Cancer Prev 20:1641. https://doi.org/10.31557/APJCP.2019.20.6.1641
Correia CN, Nalpas NC, McLoughlin KE, Browne JA, Gordon SV, MacHugh DE et al (2017) Circulating microRNAs as potential biomarkers of infectious disease. Front Immunol 8:118. https://doi.org/10.3389/fimmu.2017.00118
Xue X, Zhao Y, Wang X, Qin L, Hu R (2019) Development and validation of serum exosomal microRNAs as diagnostic and prognostic biomarkers for hepatocellular carcinoma. J Cell Biochem 120:135–142. https://doi.org/10.1002/jcb.27165
Paul S, Ruiz-Manriquez LM, Serrano-Cano FI, Estrada-Meza C, Solorio-Diaz KA, Srivastava A (2020) Human microRNAs in host–parasite interaction: a review. 3 Biotech 10:510. https://doi.org/10.1007/s13205-020-02498-6
Meningher T, Lerman G, Regev-Rudzki N, Gold D, Ben-Dov IZ, Sidi Y et al (2017) Schistosomal MicroRNAs isolated from extracellular vesicles in sera of infected patients: a new tool for diagnosis and follow-up of human schistosomiasis. J Infect Dis 215:378–386. https://doi.org/10.1093/infdis/jiw539
Samoil V, Dagenais M, Ganapathy V, Aldridge J, Glebov A, Jardim A et al (2018) Vesicle-based secretion in schistosomes: analysis of protein and microRNA (miRNA) content of exosome-like vesicles derived from Schistosoma mansoni. Sci Rep 8:3286. https://doi.org/10.1038/s41598-018-21587-4
Mu Y, Cai P, Olveda RM, Ross AG, Olveda DU, McManus DP (2020) Parasite-derived circulating microRNAs as biomarkers for the detection of human Schistosoma japonicum infection. Parasitology 147:889–896. https://doi.org/10.1017/S0031182019001690
Orsten S, Baysal İ, Yabanoglu-Ciftci S, Ciftci T, Azizova A, Akinci D et al (2021) MicroRNA expression profile in patients with cystic echinococcosis and identification of possible cellular pathways. J Helminthol 95:e1. https://doi.org/10.1017/S0022149X2000098X
Zhang X, Gong W, Cao S, Yin J, Zhang J, Cao J et al (2020) Comprehensive analysis of non-coding RNA profiles of exosome-like vesicles from the protoscoleces and hydatid cyst fluid of Echinococcus granulosus. Front Cell Infect Microbiol. https://doi.org/10.3389/fcimb.2020.00316
Siles-Lucas M, Sánchez-Ovejero C, González-Sánchez M, González E, Falcón-Pérez JM, Boufana B et al (2017) Isolation and characterization of exosomes derived from fertile sheep hydatid cysts. Vet Parasitol 236:22–33. https://doi.org/10.1016/j.vetpar.2017.01.022
Lin K, Zhou D, Li M, Meng J, He F, Yang X et al (2021) Echinococcus granulosus cyst fluid suppresses inflammatory responses by inhibiting TRAF6 signalling in macrophages. Parasitology 148:887–894. https://doi.org/10.1017/s0031182021000548
Pedrosa I, Saíz A, Arrazola J, Ferreirós J, Pedrosa CS (2000) Hydatid disease: radiologic and pathologic features and complications. Radiographics 20:795–817. https://doi.org/10.1148/radiographics.20.3.g00ma06795
Kijpaisalratana N, Nimsamer P, Khamwut A, Payungporn S, Pisitkun T, Chutinet A et al (2020) Serum miRNA125a-5p, miR-125b-5p, and miR-433-5p as biomarkers to differentiate between posterior circulation stroke and peripheral vertigo. BMC Neurol 20:372. https://doi.org/10.1186/s12883-020-01946-3
Jiang L, Huang Q, Zhang S, Zhang Q, Chang J, Qiu X et al (2010) Hsa-miR-125a-3p and hsa-miR-125a-5p are downregulated in non-small cell lung cancer and have inverse effects on invasion and migration of lung cancer cells. BMC Cancer 10:318. https://doi.org/10.1186/1471-2407-10-318
Wang R, Wang C, Meng X-J, Wei L (2021) miR-125-5p inhibits thyroid cancer growth and metastasis by suppressing the ERK/PI3K/AKT signal pathway. Mol Cell Toxicol. https://doi.org/10.1007/s13273-021-00175-0
Wu QB, Chen J, Zhu JW, Yin X, You HY, Lin YR et al (2018) MicroRNA125 inhibits RKO colorectal cancer cell growth by targeting VEGF. Int J Mol Med 42:665–673. https://doi.org/10.3892/ijmm.2018.3632
Acknowledgements
The authors would like to thank all the patients participating in the present study and the healthcare providers of the hospitals in Ahvaz for their contributions in the process of sampling. Moreover, the present study was a part of a Ph.D. thesis by M. Fasihi Karami.
Funding
The Cellular and Molecular Research Center, Medical Basic Sciences Research Institute, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran, provided financial support for this project under the grant number CMRC-0022.
Author information
Authors and Affiliations
Contributions
The present study was designed by MB and AR. All examinations performed by MFK. Data analysis was performed by MB, MFK, DD and MR. AB and AD were the surgeons who operated on the patients. MB, AR, and MFK wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Ethical Approval
This paper has not been published or is not under consideration for publication elsewhere. The submission of this manuscript for publication has been approved by all of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karami, M.F., Beiromvand, M., Rafiei, A. et al. Serum Level of egr-miR-2a-3p as a Potential Diagnostic Biomarker for Cystic Echinococcosis. Acta Parasit. 68, 114–121 (2023). https://doi.org/10.1007/s11686-022-00641-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11686-022-00641-2