Abstract
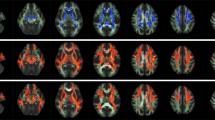
Purpose: The study aimed to unravel abnormal cerebral blood flow (CBF) in patients with Leber’s hereditary optic neuropathy (LHON) using arterial spin labeling (ASL) and to investigate the associations among disrupted CBF, disease duration, and neuro-ophthalmological impairment. Methods: ASL perfusion imaging data was collected from 20 patients with acute LHON, 29 patients with chronic LHON, and 37 healthy controls. We used a one-way analysis of covariance to test the intergroup differences in CBF. Linear and nonlinear curve fit models were applied to explore the associations among CBF, disease duration, and neuro-ophthalmological metrics. Results: Brain regions differed in LHON patients, including the left sensorimotor and bilateral visual areas (p < 0.05, cluster-wise family-wise error correction). Acute and chronic LHON patients demonstrated lower CBF in bilateral calcarine than the healthy controls. Chronic LHON had lower CBF in the left middle frontal gyrus and sensorimotor cortex, and temporal-partial junction than the healthy controls and acute LHON. A significant logarithmic negative correlation was shown between CBF of left middle frontal gyrus and disease duration. A significant linear positive correlation was found between retinal nerve fiber layer thickness and CBF in left middle frontal gyrus, and negative correlations between loss of variance and CBF in left middle frontal gyrus and sensorimotor cortex (p < 0.05, Bonferroni correction). Conclusion: LHON patients exhibited reduced CBF in the visual pathway, sensorimotor and higher-tier cognitive areas. Disease duration and neuro-ophthalmological impairments can influence the metabolism of non-visual areas.




Similar content being viewed by others
Data Availability
The individual original MRI data will not be shared with the public because the subjects’ signed permission of personal data spread had not been approved. We promised that the imaging protocols and statistical analysis results would be accessed to public once the draft has been accepted for publication. Any reader can get the shareable data by email to the corresponding author.
Code Availability
Not applicable.
References
Alsop, D. C., Detre, J. A., Golay, X., Gunther, M., Hendrikse, J., Hernandez-Garcia, L., & Zaharchuk, G. (2015). Recommended implementation of arterial spin-labeled perfusion MRI for clinical applications: A consensus of the ISMRM perfusion study group and the european consortium for ASL in dementia. Magnetic Resonance In Medicine, 73(1), 102–116. https://doi.org/10.1002/mrm.25197.
Anurova, I., Renier, L. A., De Volder, A. G., Carlson, S., & Rauschecker, J. P. (2015). Relationship between cortical thickness and functional activation in the early blind. Cerebral Cortex, 25(8), 2035–2048. https://doi.org/10.1093/cercor/bhu009.
Asanad, S., Tian, J. J., Frousiakis, S., Jiang, J. P., Kogachi, K., Felix, C. M., & Sadun, A. A. (2019). Optical coherence tomography of the retinal ganglion cell complex in Leber’s Hereditary Optic Neuropathy and Dominant Optic Atrophy. Current Eye Research, 44(6), 638–644. https://doi.org/10.1080/02713683.2019.1567792.
Balducci, N., Savini, G., Cascavilla, M. L., La Morgia, C., Triolo, G., Giglio, R., & Barboni, P. (2016). Macular nerve fibre and ganglion cell layer changes in acute Leber’s hereditary optic neuropathy. British Journal Of Ophthalmology, 100(9), 1232–1237. https://doi.org/10.1136/bjophthalmol-2015-307326.
Barbiroli, B., Montagna, P., Cortelli, P., Iotti, S., Lodi, R., Barboni, P., & Zaniol, P. (1995). Defective brain and muscle energy metabolism shown by in vivo 31P magnetic resonance spectroscopy in nonaffected carriers of 11778 mtDNA mutation. Neurology, 45(7), 1364–1369. https://doi.org/10.1212/wnl.45.7.1364.
Barcella, V., Rocca, M. A., Bianchi-Marzoli, S., Milesi, J., Melzi, L., Falini, A., & Filippi, M. (2010). Evidence for retrochiasmatic tissue loss in Leber’s hereditary optic neuropathy. Human Brain Mapping, 31(12), 1900–1906. https://doi.org/10.1002/hbm.20985.
Bhoyroo, R., Hands, B., Caeyenberghs, K., de Luca, A., Leemans, A., Wigley, A., & Hyde, C. (2022). Association between Motor Planning and the Frontoparietal Network in Children: An exploratory Multimodal Study. Journal Of The International Neuropsychological Society, 28(9), 926–936. https://doi.org/10.1017/S1355617721001168.
Blanke, O., Landis, T., Spinelli, L., & Seeck, M. (2004). Out-of-body experience and autoscopy of neurological origin. Brain, 127(Pt 2), 243–258. https://doi.org/10.1093/brain/awh040.
Borrelli, E., Triolo, G., Cascavilla, M. L., La Morgia, C., Rizzo, G., Savini, G., & Barboni, P. (2016). Changes in Choroidal Thickness follow the RNFL changes in Leber’s Hereditary Optic Neuropathy. Scientific Reports, 6, 37332. https://doi.org/10.1038/srep37332.
Brugger, P. (2002). Reflective mirrors: Perspective-taking in autoscopic phenomena. Cognitive Neuropsychiatry, 7(3), 179–194. https://doi.org/10.1080/13546800244000076.
Burton, H., Diamond, J. B., & McDermott, K. B. (2003). Dissociating cortical regions activated by semantic and phonological tasks: A FMRI study in blind and sighted people. Journal Of Neurophysiology, 90(3), 1965–1982. https://doi.org/10.1152/jn.00279.2003.
Burton, H., Sinclair, R. J., & McLaren, D. G. (2004). Cortical activity to vibrotactile stimulation: An fMRI study in blind and sighted individuals. Human Brain Mapping, 23(4), 210–228. https://doi.org/10.1002/hbm.20064.
Carelli, V., Carbonelli, M., de Coo, I. F., Kawasaki, A., Klopstock, T., Lagreze, W. A., & Barboni, P. (2017). International Consensus Statement on the clinical and therapeutic management of Leber Hereditary Optic Neuropathy. Journal Of Neuro-Ophthalmology, 37(4), 371–381. https://doi.org/10.1097/WNO.0000000000000570.
Chinnery, P. F., Johnson, M. A., Wardell, T. M., Singh-Kler, R., Hayes, C., Brown, D. T., & Turnbull, D. M. (2000). The epidemiology of pathogenic mitochondrial DNA mutations. Annals Of Neurology, 48(2), 188–193.
Collignon, O., Charbonneau, G., Lassonde, M., & Lepore, F. (2009). Early visual deprivation alters multisensory processing in peripersonal space. Neuropsychologia, 47(14), 3236–3243. https://doi.org/10.1016/j.neuropsychologia.2009.07.025.
Collignon, O., Dormal, G., Albouy, G., Vandewalle, G., Voss, P., Phillips, C., & Lepore, F. (2013). Impact of blindness onset on the functional organization and the connectivity of the occipital cortex. Brain, 136(Pt 9), 2769–2783. https://doi.org/10.1093/brain/awt176.
Cui, S., Yang, L., Jiang, H., Peng, J., Shang, J., Wang, J., & Zhang, X. (2020). Clinical features of chinese sporadic Leber Hereditary Optic Neuropathy caused by Rare primary mtDNA mutations. Journal Of Neuro-Ophthalmology, 40(1), 30–36. https://doi.org/10.1097/WNO.0000000000000799.
d’Almeida, O. C., Mateus, C., Reis, A., Grazina, M. M., & Castelo-Branco, M. (2013). Long term cortical plasticity in visual retinotopic areas in humans with silent retinal ganglion cell loss. Neuroimage, 81, 222–230. https://doi.org/10.1016/j.neuroimage.2013.05.032.
De Volder, A. G., Bol, A., Blin, J., Robert, A., Arno, P., Grandin, C., & Veraart, C. (1997). Brain energy metabolism in early blind subjects: Neural activity in the visual cortex. Brain Research, 750(1–2), 235–244. https://doi.org/10.1016/s0006-8993(96)01352-2.
Ebbesen, C. L., Insanally, M. N., Kopec, C. D., Murakami, M., Saiki, A., & Erlich, J. C. (2018). More than just a “Motor”: Recent Surprises from the Frontal Cortex. Journal Of Neuroscience, 38(44), 9402–9413. https://doi.org/10.1523/JNEUROSCI.1671-18.2018.
Fieger, A., Roder, B., Teder-Salejarvi, W., Hillyard, S. A., & Neville, H. J. (2006). Auditory spatial tuning in late-onset blindness in humans. Journal Of Cognitive Neuroscience, 18(2), 149–157. https://doi.org/10.1162/089892906775783697.
Finsterer, J., & Zarrouk-Mahjoub, S. (2018). Neuropathy of peripheral nerves in Leber’s hereditary optic neuropathy. Journal Of The Neurological Sciences, 390, 193–194. https://doi.org/10.1016/j.jns.2018.04.041.
Funakawa, I., Kato, H., Terao, A., Ichihashi, K., Kawashima, S., Hayashi, T., & Miyazaki, S. (1995). Cerebellar ataxia in patients with Leber’s hereditary optic neuropathy. Journal Of Neurology, 242(2), 75–77. https://doi.org/10.1007/BF00887819.
Grochowski, C., Jonak, K., Maciejewski, M., Stepniewski, A., & Rahnama-Hezavah, M. (2020a). Alteration within the hippocampal volume in patients with LHON Disease-7 Tesla MRI Study. J Clin Med, 10(1), 10.3390/jcm10010014.
Grochowski, C., Symms, M., Jonak, K., Krukow, P., Wood, T., Ljungberg, E., & Barker, G. J. (2020b). The evaluation of Optic Nerves using 7 Tesla “Silent” Zero Echo Time Imaging in patients with Leber’s Hereditary Optic Neuropathy with or without Idebenone Treatment. J Clin Med, 9(4), 10.3390/jcm9041112.
Hays, C. C., Zlatar, Z. Z., & Wierenga, C. E. (2016). The Utility of Cerebral Blood Flow as a biomarker of preclinical Alzheimer’s Disease. Cellular And Molecular Neurobiology, 36(2), 167–179. https://doi.org/10.1007/s10571-015-0261-z.
Jann, K., Hernandez, L. M., Beck-Pancer, D., McCarron, R., Smith, R. X., Dapretto, M., & Wang, D. J. (2015). Altered resting perfusion and functional connectivity of default mode network in youth with autism spectrum disorder. Brain Behav, 5(9), e00358. https://doi.org/10.1002/brb3.358.
Jonak, K. (2020). Widespread reductions of spontaneous neurophysiological activity in Leber’s Disease-An Application of EEG Source Current Density Reconstruction. Brain Sci, 10(9), https://doi.org/10.3390/brainsci10090622.
Jonak, K., Krukow, P., Jonak, K. E., Radzikowska, E., Baj, J., Niedzialek, A., & Grochowski, C. (2020). Decreased volume of lateral and medial geniculate nuclei in patients with LHON Disease-7 Tesla MRI Study. J Clin Med, 9(9), https://doi.org/10.3390/jcm9092914.
Kirkman, M. A., Korsten, A., Leonhardt, M., Dimitriadis, K., De Coo, I. F., Klopstock, T., & Yu-Wai-Man, P. (2009). Quality of life in patients with leber hereditary optic neuropathy. Invest Ophthalmol Vis Sci, 50(7), 3112–3115. https://doi.org/10.1167/iovs.08-3166.
Kombos, T., Suess, O., Kern, B. C., Funk, T., Hoell, T., Kopetsch, O., & Brock, M. (1999). Comparison between monopolar and bipolar electrical stimulation of the motor cortex. Acta Neurochir (Wien), 141(12), 1295–1301. https://doi.org/10.1007/s007010050433.
Leng, Y., Liu, Y., Fang, X., Li, Y., Yu, L., Yuan, Y., & Wang, Z. (2015). The mitochondrial DNA 10197 G > A mutation causes MELAS/Leigh overlap syndrome presenting with acute auditory agnosia. Mitochondrial DNA, 26(2), 208–212. https://doi.org/10.3109/19401736.2014.905860.
Liu, Y., Yu, C., Liang, M., Li, J., Tian, L., Zhou, Y., & Jiang, T. (2007). Whole brain functional connectivity in the early blind. Brain, 130(Pt 8), 2085–2096. https://doi.org/10.1093/brain/awm121.
Liu, L., Yuan, C., Ding, H., Xu, Y., Long, M., Li, Y., & Yu, C. (2017). Visual deprivation selectively reshapes the intrinsic functional architecture of the anterior insula subregions. Scientific Reports, 7, 45675. https://doi.org/10.1038/srep45675.
Long, M., Wang, L., Tian, Q., Ding, H., Qin, W., Shi, D., & Yu, C. (2019). Brain white matter changes in asymptomatic carriers of Leber’s hereditary optic neuropathy. Journal Of Neurology, 266(6), 1474–1480. https://doi.org/10.1007/s00415-019-09284-2.
Ma, G., Yang, D., Qin, W., Liu, Y., Jiang, T., & Yu, C. (2016). Enhanced functional coupling of hippocampal sub-regions in congenitally and late blind subjects. Front Neurosci, 10, 612. https://doi.org/10.3389/fnins.2016.00612.
Manners, D. N., Rizzo, G., La Morgia, C., Tonon, C., Testa, C., Barboni, P., & Lodi, R. (2015). Diffusion Tensor Imaging Mapping of Brain White Matter Pathology in mitochondrial Optic Neuropathies. Ajnr. American Journal Of Neuroradiology, 36(7), 1259–1265. https://doi.org/10.3174/ajnr.A4272.
Manners, D. N., Gramegna, L. L., La Morgia, C., Sighinolfi, G., Fiscone, C., Carbonelli, M., & Lodi, R. (2022). Multishell Diffusion MR Tractography yields morphological and Microstructural Information of the Anterior Optic Pathway: A proof-of-Concept study in patients with Leber’s Hereditary Optic Neuropathy. International Journal Of Environmental Research And Public Health, 19(11), https://doi.org/10.3390/ijerph19116914.
Mateus, C., d’Almeida, O. C., Reis, A., Silva, E., & Castelo-Branco, M. (2016). Genetically induced impairment of retinal ganglion cells at the axonal level is linked to extrastriate cortical plasticity. Brain Struct Funct, 221(3), 1767–1780. https://doi.org/10.1007/s00429-015-1002-2.
Matsuzaki, M., Hirami, Y., Uyama, H., & Kurimoto, Y. (2018). Optical coherence tomography angiography changes in radial peripapillary capillaries in Leber hereditary optic neuropathy. Am J Ophthalmol Case Rep, 9, 51–55. https://doi.org/10.1016/j.ajoc.2018.01.003.
Mercuri, M. A., White, H., & Oliveira, C. (2017). Vision loss and symmetric basal ganglia lesions in Leber Hereditary Optic Neuropathy. Journal Of Neuro-Ophthalmology, 37(4), 411–413. https://doi.org/10.1097/WNO.0000000000000524.
Milesi, J., Rocca, M. A., Bianchi-Marzoli, S., Petrolini, M., Pagani, E., Falini, A., & Filippi, M. (2012). Patterns of white matter diffusivity abnormalities in Leber’s hereditary optic neuropathy: A tract-based spatial statistics study. Journal Of Neurology, 259(9), 1801–1807. https://doi.org/10.1007/s00415-011-6406-1.
Miyaue, N., Yamanishi, Y., Tada, S., Ando, R., Yabe, H., Nagai, M., & Nomoto, M. (2019). Repetitive brainstem lesions in mitochondrial DNA 11778G > A mutation of Leber hereditary optic neuropathy. eNeurologicalSci, 14, 74–76. https://doi.org/10.1016/j.ensci.2019.01.002.
Mizoguchi, A., Hashimoto, Y., Shinmei, Y., Nozaki, M., Ishijima, K., Tagawa, Y., & Ishida, S. (2015). Macular thickness changes in a patient with Leber’s hereditary optic neuropathy. Bmc Ophthalmology, 15, 27. https://doi.org/10.1186/s12886-015-0015-1.
Nakaso, K., Adachi, Y., Fusayasu, E., Doi, K., Imamura, K., Yasui, K., & Nakashima, K. (2012). Leber’s Hereditary Optic Neuropathy with Olivocerebellar Degeneration due to G11778A and T3394C mutations in the mitochondrial DNA. J Clin Neurol, 8(3), 230–234. https://doi.org/10.3988/jcn.2012.8.3.230.
Ogawa, S., Takemura, H., Horiguchi, H., Terao, M., Haji, T., Pestilli, F., & Masuda, Y. (2014). White matter consequences of retinal receptor and ganglion cell damage. Invest Ophthalmol Vis Sci, 55(10), 6976–6986. https://doi.org/10.1167/iovs.14-14737.
Orru, G., Bertelloni, D., Cesari, V., Conversano, C., & Gemignani, A. (2021). Targeting temporal parietal junction for assessing and treating disembodiment phenomena: A systematic review of TMS effect on depersonalization and derealization disorders (DPD) and body illusions. AIMS Neurosci, 8(2), 181–194. https://doi.org/10.3934/Neuroscience.2021009.
Ostojic, J., Jancic, J., Kozic, D., Semnic, R., Koprivsek, K., Prvulovic, M., & Kostic, V. (2009). Brain white matter 1 H MRS in Leber optic neuropathy mutation carriers. Acta Neurol Belg, 109(4), 305–309.
Pemp, B., Mitsch, C., Kircher, K., & Reitner, A. (2021). Changes in visual function and correlations with inner retinal structure in Acute and Chronic Leber’s Hereditary Optic Neuropathy Patients after treatment with Idebenone. J Clin Med, 10(1), https://doi.org/10.3390/jcm10010151.
Poincenot, L., Pearson, A. L., & Karanjia, R. (2020). Demographics of a large International Population of Patients affected by Leber’s Hereditary Optic Neuropathy. Ophthalmology, 127(5), 679–688. https://doi.org/10.1016/j.ophtha.2019.11.014.
Ptito, M., Moesgaard, S. M., Gjedde, A., & Kupers, R. (2005). Cross-modal plasticity revealed by electrotactile stimulation of the tongue in the congenitally blind. Brain, 128(Pt 3), 606–614. https://doi.org/10.1093/brain/awh380.
Qin, W., Xuan, Y., Liu, Y., Jiang, T., & Yu, C. (2015). Functional connectivity density in congenitally and late blind subjects. Cerebral Cortex, 25(9), 2507–2516. https://doi.org/10.1093/cercor/bhu051.
Rance, G., Kearns, L. S., Tan, J., Gravina, A., Rosenfeld, L., Henley, L., & Mackey, D. A. (2012). Auditory function in individuals within Leber’s hereditary optic neuropathy pedigrees. Journal Of Neurology, 259(3), 542–550. https://doi.org/10.1007/s00415-011-6230-7.
Rizzo, G., Tozer, K. R., Tonon, C., Manners, D., Testa, C., Malucelli, E., & Lodi, R. (2012). Secondary post-geniculate involvement in Leber’s hereditary optic neuropathy. Plos One, 7(11), e50230. https://doi.org/10.1371/journal.pone.0050230.
Rocca, M. A., Valsasina, P., Pagani, E., Bianchi-Marzoli, S., Milesi, J., Falini, A., & Filippi, M. (2011). Extra-visual functional and structural connection abnormalities in Leber’s hereditary optic neuropathy. Plos One, 6(2), e17081. https://doi.org/10.1371/journal.pone.0017081.
Saracchi, E., Difrancesco, J. C., Brighina, L., Marzorati, L., Curto, N. A., Lamperti, C., & Ferrarese, C. (2013). A case of Leber hereditary optic neuropathy plus dystonia caused by G14459A mitochondrial mutation. Neurological Sciences : Official Journal Of The Italian Neurological Society And Of The Italian Society Of Clinical Neurophysiology, 34(3), 407–408. https://doi.org/10.1007/s10072-012-1013-1.
Selya, A. S., Rose, J. S., Dierker, L. C., Hedeker, D., & Mermelstein, R. J. (2012). A practical guide to calculating Cohen’s f(2), a measure of local effect size, from PROC MIXED. Frontiers In Psychology, 3, 111. https://doi.org/10.3389/fpsyg.2012.00111.
Tian, Q., Wang, L., Zhang, Y., Fan, K., Liang, M., Shi, D., & Ding, H. (2022). Brain Gray Matter Atrophy and functional connectivity remodeling in patients with chronic LHON. Front Neurosci, 16, 885770. https://doi.org/10.3389/fnins.2022.885770.
Vacchiano, V., Tonon, C., Mitolo, M., Evangelisti, S., Carbonelli, M., Liguori, R., & La Morgia, C. (2019). Functional MRI study in a case of Charles Bonnet syndrome related to LHON. Bmc Neurology, 19(1), 350. https://doi.org/10.1186/s12883-019-1579-9.
Vaphiades, M. S. (2011). Magnetic resonance findings in the pregeniculate visual pathways in Leber hereditary optic neuropathy. J Neuroophthalmol, 31(2), 194; author reply 194. doi:https://doi.org/10.1097/WNO.0b013e31821bca86
Veraart, C., De Volder, A. G., Wanet-Defalque, M. C., Bol, A., Michel, C., & Goffinet, A. M. (1990). Glucose utilization in human visual cortex is abnormally elevated in blindness of early onset but decreased in blindness of late onset. Brain Research, 510(1), 115–121. https://doi.org/10.1016/0006-8993(90)90735-t.
Vrieze, S. I. (2012). Model selection and psychological theory: A discussion of the differences between the Akaike information criterion (AIC) and the bayesian information criterion (BIC). Psychological Methods, 17(2), 228–243. https://doi.org/10.1037/a0027127.
Wang, L., Fan, K., Zhang, Y., Chen, Y., Tian, Q., & Shi, D. (2017). Quantitative assessment of optic nerve in patients with Leber’s hereditary optic neuropathy using reduced field-of-view diffusion tensor imaging. European Journal Of Radiology, 93, 24–29. https://doi.org/10.1016/j.ejrad.2017.05.025.
Wang, H., Li, S., Chen, X., Wang, Y., Li, J., & Wang, Z. (2020). Cerebral Blood Flow Alterations in High Myopia: An Arterial Spin Labeling Study. Neural Plast, 2020, 6090262. doi:https://doi.org/10.1155/2020/6090262
Wang, L., Ding, H., Chen, B. T., Fan, K., Tian, Q., Long, M., & Qin, W. (2021). Occult primary white matter impairment in Leber hereditary optic neuropathy. European Journal Of Neurology, 28(9), 2871–2881. https://doi.org/10.1111/ene.14995.
Wu, B., Lou, X., Wu, X., & Ma, L. (2014). Intra- and interscanner reliability and reproducibility of 3D whole-brain pseudo-continuous arterial spin-labeling MR perfusion at 3T. Journal Of Magnetic Resonance Imaging, 39(2), 402–409. https://doi.org/10.1002/jmri.24175.
Xu, L., Qin, W., Zhuo, C., Liu, H., Zhu, J., & Yu, C. (2017). Combination of volume and perfusion parameters reveals different types of grey matter changes in schizophrenia. Scientific Reports, 7(1), 435. https://doi.org/10.1038/s41598-017-00352-z.
Yen, M. Y., Wang, A. G., & Wei, Y. H. (2006). Leber’s hereditary optic neuropathy: A multifactorial disease. Progress In Retinal And Eye Research, 25(4), 381–396. https://doi.org/10.1016/j.preteyeres.2006.05.002.
Yu, C., Liu, Y., Li, J., Zhou, Y., Wang, K., Tian, L., & Li, K. (2008). Altered functional connectivity of primary visual cortex in early blindness. Human Brain Mapping, 29(5), 533–543. https://doi.org/10.1002/hbm.20420.
Zanna, C., Ghelli, A., Porcelli, A. M., Carelli, V., Martinuzzi, A., & Rugolo, M. (2003). Apoptotic cell death of cybrid cells bearing Leber’s hereditary optic neuropathy mutations is caspase independent. Annals Of The New York Academy Of Sciences, 1010, 213–217. https://doi.org/10.1196/annals.1299.037.
Zhang, Y., Huang, H., Wei, S., Qiu, H., Gong, Y., Li, H., & Liu, Z. (2014). Characterization of retinal nerve fiber layer thickness changes associated with Leber’s hereditary optic neuropathy by optical coherence tomography. Exp Ther Med, 7(2), 483–487. https://doi.org/10.3892/etm.2013.1430.
Zhang, J., Wang, L., Ding, H., Fan, K., Tian, Q., Liang, M., & Qin, W. (2021). Abnormal large-scale structural rich club organization in Leber’s hereditary optic neuropathy. Neuroimage Clin, 30, 102619. https://doi.org/10.1016/j.nicl.2021.102619.
Acknowledgements
We appreciated members of the Department of Radiology & Tianjin Key Lab of Functional Imaging of Tianjin Medical University General Hospital for their technical support. We also appreciated members of the Department of Radiology of Henan Provincial People’s Hospital for data collection.
Funding
This work was supported by the National Natural Science Foundation of China (81971599, 81771818, 82030053, 81271534, 81601473), National Key Research and Development Program of China (2018YFC1314300), Natural Science Foundation of Tianjin City (19JCYBJC25100), and Postdoctoral Research Foundation of China (2017M611175).
Author information
Authors and Affiliations
Contributions
Wen Qin and Chunshui Yu designed research; Yi Ji, Ling Wang and Hao Ding performed research; Yi Ji, Ling Wang, Qin Tian, Dapeng Shi and Ke Fan analyzed data; Yi Ji and Ling Wang wrote the paper. Wen Qin is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding authors
Ethics declarations
Ethical approval
The research was approved by the Ethics Committees of Henan Provincial People’s Hospital and was carried out in compliance with the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Consent to participate
Informed consent was obtained from all participants or their legal guardians if they were children or adolescents.
Consent for publication
The work described has not been published before. It is not under consideration for publication elsewhere. Its publication has been approved by all co-authors, if any. Its publication has been approved by the responsible authorities at the institution where the work is carried out.
Competing Interests
All authors declare no conflict of interest.
Additional information
Ling Wang, Yi Ji and Hao Ding contributed equally to the manuscript.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, L., Ji, Y., Ding, H. et al. Abnormal cerebral blood flow in patients with Leber’s hereditary optic neuropathy. Brain Imaging and Behavior 17, 471–480 (2023). https://doi.org/10.1007/s11682-023-00775-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-023-00775-5