Abstract
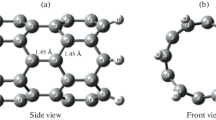
Density functional theory calculations were carried out to investigate the binding ability of transition metals (TM = Cr, Mo, W, Mn, Tc, and Re) on (5,5) armchair single-walled carbon nanotubes (SWCNTs) and their adsorption ability with ammonia (NH3) and nitrogen dioxide (NO2). The geometric, electronic, and energetic properties of pristine SWCNT doping with TM and their NH3 and NO2 adsorptions were calculated to explore their potentials as gas adsorptions and sensors. The binding abilities of TMs to the SWCNT are found to be in the order: Cr > Mo > Tc > Re > W > Mn. According to the results, compared to the pristine SWCNT, TM atom doping can significantly increase gas adsorption ability in which W-SWCNT has the strongest interaction, and the ability to absorb NO2 is higher than NH3. The density of states and orbital distributions display that the band gaps and electron delocalizations of TM-SWCNTs are significantly changed upon gas adsorption. These observations suggest that the TM-doped SWCNTs can be introduced as promising candidates in gas adsorptions and sensor devices for storage and detecting NH3 and NO2 molecules.
Similar content being viewed by others
References
S.S. Varghese, S. Lonkar, K.K. Singh, S. Swaminathan, and A. Abdalad, Sens. Actuators B 218, 160 (2015).
A. Abdelhalim, M. Winkler, F. Loghin, C. Zeiser, P. Lugli, and A. Abdellah, Sens. Actuators B 220, 1288 (2015).
J. Kim, S.W. Choi, J.H. Lee, Y. Chung, and Y.T. Byun, Sens. Actuators B 228, 688 (2016).
I.V. Zaporotskova, N.P. Boroznina, Y.N. Parkhomenko, and L.V. Kozhitov, Mod. Electron. Mater. 2, 95 (2016).
Y. Gogotsi, Carbon nanomaterials (Pennsylvania: Taylor and Francis Group, LLC, 2006).
M.A. Azam, M.W.A. Rashid, K. Isomura, A. Fujiwara, and T. Shimoda, Adv. Mater. Res. 620, 213 (2013).
R.N.A.R. Seman, M.A. Azam, and A.A. Mohamad, Renew. Sustain. Energy Rev. 75, 644 (2017).
D. Jung, M. Han, and G. Lee, Sens. Actuators A: Phys. 211, 51 (2014).
M. Roberts, M. LeMieux, and Z. Bao, ACS Nano 3, 3287 (2009).
X. Zhang, Y. Gui, H. Xiao, and Y. Zhang, Appl. Surf. Sci. 379, 47 (2016).
X. Zhang, Z. Dai, L. Wei, N. Liang, and X. Wu, Sensors (Basel) 13, 15159 (2013).
M. Yoosefian, M. Zahedi, A. Mola, and S. Naserian, Appl. Surf. Sci. 349, 864 (2015).
X. Zhang, H. Cui, X. Dong, D. Chen, and J. Tang, Appl. Surf. Sci. 420, 825 (2017).
Y. Chen, S. Yin, Y. Chen, W. Cen, J. Li, and H. Yin, Appl. Surf. Sci. 434, 382 (2018).
T.T.H. Nguyen, V.K. Le, C.L. Minh, and N.H. Nguyen, Comput. Theor. Chem. 1100, 46 (2017).
M.A. Azam, F.M. Alias, L.W. Tack, R.N.A.R. Seman, and M.F.M. Tai, J. Mol. Graph. Model. 75, 85 (2017).
W. Li, X.M. Lu, G.Q. Li, J.J. Ma, P.Y. Zeng, J.F. Chen, Z.L. Pan, and Q.Y. He, Appl. Surf. Sci. 364, 560 (2016).
H.Y. Abdullah, Results Phys. 6, 1146 (2016).
S.M. Aghaei, M.M. Monshi, I. Torres, S.M.J. Zeidi, and I. Calizo, Appl. Surf. Sci. 427, 326 (2018).
M.D. Gilan and R. Chegel, J. Electron. Mater. 47, 1009 (2018).
V. Nagarajan and R. Chandiramouli, Superlattices Microstruct. 101, 160 (2017).
A.N.A. Anasthasiya, M. Khaneja, and B.G. Jeyaprakash, J. Electron. Mater. 46, 5642 (2017).
Y. Li, M. Hodak, W. Lu, and J. Bernhol, Carbon 101, 177 (2016).
MdS Khan and A. Srivastava, J. Electrochem. Chem. 775, 243 (2016).
Y. Cheng, R. Meng, C. Tan, X. Chen, and J. Xiao, Appl. Surf. Sci. 427, 176 (2018).
J. Kong, N. Franklin, C. Zhou, M. Chapline, S. Peng, K. Cho, and H. Dai, Science 287, 622 (2000).
K. Azizi and M. Karimpanah, Appl. Surf. Sci. 285P, 102 (2013).
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, J.A. Montgomery Jr, T. Vreven, K.N. Kudin, J.C. Burant, J.M. Millam, S.S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G.A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J.E. Knox, H.P. Hratchian, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, P.Y. Ayala, K. Morokuma, G.A. Voth, P. Salvador, J.J. Dannenberg, V.G. Zakrzewski, S. Dapprich, A.D. Daniels, M.C. Strain, O. Farkas, D.K. Malick, A.D. Rabuck, K. Raghavachari, J.B. Foresman, J.V. Ortiz, Q. Cui, A.G. Baboul, S. Clifford, J. Cioslowski, B.B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R.L. Martin, D.J. Fox, T. Keith, M.A. Al-Laham, C.Y. Peng, A. Nanayakkara, M. Challacombe, P.M.W. Gill, B. Johnson, W. Chen, M.W. Wong, C. Gonzalez, and J.A. Pople, GAUSSIAN 09, Revision A.02 (Wallingford: Gaussian Inc, 2009).
A.D. Becke, Phys. Rev. A 38, 3098 (1988).
A.D. Becke, J. Chem. Phys. 98, 5648 (1993).
C. Lee, W. Yang, and R.G. Parr, Phys. Rev. B 37, 785 (1988).
P.J. Hay and W.R. Wadt, J. Chem. Phys. 82, 270 (1985).
W.R. Wadt and P.J. Hay, J. Chem. Phys. 82, 284 (1985).
P.J. Hay and W.R. Wadt, J. Chem. Phys. 82, 299 (1985).
B. Wanno and C. Tabtimsai, Superlattices Microstruct. 67, 110 (2014).
J. Arasteh and M. Naseh, Struct. Chem. 30, 97 (2019).
B.P. Giovanne, L.M. Naiara, S.L.F. Guilherme, and R.S. Julio, Theor. Chem. Acc. 138, 31 (2019).
T. Movlarooy and B. Minaie, J. Comput. Electron. 17, 1441 (2018).
Z. Zhou, M. Steigerwald, M. Hybertsen, L. Brus, and R.A. Friesner, J. Am. Chem. Soc. 126, 3597 (2004).
S. Saha, T.C. Dinadayalane, J.S. Murray, D. Leszczynska, and J. Leszczynski, J. Phys. Chem. C 116, 22399 (2012).
R.G. Parr, R.A. Donelly, M. Levy, and W.E. Palke, J. Chem. Phys. 68, 3801 (1978).
J.F. Janak, Phys. Rev. B 18, 7165 (1978).
B. Gómez and J.M. Martínez-Magadán, J. Phys. Chem. B 109, 14868 (2005).
N.M. O’Boyle, A.L. Tenderholt, and K.M. Langner, J. Comput. Chem. 29, 839 (2008).
P. Flükiger, H.P. Lüthi, and S. Portmann, MOLEKEL 4.3 (Manno: Swiss Center for Scientific Computing, 2000).
S. Banerjee, T. Hemraj-Benny, and S.S. Wong, Adv. Mater. 17, 17 (2005).
M. Malehmir and B. Khoshnevisan, Chem. Phys. 478, 62 (2016).
C. Tabtimsai, V. Ruangpornvisuti, and B. Wanno, Physica E 49, 61 (2013).
H.R. Balangi and A.A. Shokri, Physica E 74, 515 (2015).
A. Kutana, E.S. Penev, and B.I. Yakobson, Nanoscale 6, 5820 (2014).
A.S. Rad and D. Zareyee, Vacuum 130, 113 (2016).
Acknowledgments
The financial support (2561A14702104) by Research and Development Institute, Rajabhat Maha Sarakham University is gratefully acknowledged. We also thank the Computational Chemistry Center for Nanotechnology (CCCN), Department of Chemistry, Faculty of Science and Technology, Rajabhat Maha Sarakham University, and Center of Excellence for Innovation in Chemistry (PERCH-CIC), Department of Chemistry, Faculty of Science, Mahasarakham University for the facilities provided.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tabtimsai, C., Wanno, B., Utairueng, A. et al. First Principles Investigation of NH3 and NO2 Adsorption on Transition Metal-Doped Single-Walled Carbon Nanotubes. J. Electron. Mater. 48, 7226–7238 (2019). https://doi.org/10.1007/s11664-019-07537-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-019-07537-4