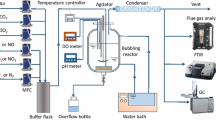
Abstract
Sodium sulfate accumulation in the water system is a common problem in the production of primary metals from sulfide minerals. In flash smelting-flash converting copper smelters, when molten copper matte quenched in industrial water to produce matte granules, Na2SO4 precipitated and entered the system, which had negative influence on the matte converting process. The reaction behaviors of copper matte powder containing Na2SO4 have been studied through thermodynamic calculation and single-particle experiments. Calculated results showed that a liquid sulfate phase, Na2S containing copper matte, and Cu–Na alloy may form in the processing conditions of the converting process. The influence of the Na2SO4 dosage, fluxing conditions, and processing temperature has been systematically studied, and the results obtained showed that: (1); Na2SO4 physically precipitated on matte particles, and the Cu–Na alloy phase was found within copper phase in reacted particles; (2); Na2SO4 contamination hindered oxygen transfer by forming a low melting sulfate phase, and the de-sulfuration rate of matte powder changed dramatically along with the variation in Na2SO4 amount; (3); CaO flux was able to promote the converting reactions and Na2SO4 transformation; (4); Na2SO4 melt and formed a sulphate layer on matte particles at 1000 °C, Cu–Na alloy phase was found in samples collected at 1200 °C, and Cu–Na–O phase was detected in blister copper at 1600 °C.








Similar content being viewed by others
References
J.A. Asteljoki, L.K. Bailey, D.B. George, and D.W. Rodolff: J. Met., 1985, vol. 20, pp. 245-250.
K.E. Sutliff, T.I. Probert, and C.T. Unger (partI) and J.E. Hoffmann, K.E. Sutliff, B.A. Wells, and D.B. George (Part II): JOM, 1996, vol. 48(8), pp. 42–44.
P. Hanniala, I. Kojo, and M. Kytö: TMS, 1994, pp. 107–19.
P. Hanniala, I. Kojo, and M. Kytö. TMS, 1998, pp. 239–47.
D. George. TMS, 2000, pp. 3–13.
[6] I. Kojo, A. Jokilaakso, and P. Hanniala. JOM, 2000, vol. 52(2), pp. 57-61.
[7] I. Kojo, A. Jokilaakso, and P. Hanniala. Rudy Met. Niezelaz., 2000, vol. 45(12), pp. 606-612.
A.D. Wan, W.S. Guo, G.S. Zhang, J.P. Zhang, and X.F. Xie. Nonferr Metall Equip., 2017, pp. 52–57. (In Chinese).
[9] J. Zhou. Nonferr Met., 2017, vol. 10, pp. 1-9. (In Chinese)
M.M. Congress and M.J. Jones: Proceedings of the Ninth Commonwealth Mining & Metallurgical Congress, 1970.
I. Kojo, M. Lahtinen, and E. Miettinen: International Peirce-Smith Converting Centennial Symposium Held at the 2009 TMS Annual Meeting and Exhibition, San Francisco, The USA, 2009, pp. 15–19.
Z.H. Liu and L.G. Xia: Miner. Process. Extr. Metall. 2018, vol. 127(1), pp. 117-124.
[13] Z.A. Wang, W. Yang, H. Liu, H. Jin, H.Q. Chen, K. Su, Y.J. Tu and W.L. Wang. J. Anal. Appl. Pyrolysis., 2019, vol. 142, pp. 1-10.
[14] R.R. Moskalyk, and A.M. Alfantazi. Miner. Eng.,2003, vol. 16, pp. 893-919.
[15] D. Freyer, and W. Voigt. .Monatsh. Chem., 2003, vol. 134(5), pp. 693-719.
P. Ren: World Nonferrous Metal. 2018;496(4), pp. 27–28
L.G. Xia, F. Yu, H.B. Shao, Y. Shen, L.T. Yue, P. Ren, and Z.H. Liu: COM 2019, Vancouver, Canada, 2019, Paper ID, 589867.
[18] C. Jeffrey, A. Hallet, and H. Kurt. J. Phys. Chem., 1980, vol. 84(13), pp. 1699-1704.
[19] Z.Q. Yan, Z.A. Wang, X.F. Wang, H. Liu, and T.R. Qiu. Trans. Nonferrous Met. Soc. China., 2015, vol. 25, pp. 3490-3497.
[20] S. Aggoun, M. Cheikh-Zouaoui, N. Chikh, and R. Duval. Constr. Build. Mater., 2008, vol. 22, pp. 106-110.
[21] F. Yu, Z.H. Liu, F.C. Ye, L.G. Xia, and A. Jokilaakso: JOM., 2020, vol. 73, pp. 694-702.
M. Shevchenko and E. Jak: CALPHAD: Comput. Coupling Phase Diagrams Thermochem., 2020, vol. 71, Paper ID:102011.
[23] L. Feng, Y.M. Sun, X.L. Zhao, R.M. Wang, K. Zhang, and J.Y. Yang: Trans. Nonferrous Met. Soc. China., 2015, vol. 25, pp. 3553-3559.
[24] A.B. Kulakov, A.N. Maljuk, M. Sofifin, C.T. Lin, B. Keimer, and M. Jansen. J. Solid State Chem., 2004, vol. 177, pp. 3274-3280
[25] A. Roine. HSC Chemistry, Outokumpu Reasearch Oy, Pori, Finland, 2020.
[26] S.E. Perez-Fontes, M. Perez-Tello and L.O. Prieto-Lopez: Miner. Metall. Process., 2007, vol. 24(4), pp. 1-7.
[27] R.O. Suominen, A. Jokilaakso, P. Taskinen and K.R. Lilius: Scand. J. Metall., 1991, vol. 20(4), pp. 245-250.
[28] M. Perez-Tello, H.Y. Sohn, and J. Lottiger. Miner. Metall. Process., 1999, vol. 16(2), pp. 1-7.
[29] M. Perez-Tello, H.Y. Sohn, and P.J. Smith: Metall. Mater. Trans. B, 2001, vol. 32B, pp. 869-886.
[30] M. Perez-Tello, H.Y. Sohn, K.S. Marie and A. Jokilaakso. Metall. Mater. Trans. B, 2001, vol. 32B, pp. 847-868
[31] M. Pérez-Tello, V.R. Parra-Sánchez, V.M. Sánchez-Corrales, A. Gomez-Alvarez, F. Brown-Bojorquez, P.A. Parra-Figueroa, E.R. Balladares-Varela and E.A. Araneda-Hernandez. Metall. Mater. Trans. B, 2018, vol. 47(2), pp. 627-643.
[32] R.L. Jones, and K.H. Stern. Ind. Eng. Chem. Prod. Res. Dev., 1980, vol. 79, pp. 158-165.
M. Petrowsky and R. Frech (2009) J. Phys. Chem. B 113(17), pp. 5996-6000.
[34] W.G. Davenport, M. King, M. Schlesinger, and A.K. Biswas: Extractive Metallurgy of Copper, 4th ed., Elsevier Science, Oxford, 2002.
[35] M. Goto, S. Kawakita, N. Kikumoto, and O. Iida. JOM, 1986, vol. 38 (9), pp. 43-46.
[36] Y. Takeda, S. Nakazawa, and A. Yazawa: Can. Metall. Q., 1980, vol. 19 (3), pp. 297-305.
F. Tanaka, O. Iida, and Y. Takeda: International Symposium on Metallurgical and Materials Processing, San Diego, 2003, vol. 2, pp. 495–508.
[38] S. Nikolic, P.C. Hayes, E. Jak. Metall. Mater. Trans. B, 2009, vol. 40(6), pp. 892-899.
N. Mihara, D. Kuchar, Y. Kojima, and H. Matsuda (2007) J. Mater. Cycles Waste Manag. vol. 9(1), pp. 21-26.
Z.A. Wang, W. Yang, H. Liu, H. Jin, H.Q. Chen, K. Su, Y.J. Tu, and W.L. Wang: J. Anal. Appl. Pyrol., 2019, vol. 142, Paper ID: 104617.
[41] I. Gruncharov, Y. Pelovski, G. Bechev, and I. Dombalov. J. Therm. Anal. Calori.,1988, vol. 33(3), pp. 597-602.
M. Viale, O. Martin, F. Muratoria, U. Bertezzoloa, J. Perezc, C. Partemioc, and J. Usartb. Proc. SPIE, 2007, vol. 6541, pp. 65410H1-11.
M.E. Schlesinger, M.J. King, K.C. Sole, and W.G. Davenport: Extractive Metallurgy of Copper (Fifth Edition), 2011
[44] A.Yazawa. J. Min. Inst. Jpn., 1956, vol. 72, pp. 305-311.
Acknowledgments
The research was funded by the National Natural Science Foundation of China (No. 52004341) and the National Solid Waste Project (No. 2018YFC1902503). The financial support from the “111” project (Green and Value-Added Metallurgy of Non-ferrous Resources) and Central South University Starting-up fund (Faculty No. 217030) are also acknowledged. The authors also thank Mr Peng Ren for the help in the experimental part.
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Manuscript submitted February 16, 2021; accepted June 30, 2021.
Rights and permissions
About this article
Cite this article
Yu, F., Xia, L., Zhu, Y. et al. Reaction Behavior of Na2SO4-Containing Copper Matte Powders in a Simulated Flash Converting Process. Metall Mater Trans B 52, 3468–3476 (2021). https://doi.org/10.1007/s11663-021-02275-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-021-02275-7