Abstract
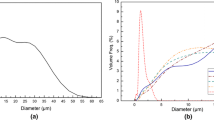
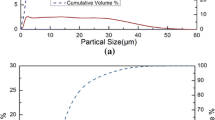
The kinetic modeling for the carbothermal reduction reaction rate in quartz and carbon black pellets is studied at different temperatures, under varying CO partial pressures in ambient atmosphere, varying carbon contents, different quartz particle sizes, and different crucible opening areas. Carbon black is produced by the cracking of natural gas. The activation energy of the SiC-producing step was determined to be 594 kJ/mol. The averaged pre-exponential factor A obtained from 1898 K, 1923 K, and 1948 K (1625 °C, 1650 °C, and 1675 °C) is 2.62E+16 min−1. The reaction rate of the gas-solid interface factor, fix-C content (Xfix-C), temperature (T), and CO partial pressure (XCO) can be expressed as follows:













Similar content being viewed by others
References
F. Li and M. Tangstad: In 2016 Annual Meeting of The Minerals, Metals & Materials Society, John Wiley & Sons, Inc., Nashville, USA, 2016.
F. Li and M. Tangstad, Metallurgical and Materials Transactions B 2017, vol. 48, pp. 853-869.
A. W. Weimer, K. J. Nilsen, G. A. Cochran, R. P. Roach, AIChE Journal 1993, vol. 39, pp. 493–503.
V.L. Kuznetsova, V.A. Dmitrenko, and A.D. Kokurin: Zh. Vses. Khim. O-Va (Proc. Mendeleev Chem. Soc.), 1980, vol. 25, pp. 118–19.
A. Agarwal and U. Pad, Metallurgical and Materials Transactions B 1999, vol. 30, pp. 295-306.
W. Kjell: Kinetics of reactions between silica and carbon. (Norwegian University of Science and Technology, Trondheim, Norway, 1990).
H. Lindgaard: High Temperature Decomposition of Methane on Quartz Pellets. (Norwegian University of Science and Technology, Trondheim, Norway, 2015).
H. L. Friedman, Journal of Polymer Science Part C: Polymer Symposia 1964, vol. 6, pp. 183-195.
F. Ni: Kinetics of the reaction between quartz and silicon carbide in different gas atmospheres. (Norwegian University of Science and Technology, Trondheim, Norway, 2015).
R. Altorfer, Thermochimica Acta 1978, vol. 24, pp. 17-37.
S. Li and P. Järvelä, Journal of Polymer Science Part B: Polymer Physics 2001, vol. 39, pp. 1525-1528.
J.R. Opfermann, E Kaisersberger and HJ Flammersheim, Thermochimica acta 2002, vol. 391, pp. 119-127.
V. Andersen: Reaction mechanism and kinetics of the high temperature reactions in the silicon process. (Norwegian University of Science and Technology, Trondheim, Norway, 2010).
A. Khawam and D. R. Flanagan, Journal of Pharmaceutical Sciences 2006, vol. 95, pp. 472-498.
A. Wilkinson and A. D. McNaught: Compendium of Chemical Terminology. (Blackwell Science, Oxford, 1997).
Acknowledgments
The authors acknowledge Elkem and Norwegian Research Council for the financial support through the project “Silicon Production with use of Natural Gas (235123).”
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted February 11, 2017.
Rights and permissions
About this article
Cite this article
Li, F., Tangstad, M. Kinetic Modeling of the Reaction Rate for Quartz and Carbon Black Pellet. Metall Mater Trans B 49, 1101–1108 (2018). https://doi.org/10.1007/s11663-018-1203-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-018-1203-1