Abstract
Summary
This study demonstrated a large treatment gap in elderly subjects experiencing fragility fracture in Spanish primary care, a low treatment persistence among subjects who do receive treatment, and more than one-quarter having no follow-up visits post-fracture. These data highlight the need to improve secondary fracture prevention in primary care.
Purpose
To describe osteoporosis (OP) treatment patterns and follow-up in subjects with fragility fracture seen in Spanish primary care (PC).
Methods
This observational, retrospective chart review included subjects aged ≥ 70 years listed in the centers' records (November 2018 to March 2020), with ≥ 1 fragility fracture and prior consultation for any reason; subjects who had participated in another study were excluded. Outcomes included OP treatments and follow-up visits post-fragility fracture.
Results
Of 665 subjects included, most (87%) were women; overall mean (SD) age, 82 years. Fewer than two thirds (61%) had received any prior OP treatment (women, 65%; men, 38%); of these, 38% had received > 1 treatment (women, 25%; men, 13%). Among treated subjects, the most frequent first-line treatments were alendronate (43%) and RANKL inhibitor denosumab (22%), with a higher discontinuation rate and shorter treatment duration observed for alendronate (discontinuation, 42% vs 16%; median treatment duration, 2.5 vs 2.1 years). Over one-quarter (26%) of subjects had no follow-up visits post-fragility fracture, with this gap higher in women than men (35% versus 25%). The most common schedule of follow-up visits was yearly (43% of subjects with a fragility fracture), followed by half-yearly (17%) and biennial (10%), with a similar trend in men and women. Most OP treatments were prescribed by PC physicians, other than teriparatide and zoledronate.
Conclusions
Across Spanish PC, we observed a large gap in the treatment and follow-up of elderly subjects experiencing a fragility fracture. Our data highlights the urgent need to improve secondary fracture prevention in PC.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Fragility fractures, defined as low energy trauma resulting from a fall from standing height or less, are a sign of underlying osteoporosis (OP), and increase the risk of further fractures [1, 2]. The risk of fragility fractures increases with age and, due to the fall in estrogen levels during the menopause which causes rapid acceleration in bone loss, is higher for women than men [3,4,5]. Other risk factors include a family history of OP, certain medications (including glucocorticoids), excessive alcohol intake, smoking, low body mass index, calcium and vitamin D deficiency, and sedentarism [6].
In Europe, fragility fractures are the fourth leading cause of chronic disease morbidity after ischemic heart disease, dementia and lung cancer [7], with an estimated prevalence of 6.8% and 22.5% in men and women aged ≥ 50 years, respectively [8]. We previously reported comparable prevalence from the PREFRAOS study of Spanish Primary Care (PC); men, 8.0%; women, 24.1%) [9]. The International Osteoporosis Foundation (IOF) estimate the annual incidence of fragility fractures in the largest five countries of the European Union plus Sweden (EU6) will increase by 23% between 2017 and 2030, from 2.7 million to 3.3 million [8]. Moreover, the total costs associated with fragility fractures in the European Union were estimated to be € 29.6 billion in 2010 and are expected to increase by approximately 54% in 2030 [7]. Similarly, based on existing disease management patterns across 8 European countries, Yeh and colleagues estimated annual fracture rates and associated costs in women aged ≥ 70 years will increase by 44% between 2020 and 2040 (from 1.2 million fractures costing € 12.8 billion to 2.8 million fractures costing € 18.4 billion) [10].
Despite these costs, and guidelines recommending OP treatment in subjects who have sustained a fragility fracture [11,12,13], OP treatment rates are low and many studies report a treatment gap in subjects at high fracture risk. For example, the IOF reported the treatment gap in 2017 to be 73% in women and 63% in men, with most (women, 89%; men 97%) treatment naïve at the time of their first fracture[8]. Similarly, a cross-sectional study of women aged ≥ 70 years seen in PC across 8 European countries reported an overall treatment gap of 75%, and a higher gap among women without an OP diagnosis (94%) versus those with an OP diagnosis (31%) [14]. Phase A of our study of Spanish PC data previously reported a treatment gap of 39% in men and women aged ≥ 70 years experiencing a fragility fracture [9].
Data reporting secondary fracture prevention by PC physicians is sparse. Our retrospective observational study aimed to estimate the prevalence of fragility fractures in Spanish PC and describe the characteristics, risk factors, co-morbidities, and OP diagnosis and treatment rates in subjects with a fragility fracture. With data regarding the prevalence of fragility fractures and OP diagnosis and treatment rates already published [9]; we describe OP treatment patterns and follow-up in subjects with at least a fragility fracture seen in Spanish PC.
Methods
Study design
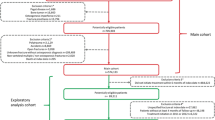
The observational, retrospective single-country PREvalence of FRAgility fractures and OSteoporosis treatment (PREFRAOS) study was conducted in 30 Spanish PC centers across 14 Spanish Regions. The study design and methodology have been published previously [9].
In brief, the study comprised two phases: Phase A included subjects aged ≥ 70 years listed in the center’s medical records between November 2018 and March 2020; Phase B included subjects from Phase A with at least a recorded fragility fracture and prior consultation at the center for any reason. Fragility fractures were identified using International Classification of Diseases (ICD) codes (ICD-9 and ICD-10) and/or open fields, according to the characteristics of each center’s database. Study outcomes were collected retrospectively from subjects’ medical records. A full list of study outcomes is given in Supplementary Methods Section. Results from Phase A and some results from Phase B were published previously [9]. Herein, we describe OP treatment patterns and follow-up in subjects with a fragility fracture included in Phase B.
This study was conducted according to the standards of the Declaration of Helsinki principles, its later amendments and with Good Clinical Practice guidelines. All subjects provided written or oral informed consent before enrolment.
Statistical analysis
All analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA). For categorical variables, the frequency and percentage with 95% confidence interval (CI) are presented. For continuous variables, summary statistics included the number of subjects, mean, median, standard deviation (SD) or standard error (SE), 25th percentile (Q1), 75th percentile (Q3), minimum, and maximum.
Results
Demographic and clinical characteristics
As reported previously, 665 subjects were included in Phase B with a mean (SD) age of 82 (6.6) years, and most subjects were women and had experienced only one fragility fracture (87% and 72%, respectively) [9]. Mean (SD) age at the time of first fragility fracture was 76 (8.6) years, with the first fracture reported to be related to a fall in over three quarters (78%) of the population. Mean (SD) time from first to second fragility fracture was numerically shorter in men than women (3.4 [4.9] versus 5.3 [4.7] years] (Table 1).
Osteoporosis treatments
Overall, 61.1% (406/665) of subjects with a fragility fracture had received prior OP treatment and three-quarters (501/665) had received calcium or vitamin D supplements. Treatment rates were higher among women than men (any prior OP treatment, 369/567 [65.1%] vs 37/98 [37.8%]; calcium or vitamin D, 440/567 [77.6%] vs 61/98 [62.2%]). Among subjects who had received prior OP treatment, 37.9% (154/406) had received more than one therapy, with women more likely than men to have received multiple therapies (141/567 [24.9%] vs 13/98 [13.3%]). The most common first-line OP treatments were alendronate (175/406, 43.1%) followed by RANKL inhibitor denosumab (89/406, 21.9%)] (Fig. 1).
At study inclusion, fewer than half (287/665 [43.2%)] of subjects with a fragility fracture were on active OP treatment, with treatment rates higher in women than men (45.7% vs 28.6%) [9] and the most common treatments being denosumab and alendronate (149/287 [51.9%] and 97/287 [33.8%], respectively). Denosumab use was numerically higher in women than in men ([138/259 [53.3%] vs 11/28 [39.3%]) (Table 2).
OP treatment rates by OP diagnosis are displayed in Table 2. Compared with subjects without a diagnosis, OP treatment rates (at any time) were higher in subjects with an OP diagnosis (79.4% vs 25.9%). The same trend was observed in women and in men: 80.8% of women and 66.7% of men with an OP diagnosis had received an OP treatment compared with 29.1% of women and 16.1% of men without an OP diagnosis. A higher OP treatment rate was reported in women (with and without OP diagnosis) compared to men.
As reported previously, almost half (298/661 [45.1%]) of all OP treatments prescribed were discontinued, most commonly at the decision of the treating specialist (95/298, 31.9%) or the subject (62/298, 20.8%), or due to tolerability issues (57/298, 19.1%) [9]. A total of 42.3% (96/227) of alendronate prescriptions were discontinued, with a numerically higher discontinuation rate in women than men (94/212 [44.3%] vs. 2/15 [13.3%]). Alendronate was most commonly discontinued at the decision of the treating specialist or the subject (23/96 [24.0%] and 21/96 [21.9%], respectively), or due to tolerability issues (21/96 [21.9%]). Only 15.5% (27/174) of denosumab prescriptions were discontinued, with a numerically lower discontinuation rate in women than men (23/159 [14.5%] vs 4/15 [26.7%]); the most common reason for discontinuation was the subject’s decision (17/27 [63.0%]). Reasons for prescription discontinuations are shown in Table SI1.
Median (Q1, Q3) treatment duration was 2.5 (0.6, 5.3) years for alendronate (higher in women vs men: 2.5 [0.6, 5.6] vs 1.0 [0.4, 4.1]) and 2.1 (0.7, 4.1) years for denosumab (also higher in women vs men: 2.3 [0.7, 4.3] vs. 1.0 [0.0, 2.8] years).
PC physicians were the most common prescribers of all OP treatments except teriparatide and zoledronate, for whom rheumatologists were the most common prescribers (Fig. 2).
Figure 3 summarizes OP treatment rates after the first, second and third fragility fracture. Subjects with one fracture had a lower treatment rate than those with multiple fractures. Only 18% of subjects initiated treatment within 90 days of a first fracture, falling to 12% and 13% after a second and third fracture, respectively. Among subjects with a second fracture, over one-quarter were continuing prior OP treatment at the time of their second fracture; and among subjects with a third fracture, almost half were continuing prior OP treatment at the time of their third fracture. Compared with women, men were less likely to receive treatment or continue previous treatment after their first fragility fracture (21.4% (21/98) vs 30.3% (172/567) and 2.0% (2/98) vs 12.2% (69/567), respectively).
OP follow-up visits and diagnostic test used
Over one-quarter (174/665 [26.2%]) of subjects with a fragility fracture did not have follow-up visits post-fracture. Men were less likely to have follow-up visits than women (no follow-up visits: 34/98 [34.7%] vs 140/567 [24.7%]). The frequency of follow-up visits is summarized in Fig. 4. The most common schedule of OP follow-up visits was yearly, followed by half-yearly and biennial, with a similar trend in men and women. Older subjects (≥ 80 years) were more likely to have yearly follow-up visits (164/377 [43.5%]) than younger subjects (48/116 [41.4%] and 71/172 [41.3%] in subjects aged 70–74 and 75–79 years, respectively) and less likely to have biennial visits (32/377 [8.5%] vs 14/116 [12.1%] and 19/172 [11.0%], respectively).
The most commonly used follow-up tests were blood tests (85.9% [571/665] of subjects), radiography (48.4% [322/665]) and densitometry (39.2% [261/665]). There was no difference in rates of follow-up tests between men and women': blood tests, 86.1% (488/567) of women and 84.7% (83/98) of men; radiography, 50.3% (285/567) and 37.8% (37/98), respectively; densitometry, 41.6% (236/567) and 25.5% (25/98). Densitometry tests were more likely to be performed in younger subjects, being performed in half (50.0% [58/116]) of subjects aged 70–74 years vs 44.2% (76/172) and 33.7% (127/377) of those aged 75–79 and ≥ 80 years, respectively. Radiography tests were less common among younger subjects (41.4% [48/116] of subjects aged 70–74 years vs 51.2% [88/172] and 49.3% [186/377] of those aged 75–79 and ≥ 80 years, respectively).
Discussion
We report real-world data regarding the clinical and therapeutic management of secondary fracture prevention by PC physicians in 665 subjects aged 70 years or over. Subjects in our study had a mean age of 82 years and had typically experienced only one fragility fracture. The first fracture was reported at a mean age of 76 years and in most (78%) cases was related to a fall, with similar results by sex, except for a shorter time between first and second fragility fracture in men.
The most commonly prescribed first-line OP treatments were alendronate followed by denosumab, with treatment patterns similar in men and women. A similar trend was observed in a cross-sectional study of women aged ≥ 70 years seen in PC across 8 European countries that reported a higher use of bisphosphonates compared to denosumab as first-line OP treatment (66% [58% oral bisphosphates and 8% parenteral bisphosphonates] vs 28%) [14]. The median duration of alendronate treatment in our study was slightly higher than for denosumab (2.5 and 2.1 years, respectively). It has been suggested a drug holiday for bisphosphonates of 3 to 5 years as they are retained in bone for extended periods with extended anti-fracture effects after discontinuation. However, the optimal duration is undetermined and should be re-evaluated after 1–2 years, depending on BF used [15]. Specifically, treatment with alendronate has shown maintenance of bone density among subjects who discontinued after 5 years [16]. Conversely, the bone-maintaining effects of denosumab wear off soon after stopping [17], with subjects not receiving subsequent OP medication experiencing 5–11% loss in bone mineral density in the first year after denosumab is stopped. Compared with denosumab, we observed a higher discontinuation rate among subjects treated with alendronate (42% vs 16%), possibly due to concerns regarding the potential side effects of bisphosphonates, including osteonecrosis of the jaw and/or atypical subtrochanteric femur fractures [18]. Following treatment discontinuation, bone mineral density (BMD) and fracture risk can be expected to return to baseline or worse slowly, in the case of bisphosphonates, or quickly, in the case of non-bisphosphonates [19]. This highlights the important role that specialists and primary care physicians play in increasing adherence, monitoring patients with bone markers and BMD testing and providing adequate instructions, feedback and support, and educational materials and sessions [20].
Treatment guidelines recommend the effect of OP treatment be confirmed by BMD and the absence of new fracture [13], therefore follow-up visits in subjects with OP are vital. However, over one-quarter of subjects in our study did not have follow-up visits, with the most common schedule of OP follow-up visits being yearly. While there is no clear evidence to guide the frequency of follow-up recommendations following a diagnosis of OP and treatment initiation, it is reasonable to assess response, adherence and tolerability to therapy at least once, after no less than 24 months [21].
In general, the most common prescribers of OP treatment in our study were PC physicians, followed by rheumatologists and traumatologists, a finding which is consistent with other studies conducted in the PC setting [22, 23]. As expected, teriparatide and zoledronate were most commonly prescribed in a hospital setting, Consistent with previous studies, gynecologists were most likely to prescribe bazedoxifene, followed by raloxifene [23].
While a number of effective and well tolerated OP treatments are available in clinical practice, the OP treatment gap has remained stable or increased in recent years [24]. It is important to highlight the high OP treatment gap we observed in Spanish PC, with over half (57%) of subjects not receiving OP treatment at study entry despite having suffered a fragility fracture. Our results are consistent with previous studies which have reported a treatment gap between 53 to 91% [14, 24, 25]. Moreover, OP diagnosis reduced the treatment gap (at study entry) from 87% in subjects without a diagnosis to 41% in those with a diagnosis9 (74% vs 21% treatment gap at any time during the study). Similarly, a retrospective cross-sectional study of women aged ≥ 70 years seen in PC across 8 European countries reported a treatment gap of 94% in subjects without an OP diagnosis vs 31% in those with a diagnosis [14]. In addition, we observed a higher treatment gap (at study entry) in men compared with women (71% vs 54%), consistent with 2014–2015 data from secondary fracture prevention in Europe [24]. However, a European cohort study of secondary fracture prevention subjects reported a higher treatment gap in women vs men (overall, 73% vs 63%; being higher in women vs men in France, Italy, Spain, Sweden and the UK, while it was maintained similar between sex in Germany [8]. Among subjects in Sweden untreated within one year of fracture, the same study reported the treatment gap in treatment naïve subjects to be higher for men vs women (96% vs 89%), and the treatment gap in subjects with prior treatment to be slightly lower in men vs women (47% vs 52%). Thus, studies about treatment gap by sex showed inconsistent results.
It should be noted that the treatment gap was higher among subjects with a single fracture than those with multiple fractures. This fact could be explained because these are the group of subjects with a much higher risk of fracture. According to these results, we could suggest that an improvement of early diagnostic, OP management and follow-up of subjects in secondary fracture prevention in PC is needed to reduce the number of patients at higher risk.
Almost half of the subjects in our study discontinued OP treatment, mostly by decision of the treating specialist/investigator or subject, or due to tolerability problems or non-compliance. Several factors could have contributed to this high discontinuation rate, including concerns about rare side effects, comorbidities and inadequate long-term efficacy. Indeed, ineffectiveness of anti-resorptive drugs has been associated with an increase in the number of untreated subjects [11]. Possible factors contributing to non-compliance include difficulty in perceiving the benefits of therapy, side effects, medication costs, lack of patient motivation and/or education. In a retrospective cohort study of women aged ≥ 50 years receiving OP treatment in Spain; 1-year persistence ranged from 35.8% (strontium ranelate) to 65.8% (denosumab), before dropping to only 10.3% (strontium ranelate) to 45.4% (denosumab) by 2 years. Compared to alendronate, the risk of discontinuation within 1 year was lower for teriparatide and denosumab, and only denosumab showed a reduced risk of discontinuation at 2-years [26]. Reported adherence to OP treatment varies widely, according to the method used. For example, when assessed using the Morisky–Green questionnaire, a study from Spanish PC reported over two-thirds (68.7%) of women to be non-compliant compared with 11.2% when adherence was self-reported by the study participants [27]. Related to this, Martínez-Laguna et al. [28] reported that combining the Morisky–Green questionnaire and determination of the carboxyterminal telopeptide of type I collagen allowed better assessment [29]. While the International Osteoporosis Foundation and European Calcified Tissue Society Working Group advocate the use of bone remodeling markers to assess adherence to bisphosphonates [30], these are rarely utilised in PC. While difficult and complex to measure in clinical practice [27], improving adherence, and, in turn, persistence, is vital for treatment effectiveness. Related to this, Yeh et al. [10] estimated the impact of improvements in risk assessment and/or treatment adherence on the economic burden of OP across eight European countries They estimated that 50% increases in treatment adherence and fracture risk assessment would result in an 18% reduction in fracture incidence and a 15% reduction in net costs (saving of € 2.8 billion) by 2040. Similarly, 10% increases in treatment adherence and fracture risk assessment could result in an 8.4% reduction in fracture incidence and a 7.6% reduction in net costs (saving of € 1.4 billion) by 2040. Actions to improve adherence include patient education with counselling, adherence monitoring with feedback and dose simplification (consisting of flexible dosing regimen) [31].
This study has strengths and limitations. For example, the inclusion of centers from different Spanish regions provides a more representative sample of the population in Spain. By contrast, the observational and retrospective study design contribute to missing data and is a limitation. Moreover, the small sample of subjects in some subgroup analysis is also a limitation. Finally, the PC centers that participated tended to have high OP experience which could bias the treatment rates compared to PC centers without OP experience.
Conclusions
This observational study reports suboptimal secondary fracture prevention in Spanish primary care, with a large treatment gap in elderly subjects experiencing fragility fracture, low treatment persistence among subjects who do receive treatment, and more than one-quarter having no follow-up visits post-fracture. These data highlight the need to improve secondary fracture prevention in primary care by enhancing the early diagnostic, assessment of risk of fractures, management of OP treatment, adherence, and follow-up of OP subjects specially in subgroups with higher risk factors as those with multiple fractures.
Data availability
Qualified researchers may request data from Amgen clinical studies. Complete details are available at the following: https://wwwext.amgen.com/science/clinical-trials/clinicaldata-transparencypractices/clinical-trial-data-sharing-request/
References
Fragility fractures | International Osteoporosis Foundation. https://www.osteoporosis.foundation/health-professionals/fragility-fractures. Accessed 6 Sep 2023
Klotzbuecher CM, Ross PD, Landsman PB et al (2000) Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res 15:721–739. https://doi.org/10.1359/jbmr.2000.15.4.721
Kanis JA, Johnell O, De Laet C et al (2004) A meta-analysis of previous fracture and subsequent fracture risk. Bone 35:375–382. https://doi.org/10.1016/j.bone.2004.03.024
Yong E-L, Logan S (2021) Menopausal osteoporosis: screening, prevention and treatment. Singapore Med J 62 159–166 https://doi.org/10.11622/smedj.2021036
Kanis JA, Norton N, Harvey NC et al (2021) SCOPE 2021: a new scorecard for osteoporosis in Europe. Arch Osteoporos 16:82. https://doi.org/10.1007/s11657-020-00871-9
International Osteoporosis Foundation (IOF) Risk factors. https://www.osteoporosis.foundation/patients/about-osteoporosis/risk-factors
Hernlund E, Svedbom A, Ivergård M et al (2013) Osteoporosis in the European Union: medical management, epidemiology and economic burden. A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch Osteoporos 8:136. https://doi.org/10.1007/s11657-013-0136-1
Borgström F, Karlsson L, Ortsäter G et al (2020) Fragility fractures in Europe: burden, management and opportunities. Arch Osteoporos 15:59. https://doi.org/10.1007/s11657-020-0706-y
Martínez-Laguna D, Carbonell C, Bastida J-C et al (2022) Prevalence and treatment of fragility fractures in Spanish primary care: PREFRAOS study. Arch Osteoporos 17:93. https://doi.org/10.1007/s11657-022-01124-7
Yeh EJ, Gitlin M, Sorio F, McCloskey E (2023) Estimating the future clinical and economic benefits of improving osteoporosis diagnosis and treatment among postmenopausal women across eight European countries. Arch Osteoporos 18:68. https://doi.org/10.1007/s11657-023-01230-0
Kanis JA, Svedbom A, Harvey N, McCloskey EV (2014) The osteoporosis treatment gap. J Bone Miner Res 29:1926–1928. https://doi.org/10.1002/jbmr.2301
Cosman F, de Beur SJ, LeBoff MS et al (2014) Clinician’s Guide to Prevention and Treatment of Osteoporosis. Osteoporos Int 25:2359. https://doi.org/10.1007/s00198-014-2794-2
Riancho JA, Peris P, González-Macías J et al (2022) Executive summary clinical practice guideline of postmenopausal, glucocortcioid-induced and male osteoporosis (2022 update). Spanish Society for Bone and Mineral Metabolism Investigation (SEIOMM). Rev Clin Esp (Barc) 222:432–439. https://doi.org/10.1016/j.rceng.2021.12.008
McCloskey E, Rathi J, Heijmans S et al (2021) The osteoporosis treatment gap in patients at risk of fracture in European primary care: a multi-country cross-sectional observational study. Osteoporos Int 32:251–259. https://doi.org/10.1007/s00198-020-05557-z
Wang M, Wu Y-F, Girgis CM (2022) Bisphosphonate Drug Holidays: Evidence From Clinical Trials and Real-World Studies. JBMR Plus 6:e10629. https://doi.org/10.1002/jbm4.10629
Black DM, Schwartz AV, Ensrud KE et al (2006) Effects of Continuing or Stopping Alendronate After 5 Years of TreatmentThe Fracture Intervention Trial Long-term Extension (FLEX): A Randomized Trial. JAMA 296:2927–2938. https://doi.org/10.1001/jama.296.24.2927
Anastasilakis AD, Makras P, Yavropoulou MP et al (2021) Denosumab Discontinuation and the Rebound Phenomenon: A Narrative Review. J Clin Med 10:152. https://doi.org/10.3390/jcm10010152
Miller PD (2016) Management of severe osteoporosis. Expert Opin Pharmacother 17:473–488. https://doi.org/10.1517/14656566.2016.1124856
LeBoff MS, Greenspan SL, Insogna KL et al (2022) The clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int 33:2049–2102. https://doi.org/10.1007/s00198-021-05900-y
Papaioannou A, Kennedy CC, Dolovich L et al (2007) Patient adherence to osteoporosis medications: problems, consequences and management strategies. Drugs Aging 24:37–55. https://doi.org/10.2165/00002512-200724010-00003
Sweet MG, Sweet JM, Jeremiah MP, Galazka SS (2009) Diagnosis and Treatment of Osteoporosis. afp 79:193–200
de Felipe R, Cáceres C, Cimas M et al (2010) Características clínicas de los pacientes con tratamiento para la osteoporosis en un centro de Atención Primaria: ¿a quién tratamos en nuestras consultas? Aten Primaria 42:559–563. https://doi.org/10.1016/j.aprim.2009.10.022
Martinez-Laguna D, Sancho-Almela F, Cano-Collado E, Gardeñes-Moron M, Morró J, Cos X (2011) Uso adecuado en Atención Primaria de los fármacos antirresortivos frente a la osteoporosis. Rev Osteoporos Metab Min 3(2):77–83
Skjødt MK, Khalid S, Ernst M et al (2020) Secular trends in the initiation of therapy in secondary fracture prevention in Europe: a multi-national cohort study including data from Denmark, Catalonia, and the United Kingdom. Osteoporos Int 31:1535–1544. https://doi.org/10.1007/s00198-020-05358-4
Mendis AS, Ganda K, Seibel MJ (2017) Barriers to secondary fracture prevention in primary care. Osteoporos Int 28:2913–2919. https://doi.org/10.1007/s00198-017-4131-z
Reyes C, Tebe C, Martinez-Laguna D et al (2017) One and two-year persistence with different anti-osteoporosis medications: a retrospective cohort study. Osteoporos Int 28:2997–3004. https://doi.org/10.1007/s00198-017-4144-7
Carbonell Abella C, Guañabens Gay N, Regadera Anechina L et al (2011) Analysis of Therapeutic Compliance in Women With Osteoporosis. Reumatología Clínica (English Edition) 7:299–304. https://doi.org/10.1016/j.reumae.2010.12.001
Martinez-Laguna D, Soria-Castro A, Carbonell-Abella C et al (2019) Validation of fragility fractures in primary care electronic medical records: A population-based study. Reumatol Clin 15:e1–e4. https://doi.org/10.1016/j.reuma.2017.10.013
Martínez-Laguna D, Nogués X, Carbonell-Abella C, Soria Castro A, Orozco López P, Poza Martínez R et al (2019) El papel de la determinación de un marcador de resorción, el telopéptido carboxiterminal del colágeno I, en la valoración del cumplimiento terapéutico en pacientes tratadas con bifosfonatos orales. Rev Osteoporos Metab Miner 11:19–24. https://doi.org/10.4321/S1889-836X2019000100004
Diez-Perez A, Naylor KE, Abrahamsen B et al (2017) International Osteoporosis Foundation and European Calcified Tissue Society Working Group. Recommendations for the screening of adherence to oral bisphosphonates. Osteoporos Int 28:767–774. https://doi.org/10.1007/s00198-017-3906-6
Hiligsmann M, Cornelissen D, Vrijens B et al (2019) Determinants, consequences and potential solutions to poor adherence to anti-osteoporosis treatment: results of an expert group meeting organized by the European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases (ESCEO) and the International Osteoporosis Foundation (IOF). Osteoporos Int 30:2155–2165. https://doi.org/10.1007/s00198-019-05104-5
Acknowledgements
Writing support was provided by Montse Sabaté, TFS HealthScience, funded by Amgen S.A, and Claire Desborough Amgen (Europe) GmbH.
Funding
This study was sponsored by Amgen S.A.
Author information
Authors and Affiliations
Consortia
Contributions
DML conceived and designed the study and contributed to the quality control and interpretation of the data, the data collection and to the drafting and revision of the manuscript. The remaining authors participated in data collection and manuscript review. All authors have read and agreed to the final published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by each ethics committee assigned to each center.
Consent to participate
Written or oral informed consent was obtained from all subjects before participating in this study.
Conflict of interest
DML: personal fees from Amgen, Lilly, Rubió, Gedeon Ritcher, Pierre Fabre, Grünenthal, Theramex and Italfarmaco. JCB: personal fees from AstraZeneca, Esteve, Boehringer Ingelheim, Bayer, Abbott, Almirall, Amgen, Chiessi, FAES, Grunenthal, GSK, Lilly, Menarini, MSD, Pfizer, Recordatti, Rovi, Servier; MG: personal fees from Amgen, Stada, Rubió, Grünenthal; RMP: fees from Amgen, UCB, Stada; FV: personal fees from Pfizer, Grünenthal, MSD, Amgen, GSK, Lilly, Bristol-Myers Squibb, Servier, P&G Pharmaceuticals, Novartis, Almirall, Sanofi, Mundipharma; ED and LC: Amgen employees.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Martínez-Laguna, D., Carbonell Abella, C., Bastida, JC. et al. Secondary fracture prevention in Spanish primary care: results of the PREFRAOS Study. Arch Osteoporos 19, 35 (2024). https://doi.org/10.1007/s11657-024-01394-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-024-01394-3