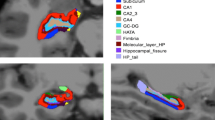
Abstract
During the caudo-rostral progression of Lewy pathology, the amygdala is involved relatively early in Parkinson’s disease (PD). However, lesser is known about the volumetric differences at the amygdala subdivisions, although the evidence mainly implicates the olfactory amygdala. We aimed to investigate the volumetric differences between the amygdala’s nuclear and sectoral subdivisions in the PD cognitive impairment continuum compared to healthy controls (HC). The volumes of nine nuclei of the amygdala were estimated with FreeSurfer (nuclear parcellation-NP) from T1-weighted images of PD patients with normal cognition (PD-CN), PD with mild cognitive impairment (PD-MCI), PD with dementia (PD-D), and HC. The appropriate nuclei were then merged to obtain three sectors of the amygdala (sectoral parcellation-SP). The nuclear and sectoral volumes were compared among the four groups and between the hyposmic and normosmic PD patients. There was a significant difference in the total amygdala volume among the four groups. In terms of nuclei, the bilateral cortico-amygdaloid transition area (CAT) and sectors superficial cortex-like region (sCLR) volumes of PD-MCI and PD-D were less than those of the PD-CN and HC. A linear discriminant analysis revealed that left CAT and left sCLR volumes classified the PD-CN and cognitively impaired PD (PD-CI: PD-MCI plus PD-D) with 90.7% accuracy according to NP and 85.2% accuracy to SP. Similarly, left CAT and sCLR volumes correctly identified the hyposmic and normosmic PD with 64.8% and 61.1% accuracies. Notably, the left olfactory amygdala volume successfully discriminated cognitive impairment in PD and could be used as neuroimaging-based support for PD-CI diagnosis.



Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AAA:
-
anterior amygdala area
- AB:
-
accessory basal nuclei of amygdala
- Ba:
-
basal nuclei of amygdala
- BLNG:
-
basolateral nuclear group of amygdala
- CAT:
-
cortico-amygdaloid transition area
- Ce:
-
central nucleus
- CMG:
-
centromedial group nucleus of amygdala
- Co:
-
cortical nuclei of amygdala
- HC:
-
healthy control
- La:
-
lateral nuclei of amygdala
- LB:
-
Lewy body
- LDA:
-
linear discriminant analysis
- LN:
-
Lewy neurite
- Me:
-
medial nuclei of amygdala
- OB:
-
Olfactory bulb
- PAC:
-
periamygdaloid cortex
- PD:
-
Parkinson’s disease
- PD-CI:
-
Parkinson’s disease with cognitive impairment
- PD-CN:
-
Parkinson’s disease with normal cognition
- PD-D:
-
Parkinson’s disease with dementia
- PD-MCI:
-
Parkinson’s disease with mild cognitive impairment
- PL:
-
paralaminar nuclei of amygdala
- sCLR:
-
superficial cortex-like region of amygdala
- UPDRS:
-
Unified Parkinson’s disease Rating Scale
References
Aarsland D, Bronnick K, Larsen JP et al (2009) Cognitive impairment in incident, untreated Parkinson disease: the Norwegian ParkWest study. Neurology 72(13):1121–1126. https://doi.org/10.1212/01.wnl.0000338632.00552.cb
Aarsland D, Kurz MW (2010) The epidemiology of dementia associated with Parkinson’s disease. Brain Pathol 20(3):633–639. https://doi.org/10.1111/j.1750-3639.2009.00369.x
Adler CH, Beach TG, Zhang N et al (2019) Unified Staging System for Lewy Body Disorders: Clinicopathologic Correlations and Comparison to Braak Staging. J Neuropathol Exp Neurol 78(10):891–899. https://doi.org/10.1093/jnen/nlz080
Amunts K, Kedo O, Kindler M et al (2005) Cytoarchitectonic mapping of the human amygdala, hippocampal region and entorhinal cortex: intersubject variability and probability maps. Anat Embryol (Berl) 210(5–6):343–352. https://doi.org/10.1007/s00429-005-0025-5
Arslan DB, Gurvit H, Genc O et al (2020) The cerebral blood flow deficits in Parkinson’s disease with mild cognitive impairment using arterial spin labeling MRI. J Neural Transm (Vienna) 127(9):1285–1294. https://doi.org/10.1007/s00702-020-02227-6
Azamat S, Arslan DB, Erdogdu E et al (2021) Detection of visual and frontoparietal network perfusion deficits in Parkinson’s disease dementia. Eur J Radiol 144:109985. https://doi.org/10.1016/j.ejrad.2021.109985
Baba T, Kikuchi A, Hirayama K et al (2012) Severe olfactory dysfunction is a prodromal symptom of dementia associated with Parkinson’s disease: a 3 year longitudinal study. Brain 135(Pt 1):161–169. https://doi.org/10.1093/brain/awr321
Beach TG, Adler CH, Lue L et al (2009) Unified staging system for Lewy body disorders: correlation with nigrostriatal degeneration, cognitive impairment and motor dysfunction. Acta Neuropathol 117(6):613–634. https://doi.org/10.1007/s00401-009-0538-8
Bertrand E, Lechowicz W, Szpak GM et al (2004) Limbic neuropathology in idiopathic Parkinson’s disease with concomitant dementia. Folia Neuropathol 42(3):141–150. https://www.ncbi.nlm.nih.gov/pubmed/15535032
Beyer MK, Janvin CC, Larsen JP, Aarsland D (2007) A magnetic resonance imaging study of patients with Parkinson’s disease with mild cognitive impairment and dementia using voxel-based morphometry. J Neurol Neurosurg Psychiatry 78(3):254–259. https://doi.org/10.1136/jnnp.2006.093849
Blair JC, Barrett MJ, Patrie J et al (2019) Brain MRI Reveals Ascending Atrophy in Parkinson’s Disease Across Severity. Front Neurol 10:1329. https://doi.org/10.3389/fneur.2019.01329
Boesveldt S, Verbaan D, Knol DL et al (2008) A comparative study of odor identification and odor discrimination deficits in Parkinson’s disease. Mov Disord 23(14):1984–1990. https://doi.org/10.1002/mds.22155
Bouchard TP, Malykhin N, Martin WR et al (2008) Age and dementia-associated atrophy predominates in the hippocampal head and amygdala in Parkinson’s disease. Neurobiol Aging 29(7):1027–1039. https://doi.org/10.1016/j.neurobiolaging.2007.02.002
Braak H, Braak E, Yilmazer D et al (1994) Amygdala pathology in Parkinson’s disease. Acta Neuropathol 88(6):493–500. https://doi.org/10.1007/BF00296485
Braak H, Del Tredici K, Bratzke H et al (2002) Staging of the intracerebral inclusion body pathology associated with idiopathic Parkinson’s disease (preclinical and clinical stages) J Neurol, 249 Suppl 3, III:1–5. https://doi.org/10.1007/s00415-002-1301-4
Braak H, Del Tredici K, Rub U et al (2003) Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging 24(2):197–211. https://doi.org/10.1016/s0197-4580(02)00065-9
Braak H, Ghebremedhin E, Rub U et al (2004) Stages in the development of Parkinson’s disease-related pathology. Cell Tissue Res 318(1):121–134. https://doi.org/10.1007/s00441-004-0956-9
Buchanan TW, Tranel D, Adolphs R (2003) A specific role for the human amygdala in olfactory memory. Learn Mem 10(5):319–325. https://doi.org/10.1101/lm.62303
Buter TC, van den Hout A, Matthews FE et al (2008) Dementia and survival in Parkinson disease: a 12-year population study. Neurology 70(13):1017–1022. https://doi.org/10.1212/01.wnl.0000306632.43729.24
Churchyard A, Lees AJ (1997) The relationship between dementia and direct involvement of the hippocampus and amygdala in Parkinson’s disease. Neurology 49(6):1570–1576. https://doi.org/10.1212/wnl.49.6.1570
Coughlin DG, Petrovitch H, White LR et al (2019) Most cases with Lewy pathology in a population-based cohort adhere to the Braak progression pattern but ‘failure to fit’ is highly dependent on staging system applied. Parkinsonism Relat Disord 64:124–131. https://doi.org/10.1016/j.parkreldis.2019.03.023
de la Monte SM, Wells SE, Hedley-Whyte T, Growdon JH (1989) Neuropathological distinction between Parkinson’s dementia and Parkinson’s plus Alzheimer’s disease. Ann Neurol 26(3):309–320. https://doi.org/10.1002/ana.410260302
De Olmos JS, Beltramino CA, Alheid G (2004) Amygdala and Extended Amygdala of the Rat: A Cytoarchitectonical, Fibroarchitectonical, and Chemoarchitectonical Survey. In G. Paxinos (Ed.), The Rat Nervous System (Third Edition) (pp. 509–603) Academic Press. https://doi.org/10.1016/B978-012547638-6/50020-1
Del Tredici K, Braak H (2012) Lewy pathology and neurodegeneration in premotor Parkinson’s disease. Mov Disord 27(5):597–607. https://doi.org/10.1002/mds.24921
Del Tredici K, Rub U, De Vos RA et al (2002) Where does parkinson disease pathology begin in the brain? J Neuropathol Exp Neurol 61(5):413–426. https://doi.org/10.1093/jnen/61.5.413
Emre M, Aarsland D, Brown R et al (2007) Clinical diagnostic criteria for dementia associated with Parkinson’s disease. Mov Disord 22(12):1689–1707 quiz 1837. https://doi.org/10.1002/mds.21507
Entis JJ, Doerga P, Barrett LF, Dickerson BC (2012) A reliable protocol for the manual segmentation of the human amygdala and its subregions using ultra-high resolution MRI. NeuroImage 60(2):1226–1235. https://doi.org/10.1016/j.neuroimage.2011.12.073
Fischl B (2012) FreeSurfer. Neuroimage 62(2):774–781. https://doi.org/10.1016/j.neuroimage.2012.01.021
Fischl B, Dale AM (2000) Measuring the thickness of the human cerebral cortex from magnetic resonance images. Proc Natl Acad Sci U S A 97(20):11050–11055. https://doi.org/10.1073/pnas.200033797
Fischl B, Salat DH, Busa E et al (2002) Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33(3):341–355. https://doi.org/10.1016/s0896-6273(02)00569-x
Gallagher M, Schoenbaum G (1999) Functions of the amygdala and related forebrain areas in attention and cognition. Ann N Y Acad Sci 877:397–411. https://doi.org/10.1111/j.1749-6632.1999.tb09279.x
Goldman JG, Postuma R (2014) Premotor and nonmotor features of Parkinson’s disease. Curr Opin Neurol 27(4):434–441. https://doi.org/10.1097/WCO.0000000000000112
Harding AJ, Stimson E, Henderson JM, Halliday GM (2002) Clinical correlates of selective pathology in the amygdala of patients with Parkinson’s disease. Brain 125(Pt 112431–2445. https://doi.org/10.1093/brain/awf251
Hawkes CH, Del Tredici K, Braak H (2007) Parkinson’s disease: a dual-hit hypothesis. Neuropathol Appl Neurobiol 33(6):599–614. https://doi.org/10.1111/j.1365-2990.2007.00874.x
Hawkes CH, Del Tredici K, Braak H (2009) Parkinson’s disease: the dual hit theory revisited. Ann N Y Acad Sci 1170:615–622. https://doi.org/10.1111/j.1749-6632.2009.04365.x
Hawkes CH, Shephard BC, Daniel SE (1997) Olfactory dysfunction in Parkinson’s disease. J Neurol Neurosurg Psychiatry 62(5):436–446. https://doi.org/10.1136/jnnp.62.5.436
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55(3):181–184. https://doi.org/10.1136/jnnp.55.3.181
Janak PH, Tye KM (2015) From circuits to behaviour in the amygdala. Nature 517(7534):284–292. https://doi.org/10.1038/nature14188
Jellinger KA (2008) A critical reappraisal of current staging of Lewy-related pathology in human brain. Acta Neuropathol 116(1):1–16. https://doi.org/10.1007/s00401-008-0406-y
Junque C, Ramirez-Ruiz B, Tolosa E et al (2005) Amygdalar and hippocampal MRI volumetric reductions in Parkinson’s disease with dementia. Mov Disord 20(5):540–544. https://doi.org/10.1002/mds.20371
Kadohisa M (2013) Effects of odor on emotion, with implications. Front Syst Neurosci 7:66. https://doi.org/10.3389/fnsys.2013.00066
Kalaitzakis ME, Graeber MB, Gentleman SM, Pearce RK (2008) Controversies over the staging of alpha-synuclein pathology in Parkinson’s disease. Acta Neuropathol 116(1):125–128 author reply 129–131. https://doi.org/10.1007/s00401-008-0381-3
Kempster PA, O’Sullivan SS, Holton JL et al (2010) Relationships between age and late progression of Parkinson’s disease: a clinico-pathological study. Brain 133(Pt 6):1755–1762. https://doi.org/10.1093/brain/awq059
Leverenz JB, Hamilton R, Tsuang DW et al (2008) Empiric refinement of the pathologic assessment of Lewy-related pathology in the dementia patient. Brain Pathol 18(2):220–224. https://doi.org/10.1111/j.1750-3639.2007.00117.x
Li X, Xing Y, Schwarz ST, Auer DP (2017) Limbic grey matter changes in early Parkinson’s disease. Hum Brain Mapp 38(7):3566–3578. https://doi.org/10.1002/hbm.23610
Litvan I, Aarsland D, Adler CH et al (2011) MDS Task Force on mild cognitive impairment in Parkinson’s disease: critical review of PD-MCI. Mov Disord 26(10):1814–1824. https://doi.org/10.1002/mds.23823
Litvan I, Goldman JG, Troster AI et al (2012) Diagnostic criteria for mild cognitive impairment in Parkinson’s disease: Movement Disorder Society Task Force guidelines. Mov Disord 27(3):349–356. https://doi.org/10.1002/mds.24893
Mattila PM, Rinne JO, Helenius H, Roytta M (1999) Neuritic degeneration in the hippocampus and amygdala in Parkinson’s disease in relation to Alzheimer pathology. Acta Neuropathol 98(2):157–164. https://doi.org/10.1007/s004010051064
Melzer TR, Watts R, MacAskill MR et al (2012) Grey matter atrophy in cognitively impaired Parkinson’s disease. J Neurol Neurosurg Psychiatry 83(2):188–194. https://doi.org/10.1136/jnnp-2011-300828
Muller CM, de Vos RA, Maurage CA et al (2005) Staging of sporadic Parkinson disease-related alpha-synuclein pathology: inter- and intra-rater reliability. J Neuropathol Exp Neurol 64(7):623–628. https://doi.org/10.1097/01.jnen.0000171652.40083.15
Nelson PT, Abner EL, Patel E et al (2018) The Amygdala as a Locus of Pathologic Misfolding in Neurodegenerative Diseases. J Neuropathol Exp Neurol 77(1):2–20. https://doi.org/10.1093/jnen/nlx099
Parkkinen L, Pirttila T, Alafuzoff I (2008) Applicability of current staging/categorization of alpha-synuclein pathology and their clinical relevance. Acta Neuropathol 115(4):399–407. https://doi.org/10.1007/s00401-008-0346-6
Pessoa L (2010) Emotion and cognition and the amygdala: from “what is it?“ to “what’s to be done?“. Neuropsychologia 48(12):3416–3429. https://doi.org/10.1016/j.neuropsychologia.2010.06.038
Pigot K, Rick J, Xie SX et al (2015) Longitudinal study of normal cognition in Parkinson disease. Neurology 85(15):1276–1282. https://doi.org/10.1212/WNL.0000000000002001
Price J, Russchen FT, Amaral DG (1987) The Limbic Region II. In: Björklund HT (ed) Handbook of Chemical Neuroanatomy, 5 edn. Elsevier, pp 279–388
Price JL (1990) Olfactory System. In: Paxinos G (ed) The Human Nervous System. Academic Press, pp 979–998
Santangelo G, Vitale C, Picillo M et al (2015) Mild Cognitive Impairment in newly diagnosed Parkinson’s disease: A longitudinal prospective study. Parkinsonism Relat Disord 21(10):1219–1226. https://doi.org/10.1016/j.parkreldis.2015.08.024
Saygin ZM, Kliemann D, Iglesias JE et al (2017) High-resolution magnetic resonance imaging reveals nuclei of the human amygdala: manual segmentation to automatic atlas. NeuroImage 155:370–382. https://doi.org/10.1016/j.neuroimage.2017.04.046
Schapira AH, Tolosa E (2010) Molecular and clinical prodrome of Parkinson disease: implications for treatment. Nat Rev Neurol 6(6):309–317. https://doi.org/10.1038/nrneurol.2010.52
Silveira-Moriyama L, Holton JL, Kingsbury A et al (2009) Regional differences in the severity of Lewy body pathology across the olfactory cortex. Neurosci Lett 453(2):77–80. https://doi.org/10.1016/j.neulet.2009.02.006
Solano-Castiella E, Schafer A, Reimer E et al (2011) Parcellation of human amygdala in vivo using ultra high field structural MRI. NeuroImage 58(3):741–748. https://doi.org/10.1016/j.neuroimage.2011.06.047
Spillantini MG, Crowther RA, Jakes R et al (1998) alpha-Synuclein in filamentous inclusions of Lewy bodies from Parkinson’s disease and dementia with lewy bodies. Proc Natl Acad Sci U S A 95(11):6469–6473. https://doi.org/10.1073/pnas.95.11.6469
Uysal-Canturk P, Hanagasi HA, Bilgic B et al (2018) An assessment of Movement Disorder Society Task Force diagnostic criteria for mild cognitive impairment in Parkinson’s disease. Eur J Neurol 25(1):148–153. https://doi.org/10.1111/ene.13467
van Mierlo TJ, Chung C, Foncke EM et al (2015) Depressive symptoms in Parkinson’s disease are related to decreased hippocampus and amygdala volume. Mov Disord 30(2):245–252. https://doi.org/10.1002/mds.26112
Vriend C, Boedhoe PS, Rutten S et al (2016) A smaller amygdala is associated with anxiety in Parkinson’s disease: a combined FreeSurfer-VBM study. J Neurol Neurosurg Psychiatry 87(5):493–500. https://doi.org/10.1136/jnnp-2015-310383
Weintraub D, Doshi J, Koka D et al (2011) Neurodegeneration across stages of cognitive decline in Parkinson disease. Arch Neurol 68(12):1562–1568. https://doi.org/10.1001/archneurol.2011.725
Williams-Gray CH, Foltynie T, Brayne CE et al (2007) Evolution of cognitive dysfunction in an incident Parkinson’s disease cohort. Brain 130(Pt 7:1787–1798. https://doi.org/10.1093/brain/awm111
Yilmazer-Hanke D (2012) Amygdala. In G. P. Jürgen K. Mai (Ed.), The Human Nervous System (Third ed., pp. 759–834) Academic Press. https://doi.org/10.1016/B978-0-12-374236-0.10022-7
Acknowledgements
Ulaş Ay acknowledges the support of Turkish Council of Higher Education for 100/2000 CoHE doctoral scholarship. We would like to acknowledge funding support from the TUBITAK grant #115S219 and the Istanbul University Research Projects Unit project #1567/42362. We would also like to Dilek Betul Arslan, Zeynep Tufekcioglu, Basar Bilgic, and Hasmet Hanagasi for their contributions to data collection. Some of the data in this paper were previously presented at the annual meeting of the Turkish Neurological Society in Antalya, Turkey, in Nov 2019, and we won first prize for an oral presentation. Therefore, we are also immensely grateful to the Turkish Neurological Society.
Funding
This study was funded by the Scientific and Technological Research Council of Turkey (TUBITAK) grant #115S219 and the Istanbul University Research Projects Unit project #1567/42362. EE received a yearly doctoral stipend (57140539) from the DAAD (German Academic Exchange Service).
Author information
Authors and Affiliations
Contributions
Ulaş Ay: Conceptualization, Methodology, Software, Formal analysis, Visualization, Writing - Original Draft. Zerrin Yıldırım: Conceptualization, Methodology, Formal analysis, Writing - Original Draft. Emel Erdoğdu: Investigation, Resources, Data Curation. Ani Kıçik: Investigation, Resources, Data Curation. Esin Öztürk-Işık: Writing - Review & Editing, Supervision, Project administration, Funding acquisition. Tamer Demiralp: Methodology, Formal analysis, Writing - Review & Editing, Supervision, Funding acquisition. Hakan Gürvit: Methodology, Writing - Review & Editing, Supervision.
Corresponding author
Ethics declarations
Competing interests
The authors report no competing interests.
Ethics approval
Ethical approval was obtained from Istanbul Medical Faculty Clinical Research Ethics Committee of Istanbul University, Turkey
Participant consent
Participants were informed about the research, and their written consents were obtained before the research.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors Ulaş Ay and Zerrin Yıldırım contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ay, U., Yıldırım, Z., Erdogdu, E. et al. Shrinkage of olfactory amygdala connotes cognitive impairment in patients with Parkinson’s disease. Cogn Neurodyn 17, 1309–1320 (2023). https://doi.org/10.1007/s11571-022-09887-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11571-022-09887-y