Abstract
The genus Calogaya (Teloschistaceae, Xanthorioideae) was established to accommodate mainly epilithic lichens with lobate thalli, previously regarded as the “Caloplaca saxicola group.” Data supporting the recognition of this new genus came from European lichens, and although the genus is soundly based, we have found in Asia numerous epiphytic lineages and lineages with reduced, non-lobate thallus in dry continental areas. The taxonomic and functional diversity of Calogaya is distinctly higher in steppe and desert areas of Asia than in the less arid regions of Europe. We sampled 238 specimens, mostly from arid regions of north-western China, Iran, southern Siberia and Turkey. Three nuclear DNA loci were analysed separately and jointly by Bayesian inference, maximum likelihood and *BEAST approaches. Delimitations of 28 putative species were tested by BP&P multispecies coalescent model with joint analysis of species delimitation and species-tree estimation. Finally, we recognised 22 taxonomic units: 16 are at species rank, 3 are treated as subspecies and 3 are complexes, treated here as a single entity, but in reality probably including more than one species. Calogaya altynis, C. biatorina subsp. asiatica, C. decipiens subsp. esorediata, C. haloxylonis, C. orientalis, C. xanthoriella and C. xinjiangis are newly described. Caloplaca zoroasteriorum is combined into Calogaya, and Calogaya persica is reduced to a subspecies. The taxonomic status of Calogaya saxicola is unclear, and the name is employed here “sensu lato” for several non-monophyletic epilithic lineages with short-lobed thalli. Calogaya biatorina and C. ferrugineoides are the two other heterogeneous taxonomic units probably including more species.





Similar content being viewed by others
References
Aras S, Cansaran D (2006) Isolation of DNA for sequence analysis from herbarium material of some lichen specimens. Turk J Bot 30:449–453
Arcadia L (2009) Large genera, small genera and lichen taxonomy. Br Lichen Soc Bull 104:13–19
Arup U, Søchting U, Frödén P (2013) A new taxonomy of the family Teloschistaceae. Nord J Bot 31:16–83
Bässler C, Cadotte MW, Beudert B, Heibl C, Blaschke M, Bradtka JH, Langbehn T, Werth S, Müller J (2015) Contrasting patterns of lichen functional diversity and species richness across an elevation gradient. Ecography 38:1–10
Crespo F, Kauff PK, Divakar R, del Prado S, Pérez-Ortega S, de Paz GA, Ferencova Z, Blanco O, Roca-Valiente B, Núńez-Zapata J, Cubas P, Argüello A, Elix JA, Esslinger TL, Hawksworth DL, Millanes AM, Molina MC, Wedin M, Ahti T, Aptroot A, Barreno E, Bungartz F, Calvelo S, Candan M, Cole MJ, Ertz D, Goffinet B, Lindblom L, Lücking R, Lutzoni F, Mattsson JE, Messuti MI, Miadlikowska J, Piercey-Normore MD, Rico VJ, Sipman H, Schmitt I, Spribille T, Thell A, Thor G, Upreti DK, Lumbsch HT (2010) Phylogenetic generic classification of parmelioid lichens (Parmeliaceae, Ascomycota) based on molecular, morphological and chemical evidence. Taxon 59:1735–1753
Drummond AJ, Rambaut A (2007) BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol Biol 7:214
Ekman S (1996) The corticolous and lignicolous species of Bacidia and Bacidina in North America. Opera Bot 127:1–148
Frolov I, Vondrák J, Fernández-Mendoza F, Wilk K, Khodosovtsev A, Halıcı MG (2016) Three new, seemingly-cryptic species in the lichen genus Caloplaca (Teloschistaceae) distinguished in two-phase phenotype evaluation. Ann Bot Fenn 53:243–262
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes—application for the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Gaya E (2009) Taxonomical revision of the Caloplaca saxicola group (Teloschistaceae, lichen-forming Ascomycota). Bibl Lichenol 101:1–191
Gaya E, Redelings BD, Navarro-Rosinés P, Llimona X, De Cáceres M, Lutzoni F (2011) Align or not to align? Resolving species complexes within the Caloplaca saxicola group as a case study. Mycologia 103:361–378
Gaya E, Fernández-Brime S, Vargas R, Lachlan RF, Gueidan C, Ramírez-Mejía M, Lutzoni F (2015) The adaptive radiation of lichen-forming Teloschistaceae is associated with sunscreening pigments and a bark-to-rock substrate shift. PNAS 112:11600–11605
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Huelsenbeck JP, Ronquist F (2001) MrBayes: Bayesian inference of phylogeny. Bioinformatics 17:754–755
Johansson V, Ranius T, Snall T (2012) Epiphyte metapopulation dynamics are explained by species traits, connectivity, and patch dynamics. Ecology 93:235–241
Johansson V, Ranius T, Snall T (2013) Epiphyte metapopulation persistence after drastic habitat decline and low tree regeneration: time-lags and effects of conservation actions. J Appl Ecol 50:414–422
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780
Katoh K, Kuma K, Toh H, Miyata T (2005) MAFFT version 5: improvement in accuracy of multiple sequence alignment. Nucleic Acids Res 33:511–518
Kondratyuk SY, Lőkös L, Zarei-Darki B, Haji Moniri M, Tchabanenko S, Galanina I, Yakovchenko L, Hooshmand F, Ezhkin A, Hur J-S (2013) Five new Caloplaca species (Teloschistaceae, Ascomycota) from Asia. Acta Bot Hungar 55:41–60
Kondratyuk SY, Kärnefelt I, Thell A, Elix JA, Kim J, Jeong M-H, Yu N-N, Kondratiuk A, Hur J-S (2014a) A revised taxonomy for the subfamily Xanthorioideae (Teloschistaceae, Ascomycota) based on molecular phylogeny. Acta Bot Hungar 56:141–178
Kondratyuk SY, Jeong M-H, Yu N-N, Kärnefelt I, Thell A, Elix JA, Kim J, Kondratiuk AS, Hur J-S (2014b) A revised taxonomy for the subfamily Caloplacoideae (Teloschistaceae, Ascomycota) based on molecular phylogeny. Acta Bot Hungar 56:93–123
Kondratyuk SY, Kärnefelt I, Thell A, Elix JA, Kim J, Kondratiuk AS, Hur J-S (2015a) Brownlielloideae, a new subfamily in the Teloschistaceae (Lecanoromycetes, Ascomycotina). Acta Bot Hungar 57:321–343
Kondratyuk SY, Lőkös L, Kim JA, Kondratiuk AS, Jeong MH, Jang SH, Oh S-O, Hur J-S (2015b) Three new monotypic genera of the Caloplacoid lichens (Teloschistaceae, lichen-forming ascomycetes). Mycobiology 43:195–202
Lücking R (2008) Foliicolous lichenized Fungi. Flora Neotropica Monograph 103:1–866
Magnusson AH (1940) Lichens from Central Asia. Part I. In: Hedin S (ed) Reports Scientific Exped. Nort-west. provinces of China (the Sino-Swedish expedition). 13, XI. Botany, 1. Aktiebolaget Thule, Stockholm
Magnusson AH (1944) Lichens from Central Asia. Part II. In: Hedin S (ed) Reports Scientific Exped. Northwest. provinces of China (the Sino-Swedish Expedition). 22, XI, Botany, 2. Aktiebolaget Thule, Stockholm
Myllys L, Lohtander K, Tehler A (2001) β-tubulin, ITS and group I intron sequences challenge the species pair concept in Physcia aipolia and P. caesia. Mycologia 93:335–343
Nelson PR, McCune B, Roland C, Stehn S (2015) Non-parametric methods reveal non-linear functional trait variation of lichens along environmental and fire age gradients. J Veg Sci 26:848–865
Posada D (2008) jModelTest: phylogenetic model averaging. Mol Biol Evol 25:1253–1256
Rambaut A, Drummond AJ (2009) TreeAnnotator. Available online at http://beast.bio.ed.ac.uk/TreeAnnotator
Rambaut A, Suchard MA, Xie D, Drummond AJ (2014) Tracer v1.6. Available online at http://beast.bio.ed.ac.uk/Tracer
Rannala B, Yang Z (2003) Bayes estimation of species divergence times and ancestral population sizes using DNA sequences from multiple loci. Genetics 164:1645–1656
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixedmodels. Bioinformatics 19:1572–1574
Shahidin H, Abbas A, Wei J (2010) Caloplaca tianshanensis (lichen-forming Ascomycota), a new species of subgenus Pyrenodesmia from China. Mycotaxon 114:1–6
Simmons MP, Ochoterena H (2000) Gaps as characters in sequence-based phylogenetic analyses. Syst Biol 49:369–381
Smith CW, Aptroot A, Coppins BJ, Fletcher A, Gilbert OL, James PW, Wolseley PA, eds (2009) The Lichens of Great Britain and Ireland. British Lichen Society, London
Søchting U, Figueras G (2007) Caloplaca lenae species nova, and other Caloplaca species with caloploicin and vicanicin. Lichenologist 39:7–14
Søchting U, Garrido-Benavent I, Seppelt R, Castello M, Pérez-Ortega S, de los Ríos Murillo A, Frödén P, Arup U (2014a) Charcotiana and Amundsenia, two new genera in Teloschistaceae (lichenized Ascomycota, subfamily Xanthorioideae) hosting two new species from continental Antarctica, and Austroplaca frigida, a new name for a continental Antarctic species. Lichenologist 46:763–782
Søchting U, Sogaard MZ, Elix JA, Arup U, Elvebakk A, Sancho LG (2014b) Catenarina (Teloschistaceae, Ascomycota), a new southern hemisphere genus with 7-chlorocatenarin. Lichenologist 46:175–187
Sokal RR, Sneath PHA (1963) Principles of numerical taxonomy. W.H. Freeman, San Francisco
Stamatakis A, Ludwig T, Meier H (2005) RAxML-III: a fast program for maximum likelihood-based interference of large phylogenetic trees. Bioinformatics 21:456–463
Steiner M, Poelt J (1982) Caloplaca sect. Xanthoriella, sect. nov.: Untersuchungen über die “Xanthoria lobulata-Gruppe” (Lichenes, Teloschistaceae). Plant Syst Evol 140:151–177
Sukumaran J, Knowles LL (2017) Multispecies coalescent delimits structure, not species. PNAS 114:1607–1612
Tehler A, Ertz D, Irestedt M (2013) The genus Dirina (Roccellaceae, Arthoniales) revisited. Lichenologist 45:427–476
Veselská T, Svoboda J, Růžičková Z, Kolařík M (2014) Application of flow cytometry for genome size determination in Geosmithia fungi: a comparison of methods. Cytometry A 85:854–861
Vondrák J, Khodosovtsev A, Šoun J, Vondráková O (2012) Two new European species from the heterogeneous Caloplaca holocarpa group (Teloschistaceae). Lichenologist 44:73–89
Vondrák J, Frolov I, Arup U, Khodosovtsev A (2013a) Methods for phenotypic evaluation of crustose lichens with emphasis on Teloschistaceae. Chernomorsky Botanicheskii Zhurnal 9:382–405
Vondrák J, Frolov I, Říha P, Hrouzek P, Palice Z, Nadyeina O, Halıcı G, Khodosovtsev A, Roux C (2013b) New crustose Teloschistaceae in Central Europe. Lichenologist 45:701–722
Vondrák J, Frolov I, Davydov EA, Urbanavichene I, Chesnokov S, Zhdanov I, Muchnik E, Konoreva L, Himelbrant D, Tchabanenko S (2016) The extensive geographical range of several species of Teloschistaceae: evidence from Russia. Lichenologist 48:171–189
Yang Z (2015) The BPP program for species tree estimation and species delimitation. Curr Zool 61:854–865
Yang Z, Rannala B (2010) Bayesian species delimitation using multilocus sequence data. PNAS 107:9264–9269
Yang Z, Rannala B (2014) Unguided Species Delimitation Using DNA Sequence Data from Multiple Loci. Mol Biol Evol 31:3125–3135
Acknowledgments
Linda in Arcadia kindly revised the manuscript. We acknowledge generous support from the National natural science foundation of China (NFSC, projects no. 31093440 and no. 31260008) and a long-term research development grant RVO 67985939.
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: Gerhard Rambold
Electronic supplementary material
ESM 1
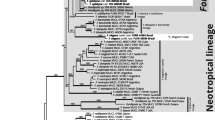
Concatenated ML tree based on analysis of alignment including all three loci (ITS, beta-tubulin, MS204). Branches with bootstrap support (BP > 0.7) have thick lines. Clades recognised at species level are displayed as cartoons (grey triangles or squares). Length of cartoons (horizontal dimension) reflects number of haplotypes within clades. Height of cartoons reflects sampling size. Numbers of samples per clade are in brackets following names of taxa. (JPG 644 kb)
ESM 3
(JPG 1923 kb)
ESM 4
(JPG 1845 kb)
ESM 5
Data for sequenced specimens (ID, locality, substrate & NCBI accession numbers). Substrate: bryo (muscicolous), calc (calcareous), sax (saxicolous), sili (siliceous). In some cases, mostly in MS204, two different sequences were generated (putative paralogs) from a single specimen. In such cases, both NCBI accession numbers are listed. (XLSX 31 kb)
Rights and permissions
About this article
Cite this article
Vondrák, J., Shahidin, H., Moniri, M.H. et al. Taxonomic and functional diversity in Calogaya (lichenised Ascomycota) in dry continental Asia. Mycol Progress 17, 897–916 (2018). https://doi.org/10.1007/s11557-018-1402-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-018-1402-9