Abstract
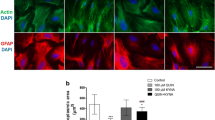
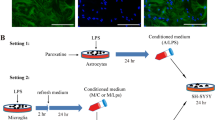
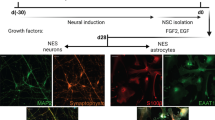
Brain glia produce neuroactive metabolites via tryptophan-kynurenine catabolism. A role for kynurenine pathway (KP) metabolites is proposed in reactive glial associated neurodegeneration. The aim of this investigation was to assess the role of KP induction and KP metabolites in driving reactive glial associated neuronal atrophy. Rat primary mixed glia, and enriched microglial and astroglial cultures were stimulated with IFNγ (10 ng/ml) for 24 hours. KP induction in mixed glial cells was confirmed by raised expression of the rate limiting KP enzyme indoleamine 2,3 dioxygenase (IDO) and raised concentrations of KP metabolites kynurenic acid (KYNA) and quinolinic acid (QUIN) in the conditioned media. Conditioned media was transferred onto immature (3 days) and mature (21 days) primary cortical neurons in vitro for 24 hours. IFNγ-stimulated mixed glial conditioned media reduced neurite outgrowth and complexity of both immature and mature neurons and co-localised expression of synaptic markers determined by immunocytochemistry. Pre-treatment of mixed glial cells with the IDO inhibitor, 1-methyltryptophan (1-MT) (L) prevented these effects of IFNγ-stimulated mixed glial conditioned media. KYNA increased complexity and synapse formation in mature cortical neurons and protected against reduced neuronal complexity and co-localised expression of synaptic markers induced by conditioned media from IFNγ-stimulated mixed glia and by treatment of neuronal cells with QUIN (1 µM). Overall, this study supports a role for the KP in driving neuronal atrophy associated with reactive glia and indicates that inhibition of the KP in glia, or raising the concentration of the astrocytic metabolite KYNA, protects against reactive microglial and QUIN-associated neuronal atrophy.
Graphical Abstract






Similar content being viewed by others
Data Availability
Data will be made available upon request.
References
Braidy N, Grant R, Brew BJ, Adams S, Jayasena T, Guillemin GJ (2009) Effects of Kynurenine Pathway Metabolites on Intracellular NAD Synthesis and Cell Death in Human Primary Astrocytes and Neurons. Int J Tryptophan Res. 2:61–9
Day JS, O’Neill E, Cawley C, Aretz NK, Kilroy D, Gibney SM, Harkin A, Connor TJ (2014) Noradrenaline acting on astrocytic beta(2)-adrenoceptors induces neurite outgrowth in primary cortical neurons. Neuropharmacology 77:234–248
DiNatale BC, Murray IA, Schroeder JC, Flaveny CA, Lahoti TS, Laurenzana EM, Omiecinski CJ, Perdew GH (2010) Kynurenic acid is a potent endogenous aryl hydrocarbon receptor ligand that synergistically induces interleukin-6 in the presence of inflammatory signaling. Toxicol Sci 115(1):89–97
Eroglu C, Barres BA (2010) Regulation of synaptic connectivity by glia. Nature 468(7321):223–231
Ferreira FS, Biasibetti-Brendler H, Pierozan P, Schmitz F, Bertó CG, Prezzi CA, Manfredini V, Wyse ATS (2018) Kynurenic acid restores Nrf2 levels and prevents quinolinic acid-induced toxicity in rat striatal slices. Mol Neurobiol 55(11):8538–8549
García-Lara L, Pérez-Severiano F, González-Esquivel D, Elizondo G, Segovia J (2015) Absence of aryl hydrocarbon receptors increases endogenous kynurenic acid levels and protects mouse brain against excitotoxic insult and oxidative stress. J Neurosci Res 93(9):1423–1433
Gutierrez H, Davies AM (2007) A fast and accurate procedure for deriving the Sholl profile in quantitative studies of neuronal morphology. J Neurosci Methods 163(1):24–30
Lob S, Konigsrainer A, Schafer R, Rammensee HG, Opelz G, Terness P (2008) Levo- but not dextro-1-methyl tryptophan abrogates the IDO activity of human dendritic cells. Blood. 111(4):2152–2154
Lugo-Huitron R, Ugalde Muniz P, Pineda B, Pedraza-Chaverri J, Rios C, Perez-de la Cruz V (2013) Quinolinic acid: an endogenous neurotoxin with multiple targets. Oxid Med Cell Longev 2013:104024
Majláth Z, Török N, Toldi J, Vécsei L (2016) Memantine and kynurenic acid: current neuropharmacological aspects. Curr Neuropharmacol 14(2):200–209
Midttun O, Hustad S, Ueland PM (2009) Quantitative profiling of biomarkers related to B-vitamin status, tryptophan metabolism and inflammation in human plasma by liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrom 23:1371–1379
Midttun O, Kvalheim G, Ueland PM (2013) High-throughput, low-volume, multianalyte quantification of plasma metabolites related to one-carbon metabolism using HPLC-MS/MS. Anal Bioanal Chem 405:2009–2017
Moroni F, Cozzi A, Sili M, Mannaioni G (2012) Kynurenic acid: a metabolite with multiple actions and multiple targets in brain and periphery. J Neural Transm (Vienna) 119(2):133–139
Nguyen NT, Kimura A, Nakahama T, Chinen I, Masuda K, Nohara K, Fujii-Kuriyama Y, Kishimoto T (2010) Aryl hydrocarbon receptor negatively regulates dendritic cell immunogenicity via a kynurenine-dependent mechanism. Proc Natl Acad Sci U S A 107(46):19961–19966
O’Farrell K, Harkin A (2017) Stress-related regulation of the kynurenine pathway: Relevance to neuropsychiatric and degenerative disorders. Neuropharmacol 112(Pt B):307–323
O’Farrell K, Fagan E, Connor TJ, Harkin A (2017) Inhibition of the kynurenine pathway protects against reactive microglial-associated reductions in the complexity of primary cortical neurons. Eur J Pharmacol 810:163–173
O’Neill E, Kwok B, Day JS, Connor TJ, Harkin A (2016) Amitriptyline protects against TNF-α-induced atrophy and reduction in synaptic markers via a Trk-dependent mechanism. Pharmacology Research Perspectives 4(2):e00195. doi:https://doi.org/10.1002/prp2.195
Pérez-De La Cruz V, Carrillo-Mora P, Santamaría A (2012) Quinolinic Acid, an endogenous molecule combining excitotoxicity, oxidative stress and other toxic mechanisms. Int J Tryptophan Res 5:1–8
Prescott C, Weeks AM, Staley KJ, Partin KM (2006) Kynurenic acid has a dual action on AMPA receptor responses. Neurosci Lett 402(1–2):108–112
Qian F, Villella J, Wallace PK, Mhawech-Fauceglia P, Tario Jr JD, Andrews C, Matsuzaki J, Valmori D, Ayyoub M, Frederick PJ, Beck A, Liao J, Cheney R, Moysich K, Lele S, Shrikant P, Old LJ, Odunsi K (2009) Efficacy of levo-1-methyl tryptophan and dextro-1-methyl tryptophan in reversing indoleamine-2,3-dioxygenase-mediated arrest of T-cell proliferation in human epithelial ovarian cancer. Cancer Res 69(13):5498–5504
Rahman A, Ting K, Cullen KM, Braidy N, Brew BJ, Guillemin GJ (2009) The excitotoxin quinolinic acid induces tau phosphorylation in human neurons. PLoS One 4(7):;e6344
Rahman A, Rao MS, Khan KM (2018) Intraventricular infusion of quinolinic acid impairs spatial learning and memory in young rats: a novel mechanism of lead-induced neurotoxicity. J Neuroinflammation 15(1):263
Schwarcz R, Bruno JP, Muchowski PJ, Wu HQ (2012) Kynurenines in the mammalian brain: when physiology meets pathology. Nat Rev Neurosci 13(7):465–477
Sholl DA (1953) Dendritic organization in the neurons of the visual and motor cortices of the cat. J Anat 87(4):387–406
Stroissnigg H, Trancíková A, Descovich L, Fuhrmann J, Kutschera W, Kostan J, Meixner A, Nothias F, Propst F (2007) S-nitrosylation of microtubule-associated protein 1B mediates nitric-oxide-induced axon retraction. Nat Cell Biol 9(9):1035–1045
Szalardy L, Zadori D, Toldi J, Fulop F, Klivenyi P, Vecsei L (2012) Manipulating kynurenic acid levels in the brain - on the edge between neuroprotection and cognitive dysfunction. Curr Top Med Chem 12(16):1797–1806
Vecsei L, Szalardy L, Fulop F, Toldi J (2013) Kynurenines in the CNS: recent advances and new questions. Nat Rev Drug Discov 12(1):64–82
Wilson C, González-Billault C (2015) Regulation of cytoskeletal dynamics by redox signaling and oxidative stress: implications for neuronal development and trafficking. Front Cell Neurosci 9:381
Funding
KO’F and KO’R were funded by a postgraduate award from Trinity College Dublin. The authors wish to acknowledge the support of the Trinity Foundation in memory of the late Professor Thomas J. Connor (1971–2013).
Author information
Authors and Affiliations
Contributions
All authors had access to the data in the study and have given final approval of the version to be published. K.O’R. and A.H. take responsibility for the integrity of the data and the accuracy of the data analysis. Conceptualization, A.H.; Methodology, A.H., K.O’R, K.O’F, J.D., O.M. Y.R.; Formal Analyses, A.H., K.O’R, K.O’F. Writing—Original Draft, A.H. and K.O’R; Writing—Review and Editing A.H., K.O’F., O.M., J.D.; Supervision, A.H.
Corresponding author
Ethics declarations
Conflicts of Interest
The authors have no conflicts of interest to declare.
Ethics Approval
The experimental protocols were in compliance with the European directive 2010/63/EU on the protection of animals used for scientific purposes, approved by the Animal Research Ethics Committee in Trinity College Dublin and performed under license granted by the Health Products Regulatory Authority.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Code Availability
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
O’Reilly, K., O’Farrell, K., Midttun, O. et al. Kynurenic Acid Protects Against Reactive Glial-associated Reductions in the Complexity of Primary Cortical Neurons. J Neuroimmune Pharmacol 16, 679–692 (2021). https://doi.org/10.1007/s11481-020-09976-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-020-09976-x