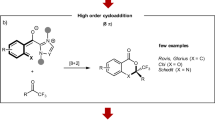
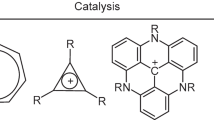
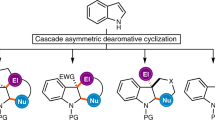
Abstract
N-heterocyclic carbene-catalyzed enantioselective kinetic resolutions, dynamic kinetic resolutions, and desymmetrization reactions are systematically reviewed. The content is organized according to the activation modes involved in these transformations. Future advances within this highly active research field are discussed from our perspectives on the topic.

Similar content being viewed by others
References
Enders D, Niemeier O, Henseler A. Chem Rev, 2007, 107: 5606–5655
Marion N, Díez-González S, Nolan S. Angew Chem Int Ed, 2007, 46: 2988–3000
Bugaut X, Glorius F. Chem Soc Rev, 2012, 41: 3511–3522
Ryan SJ, Candish L, Lupton DW. Chem Soc Rev, 2013, 42: 4906–4917
Mahatthananchai J, Bode JW. Acc Chem Res, 2014, 47: 696–707
Flanigan DM, Romanov-Michailidis F, White NA, Rovis T. Chem Rev, 2015, 115: 9307–9387
Murauski KJR, Jaworski AA, Scheidt KA. Chem Soc Rev, 2018, 47: 1773–1782
Kagan HB, Fiaud JC. Top Stereochem, 1988, 18: 249–330
Cook GR. Curr Org Chem, 2000, 4: 869–885
Pellissier H. Adv Synth Catal, 2011, 353: 1613–1666
Kitamura M, Tokunaga M, Noyori R. J Am Chem Soc, 1993, 115: 144–152
Pellissier H. Adv Synth Catal, 2011, 353: 659–676
Verho O, Bäckvall JE. J Am Chem Soc, 2015, 137: 3996–4009
Wencel-Delord J, Colobert F. Synthesis, 2016, 48: 2981–2996
Zeng XP, Cao ZY, Wang YH, Zhou F, Zhou J. Chem Rev, 2016, 116: 7330–7396
Borissov A, Davies TQ, Ellis SR, Fleming TA, Richardson MSW, Dixon DJ. Chem Soc Rev, 2016, 45: 5474–5540
Lu S, Poh S, Siau WY, Zhao Y. Synlett, 2013, 24: 1165–1169
Yang S, Fang X. Curr Org Synth, 2017, 14: 654–664
Chen S, Shi YH, Wang M. Chem Asian J, 2018, 13: 2184–2194
Wang Z, Pan D, Li T, Jin Z. Chem Asian J, 2018, 13: 2149–2163
De Risi C, Bortolini O, Di Carmine G, Ragno D, Massi A. Synthesis, 2019, 51: 1871–1891
Wu JY, Luo ZB, Dai LX, Hou XL. J Org Chem, 2008, 73: 9137–9139
Jin Z, Xu J, Yang S, Song BA, Chi YR. Angew Chem Int Ed, 2013, 52: 12354–12358
Fier PS, Maloney KM. J Am Chem Soc, 2019, 141: 1441–1445
Fier PS, Kim S, Maloney KM. J Am Chem Soc, 2019, 141: 18416–18420
Olbe L, Carlsson E, Lindberg P. Nat Rev Drug Discov, 2003, 2: 132–139
Sipos G, Drinkel EE, Dorta R. Chem Soc Rev, 2015, 44: 3834–3860
Otocka S, Kwiatkowska M, Madalińska L, Kiełba-siński P. Chem Rev, 2017, 117: 4147–4181
Chen J, Yuan P, Wang L, Huang Y. J Am Chem Soc, 2017, 139: 7045–7051
Kolb S, Oliver GA, Werz DB. Angew Chem Int Ed, 2020, 59: 22306–22310
Fischer C, Smith SW, Powell DA, Fu GC. J Am Chem Soc, 2006, 128: 1472–1473
Moss GP. Pure Appl Chem, 1996, 68: 2193–2222
Suzuki Y, Yamauchi K, Muramatsu K, Sato M. Chem Commun, 2004, 2770
Csihony S, Culkin DA, Sentman AC, Dove AP, Waymouth RM, Hedrick JL. J Am Chem Soc, 2005, 127: 9079–9084
Phillips EM, Riedrich M, Scheidt KA. J Am Chem Soc, 2010, 132: 13179–13181
Neufeld K, Henßen B, Pietruszka J. Angew Chem Int Ed, 2014, 53: 13253–13257
Kano T, Sasaki K, Maruoka K. Org Lett, 2005, 7: 1347–1349
Zeitler K. Org Lett, 2006, 8: 637–640
De Sarkar S, Biswas A, Song C, Studer A. Synthesis, 2011, 12: 1974–1983
Enders D, Niemeier O, Balensiefer T. Angew Chem Int Ed, 2006, 45: 1463–1467
De Sarkar S, Grimme S, Studer A. J Am Chem Soc, 2010, 132: 1190–1191
Iwahana S, Iida H, Yashima E. Chem Eur J, 2011, 17: 8009–8013
Kerr MS, Rovis T. J Am Chem Soc, 2004, 126: 8876–8877
Kuwano S, Harada S, Kang B, Oriez R, Yamaoka Y, Takasu K, Yamada K. J Am Chem Soc, 2013, 135: 11485–11488
Reynolds NT, Read de Alaniz J, Rovis T. J Am Chem Soc, 2004, 126: 9518–9519
Wadamoto M, Phillips EM, Reynolds TE, Scheidt KA. J Am Chem Soc, 2007, 129: 10098–10099
Liu B, Yan J, Huang R, Wang W, Jin Z, Zanoni G, Zheng P, Yang S, Chi YR. Org Lett, 2018, 20: 3447–3450
Zhao C, Li F, Wang J. Angew Chem Int Ed, 2016, 55: 1820–1824
Porey A, Mondal BD, Guin J. Angew Chem Int Ed, 2021, 60: 8786–8791
Wang Y, Yamauchi A, Hashimoto K, Fujiwara T, Inokuma T, Mitani Y, Ute K, Kuwano S, Yamaoka Y, Takasu K, Yamada K. ACS Catal, 2022, 12: 6100–6107
Bie J, Lang M, Wang J. Org Lett, 2018, 20: 5866–5871
Lu S, Poh SB, Siau WY, Zhao Y. Angew Chem Int Ed, 2013, 52: 1731–1734
Lu S, Poh SB, Zhao Y. Angew Chem Int Ed, 2014, 53: 11041–11045
Phillips E, Wadamoto M, Chan A, Scheidt K. Angew Chem Int Ed, 2007, 46: 3107–3110
Li GQ, Li Y, Dai LX, You SL. Adv Synth Catal, 2008, 350: 1258–1262
Binanzer M, Hsieh SY, Bode JW. J Am Chem Soc, 2011, 133: 19698–19701
Breslow R. J Am Chem Soc, 1958, 80: 3719–3726
Kreituss I, Bode JW. Acc Chem Res, 2016, 49: 2807–2821
Dong S, Frings M, Cheng H, Wen J, Zhang D, Raabe G, Bolm C. J Am Chem Soc, 2016, 138: 2166–2169
Enders D, Breuer K, Teles JH. Helv Chim Acta, 1996, 79: 1217–1221
Sheehan JC, Hunneman DH. J Am Chem Soc, 1966, 88: 3666–3667
Izquierdo J, Hutson GE, Cohen DT, Scheidt KA. Angew Chem Int Ed, 2012, 51: 11686–11698
Denmark SE, Wilson TW. Angew Chem Int Ed, 2012, 51: 9980–9992
Klán P, Šolomek T, Bochet CG, Blanc A, Givens R, Rubina M, Popik V, Kostikov A, Wirz J. Chem Rev, 2013, 113: 119–191
Xu W, Li Y, Liu R, Yang S, Liu J, Fang X. Org Chem Front, 2019, 6: 290–298
Zehra ST, Zhang G, Yang S, Fang X. Org Biomol Chem, 2019, 17: 2169–2173
Vora Hu, Rovis T. J Am Chem Soc, 2010, 132: 2860–2861
Zhang C, Hooper JF, Lupton DW. ACS Catal, 2017, 7: 2583–2596
Ghosh A, Biju AT. Angew Chem Int Ed, 2021, 60: 13712–13724
Maki BE, Chan A, Phillips EM, Scheidt KA. Org Lett, 2007, 9: 371–374
Mo J, Shen L, Chi YR. Angew Chem Int Ed, 2013, 52: 8588–8591
Sun FG, Sun LH, Ye S. Adv Synth Catal, 2011, 353: 3134–3138
Ryan SJ, Candish L, Lupton DW. J Am Chem Soc, 2009, 131: 14176–14177
Cheng J, Huang Z, Chi YR. Angew Chem Int Ed, 2013, 52: 8592–8596
Chen XY, Gao ZH, Song CY, Zhang CL, Wang ZX, Ye S. Angew Chem Int Ed, 2014, 53: 11611–11615
Zhu ZQ, Zheng XL, Jiang NF, Wan X, Xiao JC. Chem Commun, 2011, 47: 8670–8672
Liu B, Wang W, Huang R, Yan J, Wu J, Xue W, Yang S, Jin Z, Chi YR. Org Lett, 2018, 20: 260–263
Chen XY, Li S, Liu Q, Kumar M, Peuronen A, Rissanen K, Enders D. Chem Eur J, 2018, 24: 9735–9738
Liu J, Vasamsetty L, Anwar M, Yang S, Xu W, Liu J, Nagaraju S, Fang X. ACS Catal, 2020, 10: 2882–2893
Raup DEA, Cardinal-David B, Holte D, Scheidt KA. Nat Chem, 2010, 2: 766–771
Mo J, Chen X, Chi YR. J Am Chem Soc, 2012, 134: 8810–8813
Cohen DT, Scheidt KA. Chem Sci, 2012, 3: 53–57
De Sarkar S, Studer A. Angew Chem Int Ed, 2010, 49: 9266–9269
Axelsson A, Westerlund M, Zacharias SC, Runemark A, Haukka M, Sundén H. Eur J Org Chem, 2021, 2021: 3657–3661
He M, Struble JR, Bode JW. J Am Chem Soc, 2006, 128: 8418–8420
Barik S, Das RC, Balanna K, Biju AT. Org Lett, 2022, 24: 5456–5461
Nair V, Vellalath S, Babu BP. Chem Soc Rev, 2008, 37: 2691–2698
Menon RS, Biju AT, Nair V. Chem Soc Rev, 2015, 44: 5040–5052
Mondal S, Ghosh A, Biju AT. Chem Rec, 2022, 22: 202200054
Shao PL, Chen XY, Ye S. Angew Chem Int Ed, 2010, 49: 8412–8416
Zhang YR, He L, Wu X, Shao PL, Ye S. Org Lett, 2008, 10: 277–280
Huang XL, He L, Shao PL, Ye S. Angew Chem Int Ed, 2009, 48: 192–195
Douglas JJ, Churchill G, Slawin AMZ, Fox DJ, Smith AD. Chem Eur J, 2015, 21: 16354–16358
Jian TY, He L, Tang C, Ye S. Angew Chem Int Ed, 2011, 50: 9104–9107
Wang M, Huang Z, Xu J, Chi YR. J Am Chem Soc, 2014, 136: 1214–1217
Burstein C, Glorius F. Angew Chem Int Ed, 2004, 43: 6205–6208
Sohn SS, Rosen EL, Bode JW. J Am Chem Soc, 2004, 126: 14370–14371
Seebach D. Angew Chem Int Ed Engl, 1979, 18: 239–258
Bhattacharya A, mani Shukla P, Kumar Kaushik L, Maji B. Org Chem Front, 2019, 6: 3523–3529
Weygand F, Steglich W, Barocio de la Lama X. Tetrahedron, 1966, 22: 9–13
Noyori R, Ikeda T, Ohkuma T, Widhalm M, Kitamura M, Takaya H, Akutagawa S, Sayo N, Saito T, Taketomi T, Kumobayashi H. J Am Chem Soc, 1989, 111: 9134–9135
Turner NJ. Curr Opin Biotechnol, 2003, 14: 401–406
Schnell B, Faber K, Kroutil W. Adv Synthesis Catal, 2003, 345: 653–666
Alexeeva M, Carr R, Turner NJ. Org Biomol Chem, 2003, 1: 4133–4137
Turner NJ. Curr Opin Chem Biol, 2004, 8: 114–119
Robinson DEJE, Bull SD. Tetrahedron-Asymmetry, 2003, 14: 1407–1446
Tian SK, Chen Y, Hang J, Tang L, McDaid P, Deng L. Acc Chem Res, 2004, 37: 621–631
Connon SJ. Chem Commun, 2008, 2499
Hao L, Du Y, Lv H, Chen X, Jiang H, Shao Y, Chi YR. Org Lett, 2012, 14: 2154–2157
Fu Z, Xu J, Zhu T, Leong WWY, Chi YR. Nat Chem, 2013, 5: 835–839
Chauhan P, Enders D. Angew Chem Int Ed, 2014, 53: 1485–1487
Chen X, Fang X, Chi YR. Chem Sci, 2013, 4: 2613–2618
Chen X, Fong JZM, Xu J, Mou C, Lu Y, Yang S, Song BA, Chi YR. J Am Chem Soc, 2016, 138: 7212–7215
Liu B, Song R, Xu J, Majhi PK, Yang X, Yang S, Jin Z, Chi YR. Org Lett, 2020, 22: 3335–3338
Liu Y, Majhi PK, Song R, Mou C, Hao L, Chai H, Jin Z, Chi YR. Angew Chem Int Ed, 2020, 59: 3859–3863
Gao YY, Zhang CL, Dai L, Han YF, Ye S. Org Lett, 2021, 23: 1361–1366
Bringmann G, Hartung T. Angew Chem Int Ed Engl, 1992, 31: 761–762
Yu C, Huang H, Li X, Zhang Y, Wang W. J Am Chem Soc, 2016, 138: 6956–6959
Mori K, Itakura T, Akiyama T. Angew Chem Int Ed, 2016, 55: 11642–11646
Zhang J, Wang J. Angew Chem Int Ed, 2018, 57: 465–469
Wang G, Shi Q, Hu W, Chen T, Guo Y, Hu Z, Gong M, Guo J, Wei D, Fu Z, Huang W. Nat Commun, 2020, 11: 946–955
Guo D, Peng Q, Zhang B, Wang J. Org Lett, 2021, 23: 7765–7770
Goodman CG, Johnson JS. J Am Chem Soc, 2014, 136: 14698–14701
Vasamsetty L, Kong X, Meng M, Yang S, Xu W, Reddy PS, Fang X. Chem Asian J, 2018, 13: 3838–3844
Kolb HC, Finn MG, Sharpless KB. Angew Chem Int Ed, 2001, 40: 2004–2021
Perveen S, Yang S, Meng M, Xu W, Zhang G, Fang X. Nat Commun, 2019, 2: 8–16
Scott L, Nakano Y, Zhang C, Lupton DW. Angew Chem Int Ed, 2018, 57: 10299–10303
Candish L, Forsyth CM, Lupton DW. Angew Chem Int Ed, 2013, 52: 9149–9152
Cohen DT, Eichman CC, Phillips EM, Zarefsky ER, Scheidt KA. Angew Chem Int Ed, 2012, 51: 7309–7313
Cardinal-David B, Raup DEA, Scheidt KA. J Am Chem Soc, 2010, 132: 5345–5347
Johnston RC, Cohen DT, Eichman CC, Scheidt KA, Ha-Yeon Cheong P. Chem Sci, 2014, 5: 1974–1982
Cohen DT, Johnston RC, Rosson NT, Cheong PHY, Scheidt KA. Chem Commun, 2015, 51: 2690–2693
Lee A, Younai A, Price CK, Izquierdo J, Mishra RK, Scheidt KA. J Am Chem Soc, 2014, 136: 10589–10592
Mondal S, Mukherjee S, Das TK, Gonnade R, Biju AT. ACS Catal, 2017, 7: 3995–3999
Goodman CG, Walker MM, Johnson JS. J Am Chem Soc, 2015, 137: 122–125
Wu Z, Li F, Wang J. Angew Chem Int Ed, 2015, 54: 1629–1633
Chen K, Gao Z, Ye S. Angew Chem Int Ed, 2019, 58: 1183–1187
Liu Y, Chen Q, Mou C, Pan L, Duan X, Chen X, Chen H, Zhao Y, Lu Y, Jin Z, Chi YR. Nat Commun, 2019, 10: 1675–1683
Denmark SE, Heemstra JR, Beutner GL. Angew Chem Int Ed, 2005, 44: 4682–4698
Trost BM, Brindle CS. Chem Soc Rev, 2010, 39: 1600–1632
James T, van Gemmeren M, List B. Chem Rev, 2015, 115: 9388–9409
Yamashita Y, Yasukawa T, Yoo WJ, Kitanosono T, Kobayashi S. Chem Soc Rev, 2018, 47: 4388–4480
List B, Lerner RA, Barbas CF. J Am Chem Soc, 2000, 122: 2395–2396
Enders D, Hüttl MRM, Grondal C, Raabe G. Nature, 2006, 441: 861–863
Oisaki K, Zhao D, Kanai M, Shibasaki M. J Am Chem Soc, 2006, 128: 7164–7165
Gheewala CD, Collins BE, Lambert TH. Science, 2016, 351: 961–965
Kotani S, Yoshiwara Y, Ogasawara M, Sugiura M, Nakajima M. Angew Chem Int Ed, 2018, 57: 15877–15881
Yang X, Majhi PK, Chai H, Liu B, Sun J, Liu T, Liu Y, Zhou L, Xu J, Liu J, Wang D, Zhao Y, Jin Z, Chi YR. Angew Chem Int Ed, 2021, 60: 159–165
Liu Y, Luo G, Yang X, Jiang S, Xue W, Chi YR, Jin Z. Angew Chem Int Ed, 2020, 59: 442–448
Lv Y, Luo G, Liu Q, Jin Z, Zhang X, Chi YR. Nat Commun, 2022, 13: 36–45
Lv J, Xu J, Pan X, Jin Z, Chi YR. Sci China Chem, 2021, 64: 985–990
Ogston AG. Nature, 1948, 162: 963
Cohen SG, Altschul L. Nature, 1959, 183: 1678
García-Urdiales E, Alfonso I, Gotor V. Chem Rev, 2011, 111: PR110–PR180
Willis MC. J Chem Soc Perkin Trans 1, 1999, 1765–1784
Dílegas MD, Gálvez JA, Etayo P, Badorrey R, López-Ram-de-Viu P. Chem Soc Rev, 2011, 40: 5564–5587
Li BS, Wang Y, Proctor RSJ, Jin Z, Chi YR. Chem Commun, 2016, 52: 8313–8316
Wu Z, Wang J. ACS Catal, 2017, 7: 7647–7652
Xu B, Zhu SF, Xie XL, Shen JJ, Zhou QL. Angew Chem Int Ed, 2011, 50: 11483–11486
Lu S, Song X, Poh SB, Yang H, Wong MW, Zhao Y. Chem Eur J, 2017, 23: 2275–2281
Li S, Liu B, Chen L, Li X, Cheng JP. Org Chem Front, 2018, 5: 1101–1107
Huang Z, Huang X, Li B, Mou C, Yang S, Song BA, Chi YR. J Am Chem Soc, 2016, 138: 7524–7527
Yang G, Guo D, Meng D, Wang J. Nat Commun, 2019, 10: 3062–3068
Lu S, Poh SB, Rong ZQ, Zhao Y. Org Lett, 2019, 21: 6169–6172
Di Carmine G, Ragno D, Brandolese A, Bortolini O, Pecorari D, Sabuzi F, Mazzanti A, Massi A. Chem Eur J, 2019, 25: 7469–7474
DiRocco DA, Rovis T. Angew Chem Int Ed, 2012, 51: 5904–5906
Wu Y, Li M, Sun J, Zheng G, Zhang Q. Angew Chem Int Ed, 2022, 61: e202117340
Zhao W, Liu J, He X, Jiang H, Lu L, Xiao W. Chin J Org Chem, 2022, 42: 2504–2514
Lv X, Xu J, Sun C, Su F, Cai Y, Jin Z, Chi YR. ACS Catal, 2022, 12: 2706–2713
Mu D, Yuan W, Chen S, Wang N, Yang B, You L, Zu B, Yu P, He C. J Am Chem Soc, 2020, 142: 13459–13468
Zhou M, Liu J, Deng R, Wang Q, Wu S, Zheng P, Chi YR. ACS Catal, 2022, 12: 7781–7788
Liu J, Zhou M, Deng R, Zheng P, Chi YR. Nat Commun, 2022, 13: 4793–4801
Hu Z, Wei C, Shi Q, Hong X, Liu J, Zhou X, Han J, Cao W, Gupta AK, Zhang X, Wei D, Fu Z, Huang W. Nat Commun, 2022, 13: 4042–4051
Ohgo Y, Natori Y, Takeuchi S, Yoshimura J. Chem Lett, 1974, 3: 1327–1330
Kerr MS, Read de Alaniz J, Rovis T. J Am Chem Soc, 2002, 124: 10298–10299
Wöhler F, Liebig J. Ann Pharm, 1832, 3: 249–282
Stetter H. Angew Chem Int Ed, 1976, 15: 639–647
Liu Q, Rovis T. J Am Chem Soc, 2006, 128: 2552–2553
Li Y, Feng Z, You SL. Chem Commun, 2008, 2263
Jia MQ, You SL. Chem Commun, 2012, 48: 6363–6365
Jia MQ, Liu C, You SL. J Org Chem, 2012, 77: 10996–11001
Jia MQ, You SL. Synlett, 2013, 24: 1201–1204
Ema T, Akihara K, Obayashi R, Sakai T. Adv Synth Catal, 2012, 354: 3283–3290
Li Y, Yang S, Wen G, Lin Q, Zhang G, Qiu L, Zhang X, Du G, Fang X. J Org Chem, 2016, 81: 2763–2769
Lin Q, Li Y, Das DK, Zhang G, Zhao Z, Yang S, Fang X. Chem Commun, 2016, 52: 6459–6462
Liu H, He P, Liao X, Zhou Y, Chen X, Ou W, Wu Z, Luo C, Yang L, Xu J. ACS Catal, 2022, 12: 9864–9871
Zhuo S, Zhu T, Zhou L, Mou C, Chai H, Lu Y, Pan L, Jin Z, Chi YR. Angew Chem Int Ed, 2019, 58: 1784–1788
Mattson AE, Zuhl AM, Reynolds TE, Scheidt KA. J Am Chem Soc, 2006, 128: 4932–4933
Sun FG, Ye S. Synlett, 2011, 2011(7): 1005–1009
Barik S, Shee S, Das S, Gonnade RG, Jindal G, Mukherjee S, Biju AT. Angew Chem Int Ed, 2021, 60: 12264–12268
Shu T, Li S, Chen XY, Liu Q, von Essen C, Rissanen K, Enders D. Chem Commun, 2018, 54: 7661–7664
Knappke CEI, Imami A, Jacobi von Wangelin A. ChemCatChem, 2012, 4: 937–941
De Sarkar S, Biswas A, Samanta RC, Studer A. Chem Eur J, 2013, 19: 4664–4678
Zhu T, Liu Y, Smetankova M, Zhuo S, Mou C, Chai H, Jin Z, Chi YR. Angew Chem Int Ed, 2019, 58: 15778–15782
Hu JM, Zhang JQ, Sun BB, Chen JB, Yu JQ, Yang XP, Lv HP, Wang Z, Wang XW. Org Lett, 2019, 21: 8582–8586
Shee S, Mukherjee S, Gonnade RG, Biju AT. Org Lett, 2020, 22: 5407–5411
Wang G, Zhang M, Guan Y, Zhang Y, Hong X, Wei C, Zheng P, Wei D, Fu Z, Chi YR, Huang W. Research, 2021, 2021: 2021
Jin J, Huang X, Xu J, Li T, Peng X, Zhu X, Zhang J, Jin Z, Chi YR. Org Lett, 2021, 23: 3991–3996
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21961006, 32172459, 22371057), Science and Technology Department of Guizhou Province (Qiankehejichu-ZK [2021]Key033), Program of Introducing Talents of Discipline to Universities of China (111 Program, D20023) at Guizhou University, Frontiers Science Center for Asymmetric Synthesis and Medicinal Molecules, Department of Education, Guizhou Province (Qianjiaohe KY (2020)004), and Guizhou University (China).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, Y., Cai, H., Gan, X. et al. N-Heterocyclic carbene-catalyzed enantioselective (dynamic) kinetic resolutions and desymmetrizations. Sci. China Chem. 67, 482–511 (2024). https://doi.org/10.1007/s11426-022-1657-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-022-1657-0