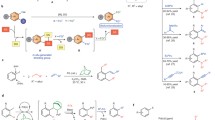
Abstract
A novel palladium-catalyzed oxidative cyclopropanation of enamides and norbornenes has been developed. The reaction proceeded through palladium-catalyzed vinyl C–H bond activation of enamides followed by two migratory insertions, β-(N)H elimination and hydrolyzation cascade steps. The reaction tolerates a range of functional groups and provides an effective method for the synthesis of cyclopropane-fused norbornanes in good yields under mild conditions.
Similar content being viewed by others
References
de Meijere A. Small Ring Compounds in Organic Synthsis VI. Berlin: Springer, 2000
Lebel H, Marcoux JF, Molinaro C, Charette AB. Chem Rev, 2003, 103: 977–1050
Chen DYK, Pouwer RH, Richard JA. Chem Soc Rev, 2012, 41: 4631–4642
Shi M, Shao LX, Lu JM, Wei Y, Mizuno K, Maeda H. Chem Rev, 2010, 110: 5883–5913
Schneider TF, Kaschel J, Werz DB. Angew Chem Int Ed, 2014, 53: 5504–5523
Nambu H, Fukumoto M, Hirota W, Yakura T. Org Lett, 2014, 16: 4012–4015
Manna S, Antonchick AP. Angew Chem Int Ed, 2016, 55: 5290–5293
Manna S, Antonchick AP. Angew Chem Int Ed, 2015, 54: 14845–14848
Trost BM, Ryan MC, Rao M, Markovic TZ. J Am Chem Soc, 2014, 136: 17422–17425
Toh KK, Biswas A, Wang YF, Tan YY, Chiba S. J Am Chem Soc, 2014, 136: 6011–6020
Charette AB. Cyclopropanation mediated by zinc organometallics. In: The Chemistry of Organozinc Compounds. Rappoport Z, Marek I, Eds. Chichester: John Wiley & Sons Ltd, 2006. 237–286
Ramazanov IR, Kadikova RN, Zosim TP, Nadrshina ZI, Dzhemilev UM. Mendeleev Commun, 2016, 26: 434–436
Chanthamath S, Iwasa S. Acc Chem Res, 2016, 49: 2080–2090
Morandi B, Carreira EM. Science, 2012, 335: 1471–1474
Xu H, Li YP, Cai Y, Wang GP, Zhu SF, Zhou QL. J Am Chem Soc, 2017, 139: 7697–7700
Feng LW, Wang P, Wang L, Tang Y. Sci Bull, 2015, 60: 210–215
Xie H, Zu L, Li H, Wang J, Wang W. J Am Chem Soc, 2007, 129: 10886–10894
Kunz RK, MacMillan DWC. J Am Chem Soc, 2005, 127: 3240–3241
White PB, Jaworski JN, Zhu GH, Stahl SS. ACS Catal, 2016, 6: 3340–3348
Qi X, Chen P, Liu G. Sci China Chem, 2015, 58: 1249–1251
Shang X, Liu ZQ. Chem Soc Rev, 2013, 42: 3253–3260
Xu YH, Chok YK, Loh TP. Chem Sci, 2011, 2: 1822–1825
Miura T, Sasaki T, Harumashi T, Murakami M. J Am Chem Soc, 2006, 128: 2516–2517
Tong X, Beller M, Tse MK. J Am Chem Soc, 2007, 129: 4906–4907
Piou T, Rovis T. J Am Chem Soc, 2014, 136: 11292–11295
Piou T, Rovis T. Nature, 2015, 527: 86–90
Guan ZH, Zhang ZY, Ren ZH, Wang YY, Zhang X. J Org Chem, 2011, 76: 339–341
Wang H, Cheng Y, Yu S. Sci China Chem, 2016, 59: 195–198
Xie JH, Zhu SF, Zhou QL. Chem Rev, 2011, 111: 1713–1760
Lei CH, Zhao L, Wang DX, Zhu J, Wang MX. Org Chem Front, 2014, 1: 909–913
Matsubara R, Kobayashi S. Acc Chem Res, 2008, 41: 292–301
He L, Zhao L, Wang DX, Wang MX. Org Lett, 2014, 16: 5972–5975
Wang M, Zhang X, Zhuang YX, Xu YH, Loh TP. J Am Chem Soc, 2015, 137: 1341–1347
Lei CH, Wang DX, Zhao L, Zhu J, Wang MX. J Am Chem Soc, 2013, 135: 4708–4711
Tong S, Wang DX, Zhao L, Zhu J, Wang MX. Angew Chem Int Ed, 2012, 51: 4417–4420
Wang K, Hu F, Zhang Y, Wang J. Sci China Chem, 2015, 58: 1252–1265
Chen M, Ren ZH, Wang YY, Guan ZH. Angew Chem Int Ed, 2013, 52: 14196–14199
Zhao MN, Yu L, Hui RR, Ren ZH, Wang YY, Guan ZH. ACS Catal, 2016, 6: 3473–3477
Zhao MN, Ren ZH, Wang YY, Guan ZH. Org Lett, 2014, 16: 608–611
Zhao MN, Ren ZH, Wang YY, Guan ZH. Chem Eur J, 2014, 20: 1839–1842
Zhao MN, Ren ZH, Wang YY, Guan ZH. Chem Commun, 2012, 48: 8105–8107
Zhao MN, Zhang MN, Ren ZH, Wang YY, Guan ZH. Sci Bull, 2017, 62: 493–496
Chen M, Zhang W, Ren ZH, Gao WY, Wang YY, Guan ZH. Sci China Chem, 2017, 60: 761–768
Du W, Gu Q, Li Z, Yang D. J Am Chem Soc, 2015, 137: 1130–1135
Guan ZH, Chen M, Ren ZH. J Am Chem Soc, 2012, 134: 17490–17493
Mao J, Xie H, Bao W. Org Lett, 2015, 17: 3678–3681
Khanna A, Premachandra IDUA, Sung PD, Van Vranken DL. Org Lett, 2013, 15: 3158–3161
Xia Y, Wang J. Chem Soc Rev, 2017, 46: 2306–2362
Yang Y, Li J, Du B, Yuan C, Liu B, Qin S. Chem Commun, 2015, 51: 6179–6182
Hu N, Zhao G, Zhang Y, Liu X, Li G, Tang W. J Am Chem Soc, 2015, 137: 6746–6749
Acknowledgments
This work was supported by the National Natural Science Foundation of China (NSFC-21622203, 21472147, 21702161), the China Postdoctoral Science Foundation Funded Project (2017M610644), and Fund of Northwest University (334100036).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Chen, M., Zhao, MN., Zhang, YD. et al. Palladium-catalyzed oxidative cyclopropanation of enamides and norbornenes initiated by C–H activation. Sci. China Chem. 61, 695–701 (2018). https://doi.org/10.1007/s11426-017-9150-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-017-9150-x