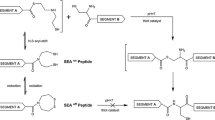
Abstract
Chemical synthesis of peptides and proteins has evolved into an indispensable tool for chemical biology. Peptide ligation is a straightforward technique for joining two short peptide fragments together via a native peptide bond to afford a larger natural peptide or protein. However, the junction sites are limited to several specific amino acids because most peptide ligations involve participation of the side-chain functional groups of the junction-site amino acids. To overcome such intrinsic limitations, “general” peptide ligations which do not rely on the side-chain functional group have been developed. This review summarized the recent developments in peptide ligations that are independent of side-chain functional group of ligation-junction-site amino acid.
Similar content being viewed by others
References
Hackenberger CPR, Schwarzer D. Angew Chem Int Ed, 2008, 47: 10030–10074
Pattabiraman VR, Bode JW. Nature, 2011, 480: 471–479
Bondalapati S, Jbara M, Brik A. Nat Chem, 2016, 8: 407–418
Huang Y, Liu L. Sci China Chem, 2015, 58: 1779–1781
Kent SBH. Chem Soc Rev, 2009, 38: 338–351
Lee CL, Li X. Sci China Chem, 2016, 59: 1061–1064
Merrifield RB. J Am Chem Soc, 1963, 85: 2149–2154
Coltart DM. Tetrahedron, 2000, 56: 3449–3491
Guzman F, Barberis S, Illanes A. Electron J Biotechnol, 2007, 10: 279–314
Nguyen GKT, Wang S, Qiu Y, Hemu X, Lian Y, Tam JP. Nat Chem Biol, 2014, 10: 732–738
Schellenberger V, Jakubke HD. Angew Chem Int Ed, 1991, 30: 1437–1449
van’t Hof W, Hansenova Manaskova S, Veerman EC, Bolscher JG. Biol Chem, 2015, 396: 283–293
Dawson PE, Muir TW, Clark-Lewis I, Kent SBH. Science, 1994, 266: 776–779
Okamoto R, Mandal K, Sawaya MR, Kajihara Y, Yeates TO, Kent SBH. Angew Chem Int Ed, 2014, 53: 5194–5198
Wang P, Dong S, Shieh JH, Peguero E, Hendrickson R, Moore MAS, Danishefsky SJ. Science, 2013, 342: 1357–1360
Blanco-Canosa JB, Dawson PE. Angew Chem Int Ed, 2008, 47: 6851–6855
Blanco-Canosa JB, Nardone B, Albericio F, Dawson PE. J Am Chem Soc, 2015, 137: 7197–7209
Boll E, Drobecq H, Ollivier N, Blanpain A, Raibaut L, Desmet R, Vicogne J, Melnyk O. Nat Protoc, 2015, 10: 269–292
Hou W, Zhang X, Li F, Liu CF. Org Lett, 2011, 13: 386–389
Mende F, Seitz O. Angew Chem Int Ed, 2011, 50: 1232–1240
Sharma RK, Tam JP. Org Lett, 2011, 13: 5176–5179
Taichi M, Hemu X, Qiu Y, Tam JP. Org Lett, 2013, 15: 2620–2623
Wang JX, Fang GM, He Y, Qu DL, Yu M, Hong ZY, Liu L. Angew Chem Int Ed, 2015, 54: 2194–2198
Zheng JS, Chang HN, Wang FL, Liu L. J Am Chem Soc, 2011, 133: 11080–11083
Fang GM, Li YM, Shen F, Huang YC, Li JB, Lin Y, Cui HK, Liu L. Angew Chem Int Ed, 2011, 50: 7645–7649
Zheng JS, Tang S, Huang YC, Liu L. Acc Chem Res, 2013, 46: 2475–2484
Zheng JS, Tang S, Qi YK, Wang ZP, Liu L. Nat Protoc, 2013, 8: 2483–2495
Chen X, Tang S, Zheng JS, Zhao R, Wang ZP, Shao W, Chang HN, Cheng JY, Zhao H, Liu L, Qi H. Nat Commun, 2015, 6: 7220
Fang GM, Wang JX, Liu L. Angew Chem Int Ed, 2012, 51: 10347–10350
Wang Z, Xu W, Liu L, Zhu TF. Nat Chem, 2016, 8: 698–704
Zheng JS, He Y, Zuo C, Cai XY, Tang S, Wang ZA, Zhang LH, Tian CL, Liu L. J Am Chem Soc, 2016, 138: 3553–3561
Ajish Kumar KS, Haj-Yahya M, Olschewski D, Lashuel HA, Brik A. Angew Chem Int Ed, 2009, 48: 8090–8094
Cergol KM, Thompson RE, Malins LR, Turner P, Payne RJ. Org Lett, 2014, 16: 290–293
Crich D, Banerjee A. J Am Chem Soc, 2007, 129: 10064–10065
Haase C, Rohde H, Seitz O. Angew Chem Int Ed, 2008, 47: 6807–6810
Ma J, Zeng J, Wan Q. Protein ligation and total synthesis II. In: Liu L, Ed. Postligation-Desulfurization: A General Approach for Chemical Protein Synthesis. Switzerland: Springer International Publishing, 2014. 57–101
Malins LR, Cergol KM, Payne RJ. ChemBioChem, 2013, 14: 559–563
Rohde H, Seitz O. Biopolymers, 2010, 94: 551–559
Yang R, Pasunooti KK, Li F, Liu XW, Liu CF. J Am Chem Soc, 2009, 131: 13592–13593
Canne LE, Bark SJ, Kent SBH. J Am Chem Soc, 1996, 118: 5891–5896
Johnson T, Quibell M, Sheppard RC. J Peptide Sci, 1995, 1: 11–25
Marinzi C, Bark SJ, Offer J, Dawson PE. Bioorgan Med Chem, 2001, 9: 2323–2328
Botti P, Carrasco MR, Kent SBH. Tetrahedron Lett, 2001, 42: 1831–1833
Low DW, Hill MG, Carrasco MR, Kent SBH, Botti P. Proc Natl Acad Sci USA, 2001, 98: 6554–6559
Li J, Cui HK, Liu L. Tetrahedron Lett, 2010, 51: 1793–1796
Bello C, Wang S, Meng L, Moremen KW, Becker CFW. Angew Chem Int Ed, 2015, 54: 7711–7715
Li J, He Q, Liu Y, Liu S, Tang S, Li C, Sun D, Li X, Zhou M, Zhu P, Bi G, Zhou Z, Zheng JS, Tian C. ChemBioChem, 2017, 18: 176–180
Pan M, Gao S, Zheng Y, Tan X, Lan H, Tan X, Sun D, Lu L, Wang T, Zheng Q, Huang Y, Wang J, Liu L. J Am Chem Soc, 2016, 138: 7429–7435
Yang R, Bi X, Li F, Cao Y, Liu CF. Chem Commun, 2014, 50: 7971–7974
Offer J, Dawson PE. Org Lett, 2000, 2: 23–26
Kawakami T, Akaji K, Aimoto S. Org Lett, 2001, 3: 1403–1405
Offer J, Boddy CNC, Dawson PE. J Am Chem Soc, 2002, 124: 4642–4646
Klán P, Šolomek T, Bochet CG, Blanc A, Givens R, Rubina M, Popik V, Kostikov A, Wirz J. Chem Rev, 2013, 113: 119–191
Pillai VR. Synthesis, 1980, 1980: 1–26
Kawakami T, Aimoto S. Tetrahedron Lett, 2003, 44: 6059–6061
Nadler C, Nadler A, Hansen C, Diederichsen U. Eur J Org Chem, 2015, 2015: 3095–3102
Bruice TC, Benkovic SJ. J Am Chem Soc, 1963, 85: 1–8
Kemp DS. Biopolymers, 1981, 20: 1793–1804
Kemp DS, Carey RI. J Org Chem, 1993, 58: 2216–2222
Loibl SF, Harpaz Z, Seitz O. Angew Chem Int Ed, 2015, 54: 15055–15059
Brik A, Wong CH. Chem Eur J, 2007, 13: 5670–5675
Brik A, Yang YY, Ficht S, Wong CH. J Am Chem Soc, 2006, 128: 5626–5627
Bennett CS, Dean SM, Payne RJ, Ficht S, Brik A, Wong CH. J Am Chem Soc, 2008, 130: 11945–11952
Payne RJ, Ficht S, Tang S, Brik A, Yang YY, Case DA, Wong CH. J Am Chem Soc, 2007, 129: 13527–13536
Yang YY, Ficht S, Brik A, Wong CH. J Am Chem Soc, 2007, 129: 7690–7701
Kemp DS, Vellaccio F. J Org Chem, 1975, 40: 3003–3004
Kemp DS, Grattan JA, Reczek J. J Org Chem, 1975, 40: 3465–3466
Wu B, Chen J, Warren JD, Chen G, Hua Z, Danishefsky SJ. Angew Chem Int Ed, 2006, 45: 4116–4125
Chen G, Warren JD, Chen J, Wu B, Wan Q, Danishefsky SJ. J Am Chem Soc, 2006, 128: 7460–7462
Liu CF, Rao C, Tam JP. J Am Chem Soc, 1996, 118: 307–312
Liu CF, Tam JP. J Am Chem Soc, 1994, 116: 4149–4153
Liu CF, Tam JP. Proc Natl Acad Sci USA, 1994, 91: 6584–6588
Li X, Lam HY, Zhang Y, Chan CK. Org Lett, 2010, 12: 1724–1727
Jin K, Sam IH, Po KHL, Lin D, Ghazvini Zadeh EH, Chen S, Yuan Y, Li X. Nat Commun, 2016, 7: 12394
Lam HY, Zhang Y, Liu H, Xu J, Wong CTT, Xu C, Li X. J Am Chem Soc, 2013, 135: 6272–6279
Lee CL, Liu H, Wong CTT, Chow HY, Li X. J Am Chem Soc, 2016, 138: 10477–10484
Wong CTT, Lam HY, Song T, Chen G, Li X. Angew Chem Int Ed, 2013, 52: 10212–10215
Zhang Y, Xu C, Lam HY, Lee CL, Li X. Proc Natl Acad Sci USA, 2013, 110: 6657–6662
Liu CF, Zhao J. Method of synthesizing peptides, proteins and bioconjugates. US Patent, 9453044, 2013-02-06
Fouché M, Masse F, Roth HJ. Org Lett, 2015, 17: 4936–4939
Ishiwata A, Ichiyanagi T, Takatani M, Ito Y. Tetrahedron Lett, 2003, 44: 3187–3190
Tung CL, Wong CTT, Li X. Org Biomol Chem, 2015, 13: 6922–6926
Raj M, Wu H, Blosser SL, Vittoria MA, Arora PS. J Am Chem Soc, 2015, 137: 6932–6940
Kemp DS, Bernstein ZW, McNeil GN. J Org Chem, 1974, 39: 2831–2835
Kemp DS, Choong SLH, Pekaar J. J Org Chem, 1974, 39: 3841–3847
Blake J, Li CH. Proc Natl Acad Sci USA, 1981, 78: 4055–4058
Aimoto S. Biopolymers, 1999, 51: 247–265
Kawakami T, Yoshimura S, Aimoto S. Tetrahedron Lett, 1998, 39: 7901–7904
Teruya K, Kawakami T, Akaji K, Aimoto S. Tetrahedron Lett, 2002, 43: 1487–1490
Chen G, Wan Q, Tan Z, Kan C, Hua Z, Ranganathan K, Danishefsky SJ. Angew Chem Int Ed, 2007, 46: 7383–7387
Payne RJ, Ficht S, Greenberg WA, Wong CH. Angew Chem Int Ed, 2008, 47: 4411–4415
Crich D, Sharma I. Angew Chem Int Ed, 2009, 48: 2355–2358
Wang P, Danishefsky SJ. J Am Chem Soc, 2010, 132: 17045–17051
Staudinger H, Meyer J. Helv Chim Acta, 1919, 2: 635–646
Köhn M, Breinbauer R. Angew Chem Int Ed, 2004, 43: 3106–3116
Saxon E, Armstrong JI, Bertozzi CR. Org Lett, 2000, 2: 2141–2143
Lin FL, Hoyt HM, van Halbeek H, Bergman RG, Bertozzi CR. J Am Chem Soc, 2005, 127: 2686–2695
Soellner MB, Nilsson BL, Raines RT. J Am Chem Soc, 2006, 128: 8820–8828
Nilsson BL, Kiessling LL, Raines RT. Org Lett, 2000, 2: 1939–1941
Nilsson BL, Kiessling LL, Raines RT. Org Lett, 2001, 3: 9–12
Soellner MB, Nilsson BL, Raines RT. J Org Chem, 2002, 67: 4993–4996
David O, Meester WJN, Bieräugel H, Schoemaker HE, Hiemstra H, van Maarseveen JH. Angew Chem Int Ed, 2003, 42: 4373–4375
Köhn M, Wacker R, Peters C, Schröder H, Soulère L, Breinbauer R, Niemeyer CM, Waldmann H. Angew Chem Int Ed, 2003, 42: 5830–5834
Soellner MB, Dickson KA, Nilsson BL, Raines RT. J Am Chem Soc, 2003, 125: 11790–11791
Nilsson BL, Hondal RJ, Soellner MB, Raines RT. J Am Chem Soc, 2003, 125: 5268–5269
Bode JW, Fox RM, Baucom KD. Angew Chem Int Ed, 2006, 45: 1248–1252
Bode JW, Sohn SS. J Am Chem Soc, 2007, 129: 13798–13799
Chiang PC, Kim Y, Bode JW. Chem Commun (Camb), 2009: 4566–4568
Dumas AM, Molander GA, Bode JW. Angew Chem Int Ed, 2012, 51: 5683–5686
Ju L, Lippert AR, Bode JW. J Am Chem Soc, 2008, 130: 4253–4255
Fukuzumi T, Bode JW. J Am Chem Soc, 2009, 131: 3864–3865
Pusterla I, Bode JW. Angew Chem Int Ed, 2012, 51: 513–516
Carrillo N, Davalos EA, Russak JA, Bode JW. J Am Chem Soc, 2006, 128: 1452–1453
Pattabiraman VR, Ogunkoya AO, Bode JW. Angew Chem Int Ed, 2012, 51: 5114–5118
Wucherpfennig TG, Rohrbacher F, Pattabiraman VR, Bode JW. Angew Chem Int Ed, 2014, 53: 12244–12247
Wucherpfennig TG, Pattabiraman VR, Limberg FRP, Ruiz-Rodríguez J, Bode JW. Angew Chem Int Ed, 2014, 53: 12248–12252
Rohrbacher F, Deniau G, Luther A, Bode JW. Chem Sci, 2015, 6: 4889–4896
Pusterla I, Bode JW. Nat Chem, 2015, 7: 668–672
Li X, Danishefsky SJ. J Am Chem Soc, 2008, 130: 5446–5448
Li X, Yuan Y, Berkowitz WF, Todaro LJ, Danishefsky SJ. J Am Chem Soc, 2008, 130: 13222–13224
Wilson RM, Stockdill JL, Wu X, Li X, Vadola PA, Park PK, Wang P, Danishefsky SJ. Angew Chem Int Ed, 2012, 51: 2834–2848
Rao Y, Li X, Danishefsky SJ. J Am Chem Soc, 2009, 131: 12924–12926
Yuan Y, Zhu J, Li X, Wu X, Danishefsky SJ. Tetrahedron Lett, 2009, 50: 2329–2333
Wu X, Park PK, Danishefsky SJ. J Am Chem Soc, 2011, 133: 7700–7703
Wu X, Stockdill JL, Park PK, Danishefsky SJ. J Am Chem Soc, 2012, 134: 2378–2384
Wu X, Stockdill JL, Wang P, Danishefsky SJ. J Am Chem Soc, 2010, 132: 4098–4100
Kellogg RM. Angew Chem Int Ed, 1984, 23: 782–794
Mashraqui SH, Kellogg RM. J Am Chem Soc, 1983, 105: 7792–7793
Leleu S, Penhoat M, Bouet A, Dupas G, Papamicaël C, Marsais F, Levacher V. J Am Chem Soc, 2005, 127: 15668–15669
Chen W, Shao J, Hu M, Yu W, Giulianotti MA, Houghten RA, Yu Y. Chem Sci, 2013, 4: 970–976
Suppo JS, Subra G, Bergès M, Marcia de Figueiredo R, Campagne JM. Angew Chem Int Ed, 2014, 53: 5389–5393
Wu W, Zhang Z, Liebeskind LS. J Am Chem Soc, 2011, 133: 14256–14259
Kato S, Akada W, Mizuta M, Ishii Y. B Chem Soc Jpn, 1973, 46: 244–248
Aspin SJ, Taillemaud S, Cyr P, Charette AB. Angew Chem Int Ed, 2016, 55: 13833–13837
Tian J, Gao WC, Zhou DM, Zhang C. Org Lett, 2012, 14: 3020–3023
Zhang C, Liu SS, Sun B, Tian J. Org Lett, 2015, 17: 4106–4109
Hu L, Xu S, Zhao Z, Yang Y, Peng Z, Yang M, Wang C, Zhao J. J Am Chem Soc, 2016, 138: 13135–13138
Sklarz B, Al-Sayyab AF. J Chem Soc (Resumed), 1964: 1318–1320
Krishnamurthy M, Vishwanatha T, Panguluri N, Panduranga V, Sureshbabu V. Synlett, 2015, 26: 2565–1569
Lundberg H, Tinnis F, Selander N, Adolfsson H. Chem Soc Rev, 2014, 43: 2714–2742
Ishihara K, Ohara S, Yamamoto H. J Org Chem, 1996, 61: 4196–4197
Gernigon N, Al-Zoubi RM, Hall DG. J Org Chem, 2012, 77: 8386–8400
Liu S, Yang Y, Liu X, Ferdousi FK, Batsanov AS, Whiting A. Eur J Org Chem, 2013, 2013: 5692–5700
Mohy El Dine T, Erb W, Berhault Y, Rouden J, Blanchet J. J Org Chem, 2015, 80: 4532–4544
El Dine TM, Rouden J, Blanchet J. Chem Commun, 2015, 51: 16084–16087
Mali SM, Jadhav SV, Gopi HN. Chem Commun, 2012, 48: 7085–7087
Leighty MW, Shen B, Johnston JN. J Am Chem Soc, 2012, 134: 15233–15236
Schwieter KE, Shen B, Shackleford JP, Leighty MW, Johnston JN. Org Lett, 2014, 16: 4714–4717
Shackleford JP, Shen B, Johnston JN. Proc Natl Acad Sci USA, 2012, 109: 44–46
Shen B, Makley DM, Johnston JN. Nature, 2010, 465: 1027–1032
Li J, Lear MJ, Kawamoto Y, Umemiya S, Wong AR, Kwon E, Sato I, Hayashi Y. Angew Chem Int Ed, 2015, 54: 12986–12990
Jbara M, Maity SK, Seenaiah M, Brik A. J Am Chem Soc, 2016, 138: 5069–5075
Maity SK, Jbara M, Laps S, Brik A. Angew Chem Int Ed, 2016, 55: 8108–8112
Tang S, Si YY, Wang ZP, Mei KR, Chen X, Cheng JY, Zheng JS, Liu L. Angew Chem Int Ed, 2015, 54: 5713–5717
Acknowledgments
This work was supported by the National Natural Science Foundation of China (21462023), and the Natural Science Foundation of Jiangxi Province (20143ACB20007, 20153BCB23018).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, J., Zhao, J. Recent developments in peptide ligation independent of amino acid side-chain functional group. Sci. China Chem. 61, 97–112 (2018). https://doi.org/10.1007/s11426-017-9056-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-017-9056-5