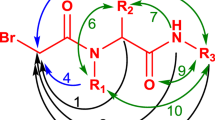
Abstract
A series of compounds containing the 1H-pyrazino[1,2-a]quinoxaline-1,5(6H)-dione scaffold have been synthesized using a one-pot procedure under microwave irradiation. This new strategy allowed for the facile synthesis of target compounds in good yields and could be readily applied to the construction of diverse libraries of compounds for high throughput screening in medicinal chemistry.
Similar content being viewed by others
References
Dömling A, Ugi I. Multicomponent reactions with isocyanides. Angew Chem Int Ed, 2000, 39: 3168–3210
Humphrey GR, Kuethe JT. Practical methodologies for the synthesis of indoles. Chem Rev, 2006, 106: 2875–2911
Dömling A, Wang W, Wang K. Chemistry and biology of multicomponent reactions. Chem Rev, 2012, 112: 3083–3135
Xu ZG, Moliner FD, Cappelli AP, Hulme C. Ugi/Aldol sequence: expeditious entry to several families of densely substituted nitrogen heterocycles. Angew Chem Int Ed, 2012, 51: 8037–8040
Xu ZG, Moliner FD, Cappelli AP, Hulme C. Aldol reactions in multicomponent reaction based domino pathways: a multipurpose enabling tool in heterocyclic chemistry. Org Lett, 2013, 15: 2738–2741
Chen ZZ, Zhang J, Tang DY, Xu ZG. Synthesis of fused benzimidazole-quinoxalinones via UDC strategy and following the intermolecular nucleophilic substitution reaction. Tetrahedron Lett, 2014, 55: 2742–2744
Richter HG, Benson GM, Blum D, Chaput E, Feng S, Gardes C, Grether U, Hartman P, Kuhn B, Martin RE, Plancher JM, Rudolph MG, Schuler F, Taylor S, Bleicher KH. Discovery of novel and orally active FXR agonists for the potential treatment of dyslipidemia & diabetes. Bioorg Med Chem Lett, 2011, 21: 191–194
Watson EL, Marsden SP, Raw SA. A robust, efficient catalyst system for enolate arylation leading to quaternary 3-aminooxindoles. Tetrahedron Lett, 2009, 50: 3318–3320
Hulme C, Chappeta S, Griffith C, Lee YS, Dietrich J. An efficient solution phase synthesis of triazadibenzoazulenones: designer isonitrile free methodology enabled by microwaves. Tetrahedron Lett, 2009, 50: 1939–1942
Faggi C, Garcia-Valverde M, Marcaccini S, Pepino R, Pozo MC. Studies on isocyanides and related compounds: a facile synthesis of 1,6-di-hydro-6-oxopyrazine-2-carboxylic acid derivatives via Ugi four-component condensation. Synthesis, 2003, 10: 1553–1558
Jhonny A, Abdelaziz EM, José MP, Alberto C, Franco F, Eddy S. Multicomponent assembly of diverse pyrazin-2(1H)-one chemotypes. J Org Chem, 2013, 78: 4402–4409
Johannes E, Horbert R, Schlosser J, Schmidt D, Peifer C. Effective synthesis of 3,5-diaryl-(1H)-pyrazin-2-ones via microwave mediated ring closure. Tetrahedron Lett, 2013, 54: 4067–4072
Ilyin A, Kysil V, Krasavin M, Kurashvili I, Ivachtchenko AV. Complexity-enhancing acid-promoted rearrangement of tricyclic products of tandem Ugi 4CC/intramolecular Diels-Alder reaction. J Org Chem, 2006, 71: 9544–9547
Oikawa M, Ikoma M, Sasaki M. Parallel synthesis of tandem Ugi/Diels-Alder reaction products on a soluble polymer support directed toward split-pool realization of a small molecule library. Tetrahedron Lett, 2005, 46: 415–418
Zhang W, Tempest P. Highly efficient microwave-assisted fluorous Ugi and post-condensation reactions for benzimidazoles and quinoxalinones. Tetrahedron Lett, 2004, 45: 6757–6760
Nixey T, Tempest P, Hulme C. Two-step solution-phase synthesis of novel quinoxalinones utilizing a UDC (Ugi/de-Boc/cyclize) strategy. Tetrahedron Lett, 2002, 43: 1637–1639
Chen S, Foss Jr FW. Aerobic organocatalytic oxidation of aryl aldehydes: flavin catalyst turnover by Hantzsch’s ester. Org Lett, 2012, 14: 5150–5153
Xu HJ, Zhao YQ, Feng T, Feng YS. Synthesis of 2,4-diamino- quinazolines and tricyclic quinazolines by cascade reductive cyclization of methyl N-cyano-2-nitrobenzimidates. J Org Chem, 2012, 77: 2649–2658
Mizuhara T, Oishi S, Fujii N, Ohno H. Efficient synthesis of pyrimido[ 1,2-c] [1,3]benzothiazin-6-imines and related tricyclic heterocycles by SNAr-Type C-S, C-N, or C-O bond formation with heterocumulenes. J Org Chem, 2010, 75: 265–268
Suchý M, Hudson RHE. Pyrimidine-fused heterocyclic frameworks based on an N4-arylcytosine scaffold: synthesis, characterization, and PNA oligomerization of the fluorescent cytosine analogue 5,6-ben-zopC. J Org Chem, 2014, 79: 3336–3347
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Zhang, X., Song, G., Cui, H. et al. Facile microwave-assisted synthesis of 1H-pyrazino[1,2-a]quinoxaline-1,5(6H)-dione derivatives via a nucleophilic substitution reaction. Sci. China Chem. 58, 1239–1242 (2015). https://doi.org/10.1007/s11426-015-5431-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-015-5431-9