Abstract
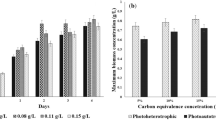
The potential success of microalgal biofuels greatly depends on the sustainability of the chosen pathway to produce them. Hydrothermal liquefaction (HTL) is a promising route to convert wet algal biomass into biocrude. Recycling the resulting HTL aqueous phase (AP) aims not only to recover nutrients from this effluent but also to use it as a substrate to close the photosynthetic loop and produce algal biomass again and process this biomass again into new biocrude. With that purpose, the response to AP recycling of five Chlorellaceae strains was monitored over five cultivation cycles. After four successive cycles of dynamic growth under nutrient-replete conditions, the microalgae were cultivated for a prolonged fifth cycle of 18 days in order to assess the impact of the AP on lipid and biomass accumulation under nutrient-limited conditions. Using AP as a substrate reduced the demand for external sources of N, S, and P while producing a significant amount of biomass (2.95–4.27 g/L) among the strains, with a lipid content ranging from 16 to 36%. However, the presence of the AP resulted in biomass with suboptimal properties, as it slowed down the accumulation of lipids and thus reduced the overall energy content of the biomass in all strains. Although Chlorella vulgaris NIES 227 did not have the best growth on AP, it did maintain the best lipid productivity of all the tested strains. Understanding the impact of AP on microalgal cultivation is essential for further optimizing biofuel production via the HTL process.
Graphical Abstract







Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Almutairi AW (2022) Full utilization of marine microalgal hydrothermal liquefaction liquid products through a closed-loop route: towards enhanced bio-oil production and zero-waste approach. 3 Biotech 12:209. https://doi.org/10.1007/s13205-022-03262-8
Aruoja V, Sihtmäe M, Dubourguier HC, Kahru A (2011) Toxicity of 58 substituted anilines and phenols to algae Pseudokirchneriella subcapitata and bacteria Vibrio fischeri: comparison with published data and QSARs. Chemosphere 84:1310–1320. https://doi.org/10.1016/j.chemosphere.2011.05.023
Bassoli SC, da Fonseca YA, Wandurraga HJL et al (2023) Research progress, trends, and future prospects on hydrothermal liquefaction of algae for biocrude production: a bibliometric analysis. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-023-03905-7
Bezergianni S, Dimitriadis A, Kikhtyanin O, Kubička D (2018) Refinery co-processing of renewable feeds. Prog Energy Combust Sci 68:29–64. https://doi.org/10.1016/j.pecs.2018.04.002
Biller P, Ross AB (2011) Potential yields and properties of oil from the hydrothermal liquefaction of microalgae with different biochemical content. Bioresour Technol 102:215–225. https://doi.org/10.1016/j.biortech.2010.06.028
Biller P, Ross AB, Skill SC et al (2012) Nutrient recycling of aqueous phase for microalgae cultivation from the hydrothermal liquefaction process. Algal Res 1:70–76. https://doi.org/10.1016/j.algal.2012.02.002
Biller P, Madsen RB, Klemmer M et al (2016) Effect of hydrothermal liquefaction aqueous phase recycling on bio-crude yields and composition. Bioresour Technol 220:190–199. https://doi.org/10.1016/j.biortech.2016.08.053
Channiwala SA, Parikh PP (2002) A unified correlation for estimating HHV of solid, liquid and gaseous fuels. Fuel 81:1051–1063. https://doi.org/10.1016/S0016-2361(01)00131-4
Chen L, Zhu T, Fernandez JSM et al (2017) Recycling nutrients from a sequential hydrothermal liquefaction process for microalgae culture. Algal Res 27:311–317. https://doi.org/10.1016/j.algal.2017.09.023
Chen PH, Venegas Jimenez JL, Rowland SM et al (2020) Nutrient recycle from algae hydrothermal liquefaction aqueous phase through a novel selective remediation approach. Algal Res 46:101776–101776. https://doi.org/10.1016/j.algal.2019.101776
Cheng F, Cui Z, Chen L et al (2017) Hydrothermal liquefaction of high- and low-lipid algae: bio-crude oil chemistry. Appl Energy 206:278–292. https://doi.org/10.1016/j.apenergy.2017.08.105
da Costa Magalhães B, Matricon L, Ramirez Romero L-A et al (2023) Catalytic hydrotreatment of bio-oil from continuous HTL of Chlorella sorokiniana and Chlorella vulgaris microalgae for biofuel production. Biomass Bioenergy 173:106798. https://doi.org/10.1016/j.biombioe.2023.106798
Dandamudi KPR, Mathew M, Selvaratnam T et al (2021) Recycle of nitrogen and phosphorus in hydrothermal liquefaction biochar from Galdieria sulphuraria to cultivate microalgae. Resour Conserv Recycl 171:105644. https://doi.org/10.1016/j.resconrec.2021.105644
Das P, AbdulQuadir M, Thaher M et al (2020) A feasibility study of utilizing hydrothermal liquefaction derived aqueous phase as nutrients for semi-continuous cultivation of Tetraselmis sp. Bioresour Technol 295:122310–122310. https://doi.org/10.1016/j.biortech.2019.122310
Di Caprio F (2021) A fattening factor to quantify the accumulation ability of microorganisms under N-starvation. New Biotechnol. https://doi.org/10.1016/j.nbt.2021.04.001
Djandja OS, Wang Z, Chen L et al (2020) Progress in hydrothermal liquefaction of algal biomass and hydrothermal upgrading of the subsequent crude bio-oil: a mini review. Energy Fuels 34:11723–11751. https://doi.org/10.1021/acs.energyfuels.0c01973
DuBois M, Gilles KA, Hamilton JK et al (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
European Committee for Standardization (2008). EN 14214:2008 - Automotive fuels - Fatty acid methyl esters (FAME) for diesel engines - Requirements and test methods. Retrieved from https://standards.iteh.ai/catalog/standards/cen/d3475989-a401-45ea-a3b3-27c9b71ab152/en-14214-2008
Gai C, Zhang Y, Chen WT et al (2015) Characterization of aqueous phase from the hydrothermal liquefaction of Chlorella pyrenoidosa. Bioresour Technol 184:328–335. https://doi.org/10.1016/j.biortech.2014.10.118
Godwin CM, Hietala DC, Lashaway AR et al (2017) Algal polycultures enhance coproduct recycling from hydrothermal liquefaction. Bioresour Technol 224:630–638. https://doi.org/10.1016/j.biortech.2016.11.105
Goncalves EC, Koh J, Zhu N et al (2016) Nitrogen starvation-induced accumulation of triacylglycerol in the green algae: evidence for a role for ROC40, a transcription factor involved in circadian rhythm. Plant J 85:743–757. https://doi.org/10.1111/tpj.13144
Hietala DC, Savage PE (2021) A molecular, elemental, and multiphase kinetic model for the hydrothermal liquefaction of microalgae. Chem Eng J 407:127007. https://doi.org/10.1016/j.cej.2020.127007
Hong Y, Yang L, You X et al (2023) Effects of light quality on microalgae cultivation: bibliometric analysis, mini-review, and regulation approaches. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-023-31192-2
Hutner SH, Provasoli L, Schatz A, Haskins CP (1950) Some approaches to the study of the role of metals in the metabolism of microorganisms. Proc Am Philos Soc 94:152–170
Jena U, Vaidyanathan N, Chinnasamy S, Das KC (2011) Evaluation of microalgae cultivation using recovered aqueous co-product from thermochemical liquefaction of algal biomass. Bioresour Technol 102:3380–3387. https://doi.org/10.1016/j.biortech.2010.09.111
Kim B, Praveenkumar R, Lee J et al (2016) Magnesium aminoclay enhances lipid production of mixotrophic Chlorella sp. KR-1 while reducing bacterial populations. Bioresour Technol 219:608–613. https://doi.org/10.1016/j.biortech.2016.08.034
Kumar V, Kumar S, Chauhan PK et al (2019) Low-temperature catalyst based hydrothermal liquefaction of harmful macroalgal blooms, and aqueous phase nutrient recycling by microalgae. Sci Rep 9:1–9. https://doi.org/10.1038/s41598-019-47664-w
Kumar V, Jaiswal KK, Vlaskin MS et al (2022) Hydrothermal liquefaction of municipal wastewater sludge and nutrient recovery from the aqueous phase. Biofuels 13:657–662. https://doi.org/10.1080/17597269.2020.1863627
LaPanse AJ, Krishnan A, Posewitz MC (2020) Adaptive laboratory evolution for algal strain improvement: methodologies and applications. Algal Res 102122. https://doi.org/10.1016/j.algal.2020.102122
Leng L, Yang L, Chen J et al (2021) Valorization of the aqueous phase produced from wet and dry thermochemical processing biomass: a review. J Clean Prod 294:126238. https://doi.org/10.1016/j.jclepro.2021.126238
Leow SR, Witter JR, Vardon D et al (2015) Prediction of microalgae hydrothermal liquefaction products from feedstock biochemical composition. Green Chem 17:3584–3599. https://doi.org/10.1039/C5GC00574D
Li T, Zheng Y, Yu L, Chen S (2013) High productivity cultivation of a heat-resistant microalga Chlorella sorokiniana for biofuel production. Bioresour Technol 131:60–67. https://doi.org/10.1016/j.biortech.2012.11.121
Marangon BB, Castro JS, Assemany PP et al (2022) Environmental performance of microalgae hydrothermal liquefaction: life cycle assessment and improvement insights for a sustainable renewable diesel. Renew Sustain Energy Rev 155:111910. https://doi.org/10.1016/j.rser.2021.111910
Marchello AE, dos Santos AC, Lombardi AT et al (2018) Physiological and ecological aspects of Chlorella sorokiniana (Trebouxiophyceae) under photoautotrophic and mixotrophic conditions. Microb Ecol 76:791–800. https://doi.org/10.1007/s00248-018-1170-8
Matricon L, Roubaud A, Haarlemmer G, Geantet C (2023) The challenge of nitrogen compounds in hydrothermal liquefaction of algae. J Supercrit Fluids 105867. https://doi.org/10.1016/j.supflu.2023.105867
Moser BR, Williams A, Haas MJ, McCormick RL (2009) Exhaust emissions and fuel properties of partially hydrogenated soybean oil methyl esters blended with ultra low sulfur diesel fuel. Fuel Process Technol 90:1122–1128. https://doi.org/10.1016/j.fuproc.2009.05.004
Omidvarborna H, Kumar A, Kim D-S (2015) NOx emissions from low-temperature combustion of biodiesel made of various feedstocks and blends. Fuel Process Technol 140:113–118. https://doi.org/10.1016/j.fuproc.2015.08.031
Ovsyannikova E, Kruse A, Becker GC (2020) Feedstock-dependent phosphate recovery in a pilot-scale hydrothermal liquefaction bio-crude production. Energies 13:379. https://doi.org/10.3390/en13020379
Pham M, Schideman L, Scott J et al (2013) Chemical and biological characterization of wastewater generated from hydrothermal liquefaction of Spirulina. Environ Sci Technol 47:2131–2138. https://doi.org/10.1021/es304532c
Pham XP (2014) Influences of Molecular Profiles of Biodiesels on Atomization, Combustion, and Emission Characteristics. Ph.D. Thesis. The University of Sydney, Sydney, Australia. http://hdl.handle.net/2123/12812
Prestigiacomo C, Scialdone O, Galia A (2022) Hydrothermal liquefaction of wet biomass in batch reactors: critical assessment of the role of operating parameters as a function of the nature of the feedstock. J Supercrit Fluids 189:105689. https://doi.org/10.1016/j.supflu.2022.105689
Přibyl P, Cepák V, Zachleder V (2012) Production of lipids in 10 strains of Chlorella and Parachlorella, and enhanced lipid productivity in Chlorella vulgaris. Appl Microbiol Biotechnol 94:549–561. https://doi.org/10.1007/s00253-012-3915-5
Ramírez-Romero A, Da Costa MB, Dimitriades-Lemaire A et al (2022) Chlorellaceae feedstock selection under balanced nutrient limitation. Fermentation 8:554. https://doi.org/10.3390/fermentation8100554
Ramírez-Romero A, Martin M, Boyer A et al (2023) Microalgae adaptation as a strategy to recycle the aqueous phase from hydrothermal liquefaction. Bioresource Technology 128631. https://doi.org/10.1016/j.biortech.2023.128631
Ratha SK, Renuka N, Abunama T et al (2022) Hydrothermal liquefaction of algal feedstocks: the effect of biomass characteristics and extraction solvents. Renew Sustain Energy Rev 156:111973. https://doi.org/10.1016/j.rser.2021.111973
Saber M, Takahashi F, Yoshikawa K (2018) Characterization and application of microalgae hydrochar as a low-cost adsorbent for Cu(II) ion removal from aqueous solutions. Environ Sci Pollut Res 25:32721–32734. https://doi.org/10.1007/s11356-018-3106-8
Shen X-F, Qin Q-W, Yan S-K et al (2019) Biodiesel production from Chlorella vulgaris under nitrogen starvation in autotrophic, heterotrophic, and mixotrophic cultures. J Appl Phycol 31:1589–1596. https://doi.org/10.1007/s10811-019-01765-1
Stablein MJ, Baracho DH, Watson JT et al (2021) Microalgal photosynthetic inhibition and mixotrophic growth in Post Hydrothermal Liquefaction Wastewater (PHW). Algal Res 60:102548. https://doi.org/10.1016/j.algal.2021.102548
SundarRajan P, Gopinath KP, Arun J et al (2021) Insights into valuing the aqueous phase derived from hydrothermal liquefaction. Renew Sustain Energy Rev 144:111019. https://doi.org/10.1016/j.rser.2021.111019
Templeton DW, Laurens LML (2015) Nitrogen-to-protein conversion factors revisited for applications of microalgal biomass conversion to food, feed and fuel. Algal Res 11:359–367. https://doi.org/10.1016/j.algal.2015.07.013
Watson J, Wang T, Si B et al (2020) Valorization of hydrothermal liquefaction aqueous phase: pathways towards commercial viability. Prog Energy Combust Sci 77:100819–100819. https://doi.org/10.1016/j.pecs.2019.100819
Yoo G, Park MS, Yang J-W, Choi M (2015) Lipid content in microalgae determines the quality of biocrude and Energy Return On Investment of hydrothermal liquefaction. Appl Energy 156:354–361. https://doi.org/10.1016/j.apenergy.2015.07.020
Yu G, Zhang Y, Schideman L et al (2011) Distributions of carbon and nitrogen in the products from hydrothermal liquefaction of low-lipid microalgae. Energy Environ Sci 4:4587–4595. https://doi.org/10.1039/c1ee01541a
Zhan H, Zhuang X, Song Y et al (2018) Insights into the evolution of fuel-N to NOx precursors during pyrolysis of N-rich nonlignocellulosic biomass. Appl Energy 219:20–33. https://doi.org/10.1016/j.apenergy.2018.03.015
Zhang J, Qian C, Yan K et al (2019) Hydrothermal liquefaction of Enteromorpha prolifera for bio-crude production: an experimental study on aqueous phase recirculation. Trans Asabe 62:1113–1119. https://doi.org/10.13031/trans.13418
Acknowledgements
The authors express their gratitude to Alexandra Dimitriades and Gatien Fleury for their contributions and assistance in the development of the analytical methods utilized in this study.
Funding
The funding for this project has been provided by the Agence Nationale de la Recherche (ANR) under the French project RAFBIOALG, with the funding number ANR 18-CE43-0009–04.
Author information
Authors and Affiliations
Contributions
Adriana Ramírez-Romero contributed to conceptualization, methodology, validation, data curation, and writing of the original draft as well as reviewing and editing the manuscript. Bruno da Costa Magalhães contributed to methodology and reviewing and editing of the manuscript. Lucie Matricon contributed to methodology. Jean-François Sassi contributed to funding acquisition and reviewing and editing of the manuscript. Jean-Philippe Steyer contributed to validation and reviewing and editing of the manuscript. Florian Delrue contributed to funding, conceptualization, methodology, validation, and reviewing and editing of the manuscript. The first draft of the manuscript was written by Adriana Ramírez-Romero, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ta Yeong Wu
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ramírez-Romero, A., da Costa Magalhães, B., Matricon, L. et al. Aqueous phase recycling: impact on microalgal lipid accumulation and biomass quality. Environ Sci Pollut Res (2024). https://doi.org/10.1007/s11356-024-32701-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11356-024-32701-7