Abstract
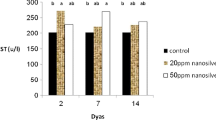
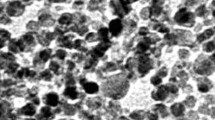
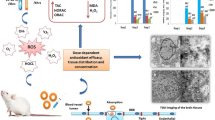
Nanosized lanthanum oxide particles (La2O3) are commonly utilized in various industries. The potential health risks associated with La2O3 nanoparticles, cytotoxic effects at varying doses and time intervals, and the mechanisms behind their induction of behavioral changes remain uncertain and necessitate further investigation. Therefore, this study examined in vivo hepatotoxicity, considering the quantity (60, 150, and 300 mg/kg) and time-dependent induction of reactive oxygen species (ROS) over one week or 21 days. The mice received intraperitoneal injections of three different concentrations in Milli-Q water. Throughout the experiments, no physical changes or weight loss were observed among the groups. However, after 21 days, only the highest concentration showed signs of anxiety in the activity cage (p < 0.05). Subsequently, all animals treated with La2O3 NPs exhibited a significant loss of learning and memory recall using the Active Avoidances test, after 21 days (p < 0.001). Markers for anti-reactive oxygen species (ROS) such as superoxide dismutase (SOD) were significantly upregulated in response to all concentrations of NPs after seven days compared to the control group. This was confirmed by a significant increase in glutathione peroxidase (Gpx1) and pro-apoptotic Caspase-3 expression at the lowest and highest doses. Additionally, both transcription and protein levels of the anti-apoptotic BCL-2 surpassed P53 protein in a dosage-dependent manner, indicating activation of the primary anti-apoptosis pathway. After 21 days, P53 levels exceeded BCL-2 protein levels, confirming a significant loss of BCL-2 mRNA, particularly at the 300 mg/kg concentration. Furthermore, a higher transcription level of Caspase-3, SOD, and Gpx1 was observed, with the highest values detected at the 300 mg/kg concentration, indicating the activation of cell death. Histopathological analysis of the liver illustrated apoptotic bodies resulting from La2O3 NP concentration. The investigation revealed multiple inflammatory foci, cytoplasmic degeneration, steatosis, and DNA fragmentation consistent with increased damage over time due to higher concentrations. Blood samples were also analyzed to determine liver enzymatic changes, including alkaline phosphatase (ALP), alanine transaminase (ALT), aspartate aminotransferase (AST), and lipid profiles. The results showed significant differences among all La2O3 NP concentrations, with the most pronounced damage observed at the 300 mg/kg dose even after 21 days. Based on an animal model, this study suggests that La2O3 hepatotoxicity is likely caused by the size and shape of nanoparticles (NPs), following a dose and time-dependent mechanism that induces the production of reactive oxygen species and behavioral changes such as anxiety and memory loss.








Similar content being viewed by others
Data availability
All relevant data are within the paper.
References
Afifi M, Saddick S, Abu Zinada OA (2016) Toxicity of silver nanoparticles on the brain of Oreochromis niloticus and Tilapia zillii. Saudi J Biol Sci 23(6):754–760. https://doi.org/10.1016/j.sjbs.2016.06.008
Alarifi S, Ali D, Alkahtani S (2017) Oxidative Stress-Induced DNA Damage by Manganese Dioxide Nanoparticles in Human Neuronal Cells. Biomed Res Int 2017:5478790. https://doi.org/10.1155/2017/5478790
Al-Ghamdi H, Dong M, Sayyed MI, Wang C, Almuqrin AH, Almasoud FI (2021) The Vital Role of La2O3 on the La2O3-CaO-B2O3-SiO2 Glass System for Shielding Some Common Gamma Ray Radioactive Sources. Materials (Basel) 14(17). https://doi.org/10.3390/ma14174776
Almukhlafi H, Ali D, Almutairi B, Yaseen KN, Alyami N, Almeer R, . . . Alarifi S (2021) Role of Oxidative Stress in La2O3 Nanoparticle-Induced Cytotoxicity and Apoptosis in CHANG and HuH-7 Cells. Int J Nanomedicine 16:3487–3496. https://doi.org/10.2147/IJN.S302478
Alyami NM, Almeer R, Alyami HM (2022a) Role of green synthesized platinum nanoparticles in cytotoxicity, oxidative stress, and apoptosis of human colon cancer cells (HCT-116). Heliyon 8(12):e11917. https://doi.org/10.1016/j.heliyon.2022.e11917
Alyami NM, Alyami HM, Almeer R (2022b) Using green biosynthesized kaempferol-coated sliver nanoparticles to inhibit cancer cells growth: An in vitro study using hepatocellular carcinoma (HepG2). Cancer Nanotechnol 13(1):26
An L, Liu S, Yang Z, Zhang T (2012) Cognitive impairment in rats induced by nano-CuO and its possible mechanisms. Toxicol Lett 213(2):220–227. https://doi.org/10.1016/j.toxlet.2012.07.007
Bedoui S, Herold MJ, Strasser A (2020) Emerging connectivity of programmed cell death pathways and its physiological implications. Nat Rev Mol Cell Biol 21(11):678–695. https://doi.org/10.1038/s41580-020-0270-8
Bi M, Zhang H, Yuan L, Zhao L, Liu R (2019) Molecular mechanisms of lead-induced changes of selenium status in mice livers through interacting with selenoprotein P. Ecotoxicol Environ Saf 175:282–288. https://doi.org/10.1016/j.ecoenv.2019.03.059
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Chen L, Zhang B, Toborek M (2013) Autophagy is involved in nanoalumina-induced cerebrovascular toxicity. Nanomedicine: Nanotechnology, Biology and Medicine 9(2):212–221
Chen YJ, Deng SM, Chen HW, Tsao CH, Chen WT, Cheng SJ, . . . Huang GJ (2021) Follistatin mediates learning and synaptic plasticity via regulation of Asic4 expression in the hippocampus. Proc Natl Acad Sci U S A 118(39). https://doi.org/10.1073/pnas.2109040118
Chen C, Zhou C, Zhang W, Liu H, Wang M, Li F, . . . Cao Y (2022) Effect and Mechanism of PINK1/Parkin-Mediated Mitochondrial Autophagy in Rat Lung Injury Induced by Nano Lanthanum Oxide. Nanomaterials (Basel) 12(15). https://doi.org/10.3390/nano12152594
Del Rio Oliva M, Kirk CJ, Groettrup M, Basler M (2022) Effective therapy of polymyositis in mice via selective inhibition of the immunoproteasome. Eur J Immunol 52(9):1510–1522. https://doi.org/10.1002/eji.202249851
Dey R, Pandey A, Rai VK (2014) The Er3+-Yb3+ codoped La2O3 phosphor in finger print detection and optical heating. Spectrochim Acta A Mol Biomol Spectrosc 128:508–513. https://doi.org/10.1016/j.saa.2014.02.175
Dhakshinamoorthy V, Manickam V, Perumal E (2017) Neurobehavioural Toxicity of Iron Oxide Nanoparticles in Mice. Neurotox Res 32(2):187–203. https://doi.org/10.1007/s12640-017-9721-1
Dhar SK, Xu Y, St Clair DK (2010) Nuclear factor kappaB- and specificity protein 1-dependent p53-mediated bi-directional regulation of the human manganese superoxide dismutase gene. J Biol Chem 285(13):9835–9846. https://doi.org/10.1074/jbc.M109.060715
Drane P, Bravard A, Bouvard V, May E (2001) Reciprocal down-regulation of p53 and SOD2 gene expression-implication in p53 mediated apoptosis. Oncogene 20(4):430–439. https://doi.org/10.1038/sj.onc.1204101
Dressler VL, Ogunmodede OT, Heidrich GM, Neves VM, Schetinger MRC, Morsch VM (2020) Investigative Analysis of Lanthanum Oxide Nanoparticles on Elements in Bone of Wistar Rats After 30 Days of Repeated Oral Administration. Biol Trace Elem Res 196(1):153–167. https://doi.org/10.1007/s12011-019-01907-z
Fan J, Shi Y, Liu H, Wang S, Luan L, Duan L, . . . Wei X (2022) Atomic Layer Deposition of Ultrathin La2O3/Al2O3 Nanolaminates on MoS2 with Ultraviolet Ozone Treatment. Materials (Basel) 15(5). https://doi.org/10.3390/ma15051794
Farmer G, Friedlander P, Colgan J, Manley JL, Prives C (1996) Transcriptional repression by p53 involves molecular interactions distinct from those with the TATA box binding protein. Nucleic Acids Res 24(21):4281–4288. https://doi.org/10.1093/nar/24.21.4281
Gomez-Perez S, McKeever L, Sheean P (2020) Tutorial: A Step-by-Step Guide (Version 2.0) for Measuring Abdominal Circumference and Skeletal Muscle From a Single Cross-Sectional Computed-Tomography Image Using the National Institutes of Health ImageJ. JPEN J Parenter Enteral Nutr 44(3):419–424. https://doi.org/10.1002/jpen.1721
He C, Jiang S, Yao H, Zhang L, Yang C, Jiang S, . . . Chen X (2019) High-content analysis for mitophagy response to nanoparticles: A potential sensitive biomarker for nanosafety assessment. Nanomedicine 15(1):59–69. https://doi.org/10.1016/j.nano.2018.09.003
Hemann MT, Lowe SW (2006) The p53-Bcl-2 connection. Cell Death Differ 13(8):1256–1259. https://doi.org/10.1038/sj.cdd.4401962
Huang GJ, Edwards A, Tsai CY, Lee YS, Peng L, Era T, . . . Flint J (2014) Ectopic cerebellar cell migration causes maldevelopment of Purkinje cells and abnormal motor behaviour in Cxcr4 null mice. PLoS One 9(2):e86471. https://doi.org/10.1371/journal.pone.0086471
Kiatsaengthong D, Jaroenpanon K, Somchuea P, Chukeaw T, Chareonpanich M, Faungnawakij K, . . . Seubsai A (2022) Effects of Mg, Ca, Sr, and Ba Dopants on the Performance of La2O3 Catalysts for the Oxidative Coupling of Methane. ACS Omega 7(2):1785–1793. https://doi.org/10.1021/acsomega.1c04738
Kunhikrishnan A, Rahman MA, Lamb D, Bolan NS, Saggar S, Surapaneni A, Chen C (2022) Rare earth elements (REE) for the removal and recovery of phosphorus: A review. Chemosphere 286(Pt 2):131661. https://doi.org/10.1016/j.chemosphere.2021.131661
Lim CH (2015) Toxicity of Two Different Sized Lanthanum Oxides in Cultured Cells and Sprague-Dawley Rats. Toxicol Res 31(2):181–189. https://doi.org/10.5487/TR.2015.31.2.181
de Lima MN, Polydoro M, Laranja DC, Bonatto F, Bromberg E, Moreira JC, . . . Schroder N (2005) Recognition memory impairment and brain oxidative stress induced by postnatal iron administration. Eur J Neurosci 21(9):2521–2528. https://doi.org/10.1111/j.1460-9568.2005.04083.x
Llewellyn BD (2009) Nuclear staining with alum hematoxylin. Biotech Histochem 84(4):159–177. https://doi.org/10.1080/10520290903052899
Lu VM, McDonald KL (2020) Lanthanum nanoparticles to target the brain: proof of biodistribution and biocompatibility with adjuvant therapies. Nanomedicine (lond) 15(22):2107–2117. https://doi.org/10.2217/nnm-2020-0165
Lu VM, McDonald KL, Townley HE (2017) Realizing the therapeutic potential of rare earth elements in designing nanoparticles to target and treat glioblastoma. Nanomedicine (lond) 12(19):2389–2401. https://doi.org/10.2217/nnm-2017-0193
Lu VM, Jue TR, McDonald KL (2020) Cytotoxic lanthanum oxide nanoparticles sensitize glioblastoma cells to radiation therapy and temozolomide: an in vitro rationale for translational studies. Sci Rep 10(1):18156. https://doi.org/10.1038/s41598-020-75372-3
Malhotra N, Hsu HS, Liang ST, Roldan MJM, Lee JS, Ger TR, Hsiao CD (2020) An Updated Review of Toxicity Effect of the Rare Earth Elements (REEs) on Aquatic Organisms. Animals (Basel) 10(9). https://doi.org/10.3390/ani10091663
Meng Z, Wang S, Wang H, Wang L, Wang S (2019) La2O3-coated Li2ZnTi3O8@C as a high performance anode for lithium-ion batteries. RSC Adv 9(36):20618–20623. https://doi.org/10.1039/c9ra03846a
Paiva AV, de Oliveira MS, Yunes SN, de Oliveira LG, Cabral-Neto JB, de Almeida CE (2009) Effects of lanthanum on human lymphocytes viability and DNA strand break. Bull Environ Contam Toxicol 82(4):423–427. https://doi.org/10.1007/s00128-008-9596-1
Pang C, Fan KS, Wei L, Kolar MK (2021) Gene therapy in wound healing using nanotechnology. Wound Repair Regen 29(2):225–239. https://doi.org/10.1111/wrr.12881
Pratt DS, Kaplan MM (2000) Evaluation of abnormal liver-enzyme results in asymptomatic patients. N Engl J Med 342(17):1266–1271. https://doi.org/10.1056/NEJM200004273421707
Rao X, Huang X, Zhou Z, Lin X (2013) An improvement of the 2^(-delta delta CT) method for quantitative real-time polymerase chain reaction data analysis. Biostat Bioinforma Biomath 3(3):71–85. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/25558171
Sahani S, Sharma YC (2021) Advancements in applications of nanotechnology in global food industry. Food Chem 342:128318. https://doi.org/10.1016/j.foodchem.2020.128318
Salama DM, Abd El-Aziz ME, Rizk FA, Abd Elwahed MSA (2021) Applications of nanotechnology on vegetable crops. Chemosphere 266:129026. https://doi.org/10.1016/j.chemosphere.2020.129026
Shin SH, Lim CH, Kim YS, Lee YH, Kim SH, Kim JC (2017) Twenty-eight-day repeated inhalation toxicity study of nano-sized lanthanum oxide in male sprague-dawley rats. Environ Toxicol 32(4):1226–1240. https://doi.org/10.1002/tox.22319
Shishkin AV, Shishkin VY, Pankratov AA, Burdina AA, Zaikov YP (2022) Electrochemical Reduction of La2O3, Nd2O3, and CeO2 in LiCl-Li2O Melt. Materials (Basel) 15(11). https://doi.org/10.3390/ma15113963
Sisler JD, Li R, McKinney W, Mercer RR, Ji Z, Xia T, . . . Qian Y (2016a) Differential pulmonary effects of CoO and La2O3 metal oxide nanoparticle responses during aerosolized inhalation in mice. Part Fibre Toxicol 13(1):42. https://doi.org/10.1186/s12989-016-0155-3
Sisler JD, Pirela SV, Shaffer J, Mihalchik AL, Chisholm WP, Andrew ME, . . . Qian Y (2016b) Toxicological Assessment of CoO and La2O3 Metal Oxide Nanoparticles in Human Small Airway Epithelial Cells. Toxicol Sci 150(2):418–428. https://doi.org/10.1093/toxsci/kfw005
Titford M (2005) The long history of hematoxylin. Biotech Histochem 80(2):73–78. https://doi.org/10.1080/10520290500138372
Tommasi F, Thomas PJ, Pagano G, Perono GA, Oral R, Lyons DM, . . . Trifuoggi M (2021) Review of Rare Earth Elements as Fertilizers and Feed Additives: A Knowledge Gap Analysis. Arch Environ Contam Toxicol 81(4):531–540. https://doi.org/10.1007/s00244-020-00773-4
Vilmosh N, Delev D, Kostadinov I, Zlatanova H, Kotetarova M, Kandilarov I, Kostadinova I (2022) Anxiolytic Effect of Satureja montana Dry Extract and its Active Compounds Rosmarinic Acid and Carvacrol in Acute Stress Experimental Model. J Integr Neurosci 21(5):124. https://doi.org/10.31083/j.jin2105124
Vincent-Johnson AJ, Schwab Y, Mann HS, Francoeur M, Hammonds JS Jr, Scarel G (2013) Origin of the low frequency radiation emitted by radiative polaritons excited by infrared radiation in planar La2O3 films. J Phys Condens Matter 25(3):035901. https://doi.org/10.1088/0953-8984/25/3/035901
Vorobyova V, Vasyliev G, Uschapovskiy D, Lyudmyla K, Skiba M (2022) Green synthesis, characterization of silver nanoparticals for biomedical application and environmental remediation. J Microbiol Methods 193:106384. https://doi.org/10.1016/j.mimet.2021.106384
Wang F, Hu E, Wang J, Yu L, Hong S, Kim JS, Zhu B (2022) Tuning La2O3 to high ionic conductivity by Ni-doping. Chem Commun (camb) 58(27):4360–4363. https://doi.org/10.1039/d1cc07183a
Wu Y, Zhou H, Wu K, Lee S, Li R, Liu X (2014) PTEN phosphorylation and nuclear export mediate free fatty acid-induced oxidative stress. Antioxid Redox Signal 20(9):1382–1395. https://doi.org/10.1089/ars.2013.5498
Xiao Z, Yue L, Wang C, Chen F, Ding Y, Liu Y, . . . Wang Z (2021) Downregulation of the photosynthetic machinery and carbon storage signaling pathways mediate La2O3 nanoparticle toxicity on radish taproot formation. J Hazard Mater 411:124971. https://doi.org/10.1016/j.jhazmat.2020.124971
Xue Y, Wu J, Sun J (2012) Four types of inorganic nanoparticles stimulate the inflammatory reaction in brain microglia and damage neurons in vitro. Toxicol Lett 214(2):91–98. https://doi.org/10.1016/j.toxlet.2012.08.009
Yasuda S, Inoue K, Matsui I, Matsumoto A, Katsuma Y, Okushima H, . . . Isaka Y (2023) Hepatic phosphate uptake and subsequent nerve-mediated phosphaturia are crucial for phosphate homeostasis following portal vein passage of phosphate in rats. Sci Rep 13(1):5794. https://doi.org/10.1038/s41598-023-32856-2
Yokoyama T, Omotehara T, Hirano T, Kubota N, Yanai S, Hasegawa C, . . . Hoshi N (2018) Identification of reference genes for quantitative PCR analyses in developing mouse gonads. J Vet Med Sci 80(10):1534–1539. https://doi.org/10.1292/jvms.18-0417
Yuan H, Duan S, Guan T, Yuan X, Lin J, Hou S, . . . Chen S (2020a) Corrigendum to "Vitexin protects against ethanol-induced liver injury through Sirt1/p53 signaling pathway" [Eur. J. Pharmacol. 873 (2020) 173007]. Eur J Pharmacol 880:173160. https://doi.org/10.1016/j.ejphar.2020.173160
Yuan L, Bai D, Meng L, Wang H, Sun Z, An T, . . . Zhang X (2020b) Effects of intragastric administration of La2O3 nanoparticles on mouse testes. J Toxicol Sci 45(8):411–422. https://doi.org/10.2131/jts.45.411
Yuan L, Bai D, Meng L, Wang H, Sun Z, An T, . . . Zhang X (2020c) Effects of intragastric administration of La(2)O(3) nanoparticles on mouse testes. J Toxicol Sci 45(8):411–422. https://doi.org/10.2131/jts.45.411
Yuan L, Li Q, Bai D, Shang X, Hu F, Chen Z, . . . Zhang X (2020d) La(2)O(3) Nanoparticles Induce Reproductive Toxicity Mediated by the Nrf-2/ARE Signaling Pathway in Kunming Mice. Int J Nanomedicine 15:3415–3431. https://doi.org/10.2147/IJN.S230949
Yuan L, Qu Y, Li Q, An T, Chen Z, Chen Y, . . . Bai D (2020e) Protective effect of astaxanthin against La2O3 nanoparticles induced neurotoxicity by activating PI3K/AKT/Nrf-2 signaling in mice. Food Chem Toxicol 144:111582. https://doi.org/10.1016/j.fct.2020.111582
Yuan L, Qu Y, Li Q, An T, Chen Z, Chen Y, . . . Bai D (2020f) Protective effect of astaxanthin against La(2)O(3) nanoparticles induced neurotoxicity by activating PI3K/AKT/Nrf-2 signaling in mice. Food Chem Toxicol 144:111582. https://doi.org/10.1016/j.fct.2020.111582
Zhang Y, Li CS, Ye Y, Johnson K, Poe J, Johnson S, . . . Madhu C (2006) Porcine brain microvessel endothelial cells as an in vitro model to predict in vivo blood-brain barrier permeability. Drug Metab Dispos 34(11):1935–1943. https://doi.org/10.1124/dmd.105.006437
Acknowledgements
The writer would like to thank the Researchers Supporting Project number (RSP2023R177), King Saud University, Riyadh, Saudi Arabia.
Funding
This project was funded by Researchers Supporting Project number (RSP2023R177) King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
NA1, HA1, DA, RA, and SA conceptualized the study; NA1 and HA1 were responsible for the data curation; HA2, ZA, NA3, and AR were responsible for the formal analysis; NA1 was accountable for the funding acquisition; NA2, NA3, SM, DE, and AR were responsible for the investigation; NA2, NA3, KY, and ZA were responsible for the methodology; SA, RA, and DA were accountable for the project administration; DA and BA were responsible for the software; SA, RA, and NA1 supervised the study; DA was responsible for the validation; NA1 wrote the original draft; and WA and HA2 reviewed, and edited the manuscript.
Corresponding author
Ethics declarations
Ethics statement
The Research Ethics Committee (REC) authorized the regulations and procedures governing the handling and care of animals, and the animals were treated under (KSU-SE-22–75).
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Conflicts of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alyami, N.M., Alobadi, H., Maodaa, S. et al. Determination of dose- and time-dependent hepatotoxicity and apoptosis of Lanthanum oxide nanoparticles in female Swiss albino mice. Environ Sci Pollut Res 31, 17124–17139 (2024). https://doi.org/10.1007/s11356-024-32209-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-32209-0