Abstract
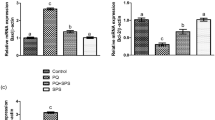
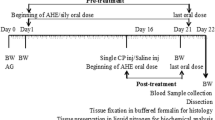
Paraquat (PQ) is an organic compound, which is commonly used as a herbicide in the agriculture sector, and it is also known to stimulate critical damages in the male reproductive system. Gossypetin (GPTN) is one of important members of the flavonoid family, which is an essential compound in flowers and calyx of Hibiscus sabdariffa with potential pharmacological properties. The current investigation was aimed to examine the ameliorative potential of GPTN against PQ-instigated testicular damages. Adult male Sprague–Dawley rats (n = 48) were distributed into four groups: control, PQ (5 mg/kg), PQ + GPTN (5 mg/kg + 30 mg/kg respectively), and GPTN (30 mg/kg). After 56 days of treatment, biochemical, spermatogenic indices, hormonal, steroidogenic, pro-or-anti-apoptotic, and histopathological parameters were estimated. PQ exposure disturbed the biochemical profile by reducing the activities of catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GPx), and glutathione reductase (GSR), while it increased the concentration of reactive oxygen species (ROS) and malondialdehyde (MDA) level. Furthermore, PQ exposure decreased the sperm motility, viability, number of hypo-osmotic tail swelled spermatozoa, and epididymal sperm count; additionally, it increased sperm morphological (head mid-piece and tail) abnormalities. Moreover, PQ lessened the follicle-stimulating hormone (FSH), luteinizing hormone (LH), and plasma testosterone levels. Besides, PQ-intoxication downregulated the gene expression of steroidogenic enzymes (StAR, 3β-HSD, and 17β-HSD) and anti-apoptotic marker (Bcl-2), whereas upregulated the gene expression of apoptotic markers (Bax and Caspase-3). PQ exposure led to histopathological damages in testicular tissues as well. Nonetheless, GPTN inverted all the illustrated impairments in testes. Taken together, GPTN could potently ameliorate PQ-induced reproductive dysfunctions due to its antioxidant, androgenic, and anti-apoptotic potential.



Similar content being viewed by others
Data availability
The datasets used/analyzed in this study are available from the corresponding author on reasonable request.
The study was approved by the Institutional Biosafety/Bioethics Committee (IBC) of the University of Agriculture, Faisalabad, in compliance with this (CEE Council 86/609) protocol.
References
Ahmed MM (2010) Radio and chemoprotective properties of hesperidin against genotoxicity induced by gamma radiation and/ or paraquat in rat bone marrow cells. J Radiol Sci Appl 23:233–244
Ali SS, Ahsan H, Zia MK et al (2020) Understanding oxidants and antioxidants: classical team with new players. J Food Biochem 44:13145. https://doi.org/10.1111/jfbc.13145
Anam C, Marhendra AP, Rahayu S (2019) Alcohol Intake Investigation of Adult Rats Based on Sperm Parameters. J Exp Life Sci 9, 128–132. https://doi.org/10.21776/ub.jels.2019.009.02.11
Asghari MH, Moloudizargari M, Bahadar H et al (2017) A review of the protective effect of melatonin in pesticide-induced toxicity. Expert Opin Drug Metab Toxicol 13:545–554. https://doi.org/10.1080/17425255.2016.1214712
Aslani BA, Ghobadi S (2016) Studies on oxidants and antioxidants with a brief glance at their relevance to the immune system. Life Sci 146:163–173. https://doi.org/10.1016/j.lfs.2016.01.014
Barbosa ML, de Meneses AAPM, de Aguiar RPS et al (2020) Oxidative stress, antioxidant defense and depressive disorders: a systematic review of biochemical and molecular markers. Neurol Psychiatry Brain Res 36:65–72. https://doi.org/10.1016/j.npbr.2020.02.006
Birben E, Sahiner UM, Sackesen C et al (2012) Oxidative stress and antioxidant defense. World Allergy Organ J 5:9–19. https://doi.org/10.1097/WOX.0b013e3182439613
Blanco-Ayala T, Anderica-Romero AC, Pedraza-Chaverri J (2014) New insights into antioxidant strategies against paraquat toxicity. Free Radic Res 48:623–640. https://doi.org/10.3109/10715762.2014.899694
Cao Z, Shao B, Xu F et al (2017) Protective effect of selenium on aflatoxin B1-induced testicular toxicity in mice. Biol Trace Elem Res 180:233–238. https://doi.org/10.1007/s12011-017-0997-z
Carlberg I, Mannervik B (1975) Purification and characterization of the flavoenzyme glutathione reductase from rat liver. J Biol Chem 250:5475–5480. https://doi.org/10.1016/S0021-9258(19)41206-4
Castillo AF, Orlando U, Helfenberger KE et al (2015) The role of mitochondrial fusion and StAR phosphorylation in the regulation of StAR activity and steroidogenesis. Mol Cell Endocrinol 408:73–79. https://doi.org/10.1016/j.mce.2014.12.011
Chance B, Maehly A (1955) [136] assay of catalases and peroxidases. Methods Enzymol 2:764–775. https://doi.org/10.1016/S0076-6879(55)02300-8
Chen Z, Zuo X, Li H et al (2017) Effects of melatonin on maturation, histone acetylation, autophagy of porcine oocytes and subsequent embryonic development. Anim Sci J 88:1298–1310. https://doi.org/10.1111/asj.12779
Chikere OU, Uchechi EE, Ikenna IM et al (2020) Ameliorative effect of aframomum melegueta (Alligator Pepper) against paraquat induced testicular damage. World J Pharm Res 9:2105–2124. https://doi.org/10.20959/wjpr20205-17442
Correa JR, Zavos PM (1994) The hypoosmotic swelling test: its employment as an assay to evaluate the functional integrity of the frozen–thawed bovine sperm membrane. Theriogenology 42:351–360. https://doi.org/10.1016/0093-691X(94)90280-1
Das J, Ghosh J, Manna P et al (2012) Taurine protects rat testes against doxorubicin–induced oxidative stress as well as p53, Fas and caspase 12–mediated apoptosis. Amino Acids 42:1839–1855. https://doi.org/10.1007/s00726-011-0904-4
Davalli P, Mitic T, Caporali A et al (2016) ROS, cell senescence, and novel molecular mechanisms in aging and age-related diseases. Oxid Med Cell Longev 2016. https://doi.org/10.1155/2016/3565127
Fois AG, Paliogiannis P, Sotgia S et al (2018) Evaluation of oxidative stress biomarkers in idiopathic pulmonary fibrosis and therapeutic applications: a systematic review. Respir Res 19:51. https://doi.org/10.1186/s12931-018-0754-7
Fortenberry GZ, Beckman J, Schwartz A et al (2016) Magnitude and characteristics of acute paraquat-and diquat-related illnesses in the US: 1998–2013. Environ Res 146:191–199. https://doi.org/10.1016/j.envres.2016.01.003
Ghasemi Y, Tayebi-Khosroshahi H, Abedi B et al (2019) Tiny non-coding RNAs in body fluids, possible biomarkers for autosomal dominant polycystic kidney disease. Iran J Kidney Dis 13:151–164
Gu YP, Yang XM, Duan ZH et al (2017) Inhibition of chemotherapy-induced apoptosis of testicular cells by squid ink polysaccharide. Exp Ther Med 14:5889–5895. https://doi.org/10.3892/etm.2017.5342
Gulsheen, Kumar, A. and Sharma, A., 2019. Antianxiety and antidepressant activity guided isolation and characterization of gossypetin from Hibiscus sabdariffa Linn. calyces. J Biol Act Prod Nat 9: 205–214. https://doi.org/10.1080/22311866.2019.1615552
Halvaei I, Roodsari HRS, Harat ZN (2012) Acute effects of Ruta graveolens L. on sperm parameters and DNA integrity in rats. J Reprod Fertil 13:33–38
Hayashi I, Morishita Y, Imai K et al (2007) High-throughput spectrophotometric assay of reactive oxygen species in serum. Mutat Res Genet Toxicol Environ Mutagen 631:55–61. https://doi.org/10.1016/j.mrgentox.2007.04.006
Huang W, Quan C, Duan P et al (2016) Nonylphenol induced apoptosis and autophagy involving the Akt/mTOR pathway in prepubertal Sprague-Dawley male rats in vivo and in vitro. Toxicology 373:41–53. https://doi.org/10.1016/j.tox.2016.11.006
Ighodaro OM, Akinloye OA (2018) First line defence antioxidants–superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria J Med 54:287–293. https://doi.org/10.1016/j.ajme.2017.09.001
Ijaz MU, Tahir A, Samad A et al (2021a) Nobiletin ameliorates nonylphenol-induced testicular damage by improving biochemical, steroidogenic, hormonal, spermatogenic, apoptotic and histological profile. Hum Exp Toxicol 40:403–416. https://doi.org/10.1177/0960327120950007
Ijaz MU, Anwar H, Iqbal S et al (2021b) Protective effect of myricetin on nonylphenol-induced testicular toxicity: biochemical, steroidogenic, hormonal, spermatogenic, and histological-based evidences. Environ Sci Pollut Res 28:22742–22757. https://doi.org/10.1007/s11356-020-12296-5
Kakkar P, Das B, Viswanathan PN (1984) A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biophys 21:130–132
Khan A, Manna K, Das DK et al (2015a) Gossypetin ameliorates ionizing radiation-induced oxidative stress in mice liver—a molecular approach. Free Radic Res 49:1173–1186. https://doi.org/10.3109/10715762.2015.1053878
Kamel F (2013) Paths from pesticides to Parkinson’s. Sciences 341: 722 –723. https://doi.org/10.1126/science.1243619
Kaur P, Dhandayuthapani S, Venkatesan T et al (2020) Molecular mechanism of C-phycocyanin induced apoptosis in LNCaP cells. Bioorg Med Chem 28:115272. https://doi.org/10.1016/j.bmc.2019.115272
Kenjale R, Shah R, Sathaye S (2008) Effects of Chlorophytum borivilianum on sexual behaviour and sperm count in male rats. Phytotherapy Research: An International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives 22: 796–801. https://doi.org/10.1002/ptr.2369
Kim JW, Kim DS (2020) Paraquat: toxicology and impacts of its ban on human health and agriculture. Weed Sci 68:208–213. https://doi.org/10.1017/wsc.2019.70
Kuan CM, Lin ST, Yen TH et al (2016) Paper-based diagnostic devices for clinical paraquat poisoning diagnosis. Biomicrofluidics 10:034118. https://doi.org/10.1063/1.4953257
Lawrence RA, Burk RF (1976) Glutathione peroxidase activity in selenium–deficient rat liver. Biochem Biophys Res Commun 71:952–958. https://doi.org/10.1016/0006-291X(76)90747-6
Lee MS, Tsai CW, Wang CP et al (2017) Anti-prostate cancer potential of gossypetin via inducing apoptotic and autophagic cell death. Mol Carcinog 56:2578–2592. https://doi.org/10.1002/mc.22702
Li H, Hong T, Zhu Q et al (2019) Paraquat exposure delays late-stage Leydig cell differentiation in rats during puberty. Environ Pollut 255:113316. https://doi.org/10.1016/j.envpol.2019.113316
Lin HH, Hsieh MC, Wang CP et al (2021) Anti-atherosclerotic effect of gossypetin on abnormal vascular smooth muscle cell proliferation and migration. Antioxidants 10:1357. https://doi.org/10.3390/antiox10091357
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Meng XX, Wang RL, Gao O (2013) Effect of alinastation on Paraquat induced oxidative stress in humans type II alveolar epithelial cells. World J Emerg Med 4:133–137. https://doi.org/10.5847/wjem.j.issn.1920-8642.2013.02.009
Momtaz S, Abdollahi M (2012) A comprehensive review of biochemical and molecular evidences from animal and human studies on the role of oxidative stress in aging: an epiphenomenon or the cause. Asian J Anim Vet Adv. https://doi.org/10.3923/ajava.2012
Mussi MA (2010) Calcaterra NB (2010) Paraquat-induced oxidative stress response during amphibian early embryonic development. Compara Biochem Physiol Part C 151:240–247. https://doi.org/10.1016/j.cbpc.2009.11.003
Nair N (2015) Dose-dependent short–term study of di-n-butyl phthalate on the testicular antioxidant system of Wistar rats. Environ Sci Pollut Res 22:2196–2204. https://doi.org/10.1007/s11356-014-3457-8
Nasibeh F, Mohammad H, Morteza H et al (2015) Effects of paraquat on testicular histomorphometry of male rats. Biol Forum 7:573–575
O’Shaughnessy PJ, Monteiro A, Verhoeven G et al (2010) Effect of FSH on testicular morphology and spermatogenesis in gonadotropin-deficient hypogonadal mice lacking androgen receptors. Reproduction 139:177–184. https://doi.org/10.1530/REP-09-0377
Ofoego UC, Ekwujuru EU, Nwakanma A et al (2018) Protective and ameliorating effects of methanolic seed extract of Mucuna Pruriens on paraquat induced testicular damage. Adv Life Sc Technol 53:8–16
Ojha A, Srivastava N (2014) In vitro studies on organophosphate pesticides induced oxidative DNA damage in rat lymphocytes. Mutat Res Genet Toxicol Environ Mutagen 761:10–17. https://doi.org/10.1016/j.mrgentox.2014.01.007
Okorondu M, Okorondu S, Alisi C et al (2019) Ameliorative effect of psidium guajava leaf extract on paraquat induced renal and reproductive hormone toxicity. Sci Res J 7:2201–2796. https://doi.org/10.31364/SCIRJ/v7.i4.2019.P0419633
O’Shaughnessy PJ (2014) Hormonal control of germ cell development and spermatogenesis. Semin Cell Dev Biol 29:55–65. https://doi.org/10.1016/j.semcdb.2014.02.010
Patel DK, Patel K (2020) P0174 Nephroprotective activity of gossypetin through inhibitory effect on xanthine oxidase, nuclear factor kappa B (NF-KB) and soluble epoxide hydrolase (SEH): in-vitro and in-silico experiment. Nephrol Dial Transplant 35:142–174. https://doi.org/10.1093/ndt/gfaa142.P0174
Patel DK, Patel K (2021a) P-MD005. Neuroprotective effects of gossypetin in Alzheimer’s disease: therapeutic approaches to evaluate the acetylcholinesterase and butyl cholinesterase inhibitory potential. Clin Neurophysiol 132:97–98. https://doi.org/10.1016/j.clinph.2021.02.225
Patel K, Patel DK (2021) Therapeutic benefit and pharmacological activities of gossypetin: biological importance in the medicine through scientific research data analysis. Metab Clin Exp 116:154550. https://doi.org/10.1016/j.metabol.2020
Pawar JS, Mustafa S, Ghosh I (2017) Gossypetin inhibits ROS generation in HeLa and HepG2 cell lines. Res Rev J Toxicol 7:25–31
Placer ZA, Cushman L, Johnson BC (1966) Estimation of products of lipid peroxidation (malonyldialdehyde) in biochemical systems. Anal Biochem 16:359–364. https://doi.org/10.1016/0003-2697(66)90167-9
Qian JY, Deng P, Yu ZP et al (2019) 8-Formylophiopogonanone B antagonizes paraquat-induced hepatotoxicity by suppressing oxidative stress. Front Pharmacol 10:1283. https://doi.org/10.3389/fphar.2019.01283
Ramaswamy S, Weinbauer GF (2015) Endocrine control of spermatogenesis: Role of FSH and LH/ testosterone. Spermatogenesis 4: 996025. https://doi.org/10.1080/21565562.2014.996025
Rasooli R, Kamali Y, Mandegary A (2020) Effects of pirfenidone, vitamin E, and pirfenidone–vitamin E combination in paraquat-induced pulmonary fibrosis. Comp Clin Path 29:667–673
Salvamani S, Gunasekaran B, Shaharuddin NA et al (2014) Antiartherosclerotic effects of plant flavonoids. Biomed Res Int: 2014: 480258. https://doi.org/10.1155/2014/480258
Sandström J, Broyer A, Zoia D et al (2017) Potential mechanisms of development-dependent adverse effects of the herbicide paraquat in 3D rat brain cell cultures. Neurotoxicology 60:116–124. https://doi.org/10.1016/j.neuro.2017.04.010
Tremblay JJ (2015) Molecular regulation of steroidogenesis in endocrine Leydig cells. Steroids 103:3–10. https://doi.org/10.1016/j.steroids.2015.08.001
Venkatadri R, Muni T, Iyer AKV et al (2016) Role of apoptosis-related miRNAs in resveratrol-induced breast cancer cell death. Cell Death Dis 7:2104. https://doi.org/10.1038/cddis.2016.6
Xie X, Liu K, Liu F et al (2019) Gossypetin is a novel MKK3 and MKK6 inhibitor that suppresses esophageal cancer growth in vitro and in vivo. Cancer Lett 442:126–136. https://doi.org/10.1016/j.canlet.2018.10.016
Ye L, Su Z, Ge R (2011) Inhibitors of testosterone biosynthetic and metabolic activation enzymes. Molecules 16:9983–10001. https://doi.org/10.3390/molecules16129983
Yokoi K, Uthus EO, Nielsen FH (2003) Nickel deficiency diminishes sperm quantity and movement in rats. Biol Trace Elem Res 93:141–154. https://doi.org/10.1385/BTER:93:1-3:141
Zhao M, Gu L, Li Y et al (2019) Chitooligosaccharides display anti-tumor effects against human cervical cancer cells via the apoptotic and autophagic pathways. Carbohydr Polym 224:115171. https://doi.org/10.1016/j.carbpol.2019.115171
Zirkin BR, Papadopoulos V (2018) Leydig cells: formation, function, and regulation. Biol Reprod 99:101–111. https://doi.org/10.1093/biolre/ioy059
Funding
The authors did not receive any financial support for this research.
Author information
Authors and Affiliations
Contributions
SM and MUI conceived the idea and designed the study. MUI, SM, HA1, and SI performed the experiments. HA2 and SI helped in statistical analysis. MUI, SM, and QA wrote the manuscript. All the authors read and approved the final version of manuscript.
Corresponding author
Ethics declarations
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mustafa, S., Anwar, H., Ain, Q.u. et al. Therapeutic effect of gossypetin against paraquat-induced testicular damage in male rats: a histological and biochemical study. Environ Sci Pollut Res 30, 62237–62248 (2023). https://doi.org/10.1007/s11356-023-26469-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-26469-5