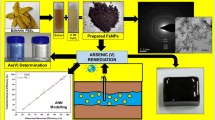
Abstract
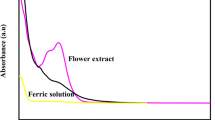
The optimal conditions for the green synthesis of nano zero-valent iron (G-NZVI) using mango peel extract were investigated using a Box-Behnken design approach. Three factors were considered, namely the ratio of iron solution to mango peel extract ratio (1:1–1:3), feeding rate of mango peel extract (1–5 mL min−1), and agitation speed (300–350 rpm). The results showed that the optimal conditions for the synthesis of G-NZVI for arsenate removal were a 1:1 ratio of iron solution to mango peel extract, a mango peel extract feeding rate of 5 mL min−1, and an agitation speed of 300 rpm. Under these conditions, nearly 100% arsenate removal was achieved. X-ray diffractometry (XRD), Fourier transform infrared spectroscopy (FT-IR), transmission electron microscopy (TEM), Brunauer–Emmett–Teller (BET), and scanning electron microscopy (SEM) with energy-dispersive X-ray analysis (EDX) methods were used to characterize the properties of the G-NZVI. Finally, the arsenate removal efficiency of the G-NZVI was compared against that of commercial nano zero-valent iron (C-NZVI). The results revealed that the G-NZVI was roughly five times more efficient at arsenate removal than the C-NZVI. The influence of background species such as chloride (Cl−), phosphate (PO43−), calcium (Ca2+), and sulfate (SO42−) was studied to evaluate their effects on arsenate removal. As a result, Cl− and Ca2+ were shown to play a role in promoting arsenate removal, whereas SO42− and PO43− were observed to play an inhibiting role.






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Ajila CM, Aalami M, Leelavathi K, Prasada Rao UJS (2010) Mango peel powder: a potential source of antioxidant and dietary fiber in macaroni preparations. IFSET 11(1):219–224. https://doi.org/10.1016/j.ifset.2009.10.004
Berardini N, Fezer R, Conrad J, Beifuss U, Carle R, Schieber A (2005) Screening of mango (Mangifera indica L.) cultivars for their contents of flavonol O- and xanthone C-glycosides, anthocyanins, and pectin. J Agric Food Chem 53(5):1563–1570. https://doi.org/10.1021/jf0484069
Choe S, Liljestrand HM, Khim J (2004) Nitrate reduction by zero-valent iron under different pH regimes. Appl Geochem 19(3):335–342. https://doi.org/10.1016/j.apgeochem.2003.08.001
Deewan R, Tanboonchuy V (2022) Green synthesis of zero-valent iron nanoparticle using mango peel extract by Box-Behnken design experimental. Proceeding of 11th International Conference on Environmental Engineering, Science and Management 12–13 May 2022
Desalegn B, Megharaj M, Chen Z, Naidu R (2019) Green synthesis of zero valent iron nanoparticle using mango peel extract and surface characterization using XPS and GC-MS. Heliyon 5(5):1–9. https://doi.org/10.1016/j.heliyon.2019.e01750
Eljamal R, Eljamal O, Maamoun I, Yilmaz G, Sugihara Y (2020) Enhancing the characteristics and reactivity of nZVI: polymers effect and mechanisms. J Mol Liq 315:113714. https://doi.org/10.1016/j.molliq.2020.113714
Fatoki JO, Badmus JA, Fatoki CO, Adekunle AS, Adeleke GE, Kehinde SA (2019) Induction of oxidative stress: a possible mechanism for the arsenic induced catastrophes in male wistar rats. Life Sci Adv 75:23–32. https://doi.org/10.7176/alst/75-04
Fazlzadeh M, Rahmani K, Zarei A, Abdoallahzadeh H, Nasiri F, Khosravi R (2017) A novel green synthesis of zero valent iron nanoparticles (NZVI) using three plant extracts and their efficient application for removal of Cr(VI) from aqueous solutions. Adv Powder Technol 28(1):122–130. https://doi.org/10.1016/j.apt.2016.09.003
Fu F, Lu J, Cheng Z, Tang B (2016) Removal of selenite by zero-valent iron combined with ultrasound: Se(IV) concentration changes, Se(VI) generation, and reaction mechanism. Ultrason Sonochem 29:328–336. https://doi.org/10.1016/j.ultsonch.2015.10.007
Hoag GE, Collins JB, Holcomb JL, Hoag JR, Nadagoudab MN, Varma RS (2009) Degradation of bromothymol blue by “greener” nano-scale zero-valent iron synthesized using tea polyphenols. J Mater Chem 19(45):8671–8677. https://doi.org/10.1039/B909148C
Hsu JC, Lin CJ, Liao CH, Chen ST (2008) Evaluation of the multiple-ion competition in the adsorption of As(V) onto reclaimed iron-oxide coated sands by fractional factorial design. Chemosphere 72(7):1049–1055. https://doi.org/10.1016/j.chemosphere.2008.03.060
Ignat I, Volf I, Popa VI (2011) A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem 126(4):1821–1835. https://doi.org/10.1016/j.foodchem.2010.12.026
Jegadeesan G, Mondal K, Lalvani SB (2005) Arsenate remediation using nanosized modified zerovalent iron particles. Environ Prog 24(3):289–296. https://doi.org/10.1002/ep.10072
Karavasilis M, Tsakiroglou CD (2019) Synthesis of aqueous suspensions of zero-valent iron nanoparticles (NZVI) from plant extracts: experimental study and numerical modeling. Emerg Sci J 3(6):344–360. https://doi.org/10.28991/esj-2019-01197
Kim H, Yong Moon J, Kim H, Lee DS, Cho M, Choi HK, Kim YS, Mosaddik A, Cho SM (2010) Antioxidant and antiproliferative activities of mango (Mangifera Indica L.) flesh and peel. Food Chem 121(2):429–436. https://doi.org/10.1016/j.foodchem.2009.12.060
Khamdahsag P, Yan DYS, Poompang P, Supannafai N, Tanboonchuy V (2021) Continuous fixed-bed column studies of arsenite removal via oxidation and adsorption coprocesses. J Water Process Eng 42:102176. https://doi.org/10.1016/j.jwpe.2021.102176
Khashij M, Dalvand A, Mehralian M, Ebrahimi AA, Khosravi R (2020) Removal of reactive black 5 dye using aero valent iron nanoparticles produced by a novel green synthesis method. Pigment Resin Technol 49(3):215–221. https://doi.org/10.1108/PRT-10-2019-0092
Lin J, Sun M, Liu X, Chen Z (2017) Functional kaolin supported nanoscale zero-valent iron as a Fenton-like catalyst for the degradation of Direct Black G. Chemosphere 184:664–672. https://doi.org/10.1016/j.chemosphere.2017.06.038
Mystrioti C, Sparis D, Papasiopi N, Xenidis A, Dermatas D, Chrysochoou M (2015) Assessment of polyphenol coated nano zero valent iron for hexavalent chromium removal from contaminated waters. BECT 94(3):302–307. https://doi.org/10.1007/s00128-014-1442-z
Nadagouda MN, Castle AB, Murdock RC, Hussain SM, Varma RS (2010) In vitro biocompatibility of nanoscale zerovalent iron particles (NZVI) synthesized using tea polyphenols. Green Chem 12(1):114–122. https://doi.org/10.1039/B921203P
Nakseedee P, Tanboonchuy V, Pimpha N, Khemthong P, Liao CH, Grisdanurak N (2015) Arsenic removal by nanoiron coupled with gas bubbling system. J Taiwan Inst Chem Eng 47:182–189. https://doi.org/10.1016/j.jtice.2014.10.006
Parks JL, Novak J, Macphee M, Itle C, Edwards M (2003) Effect of Ca on As releases from ferric and alum residuals. J AWWA 95(6):108–118. https://doi.org/10.1002/j.1551-8833.2003.tb10391.x
Phanthasri J, Khamdahsag P, Jutaporn P, Sorachoti K, Wantala K, Tanboonchuy V (2018) Enhancement of arsenite removal using manganese oxide coupled with iron (III) trimesic. Appl Surf Sci 427:545–552. https://doi.org/10.1016/j.apsusc.2017.09.032
Phanthasri J, Yan DYS, Wantala K, Khunphonoi R, Tanboonchuy V (2022) Reduction and adsorption co-processes for selenate removal by zeolite-supported nanoscale zero-valent iron. EASR 49(3):363–372. https://ph01.tci-thaijo.org/index.php/easr/article/view/246567. Accessed 1 Jun 2022
Poguberović SS, KrčmaraSnežan DM, Maletić SP, Kónya Z, Tomašević Pilipovic DD, Kerkez DV, Rončević SD (2016) Removal of As (III) and Cr (VI) from aqueous solutions using “ green ” zero-valent iron nanoparticles produced by oak, mulberry and cherry leaf extracts. Ecol Eng 90:42–49. https://doi.org/10.1016/j.ecoleng.2016.01.083
Rahaman MS, Mise N, Ichihara S (2022) Arsenic contamination in food chain in Bangladesh: a review on health hazards, socioeconomic impacts and implications. Hyg Environ Health Adv 2:100004. https://doi.org/10.1016/j.heha.2022.100004
Rakhmania KH, Yuzir MA, Fouad FA, Amalia Purba LA, Riyadi FA (2021) Application of Box-Behnken design to mineralization and color removal of palm oil mill effluent by electrocoagulation process. Environ Sci Pollut Res 27:12755–12766. https://doi.org/10.1007/s11356-021-16197-z
Rashtbari Y, Hazrati S, Azari A, Afshin A, Fazlzadeh M, Vosoughi M (2020) A novel, eco-friendly and green synthesis of PPAC-ZnO and PPAC-nZVI nanocomposite using pomegranate peel: cephalexin adsorption experiments, mechanisms, isotherms and kinetics. Adv Powder Technol 31(4):1612–1623. https://doi.org/10.1016/j.apt.2020.02.001
Sasaki K, Nakano H, Wilopo W, Maiura Y, Hirajima T (2009) Sorption and speciation of arsenic by zero-valent iron. Colloids Surf A Physicochem Eng 347(1–3):8–17. https://doi.org/10.1016/j.colsurfa.2008.10.033
Su C, Puls RW (2001) Arsenate and arsenite removal by zerovalent iron: effect of phosphate, silicate, carbonate, borate, sulfate, chromate, molybdate, and nitrate, relative to chloride. Environ Sci Technol 35(22):4562–4568. https://doi.org/10.1021/es010768z
Suwannatrai S, Yan DYS, Phanthasri J, Khamdahsag P, Wannapaiboon S, Tanboonchuy V (2020) Oxidation–adsorption of arsenite contaminated water over ceria nanorods. Desalin Water Treat 200:252–261. https://doi.org/10.5004/dwt.2020.26138
Tanboonchuy V, Grisdanurak N, Liao CH (2012) Nitrate probe for quantifying reducing power of nanoscale zero-valent iron. Sustain Environ Res 22(3):185–191
Tanboonchuy V, Hsu JC, Grisdanurak N, Liao CH (2010) Arsenate removal by nano zero-valent iron in the gas bubbling system. World Acad Sci Eng Technol 65:263–265. https://doi.org/10.5281/zenodo.1078849
Tanboonchuy V, Hsu JC, Grisdanurak N, Liao CH (2011a) Gas-bubbled nano zero-valent iron process for high concentration arsenate removal. J Hazard Mater 186(2–3):2123–2128. https://doi.org/10.1016/j.jhazmat.2010.12.125
Tanboonchuy V, Hsu JC, Grisdanurak N, Liao CH (2011b) Impact of selected solution factors on arsenate and arsenite removal by nanoiron particles. Environ Sci Pollut Res 18(6):857–864. https://doi.org/10.1007/s11356-011-0442-3
Vaishya RC, Gupta SK (2004) Modeling arsenic(V) removal from water by sulfate modified iron-oxide coated sand (SMIOCS). Sep Sci Technol 39(3):645–666. https://doi.org/10.1081/SS-120027999
Wang CB, Zhang WX (1997) Synthesizing nanoscale iron particles for rapid and complete dechlorination of TCE and PCBs. Environ Sci Technol 31(7):2154–2156. https://doi.org/10.1021/es970039c
Xiao C, Li H, Zhao Y, Zhang X, Wang X (2020) Green synthesis of iron nanoparticle by tea extract (polyphenols) and its selective removal of cationic dyes. J Environ Manage 275:111262. https://doi.org/10.1016/j.jenvman.2020.111262
Zhang C, Sun G, Quan B, Shi X, Xiao N, Zhang Y, Tong J, Wang W, Tang Y, Xiao B, Zhang C (2022) Preparation of Mn/Ti-modified zeolite and its performance for removing iron and manganese. Environ Sci Pollut Res 29(1–3):1–16. https://doi.org/10.1007/s11356-022-21309-4
Zhang W, Singh P, Paling E, Delides S (2004) Arsenic removal from contaminated water by natural iron ores. Miner Eng 17(4):517–524. https://doi.org/10.1016/j.mineng.2003.11.020
Zhu F, Ma S, Liu T, Deng X (2018) Green synthesis of nano zero-valent iron/Cu by green tea to remove hexavalent chromium from groundwater. J Clean Prod 174:184–190. https://doi.org/10.1016/j.jclepro.2017.10.302
Acknowledgements
This work was conducted under research focused on the development of novel technologies for safe agriculture by the Faculty of Engineering, Khon Kaen University, which has received funding support from the Fundamental Fund (The National Science, Research and Innovation Fund (NSRF), Thailand).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Ratthiwa Deewan, Dickson Yuk-Shing Yan, and Visanu Tanboonchuy. The first draft of the manuscript was written by Ratthiwa Deewan, Pummarin Khamdahsag, and Visanu Tanboonchuy. The manuscript was reviewed, revised, and approved by all authors.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent to publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deewan, R., Yan, D.YS., Khamdahsag, P. et al. Remediation of arsenic-contaminated water by green zero-valent iron nanoparticles. Environ Sci Pollut Res 30, 90352–90361 (2023). https://doi.org/10.1007/s11356-022-24535-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-24535-y