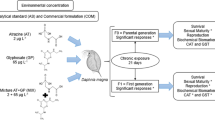
Abstract
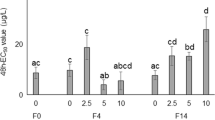
We evaluated the individual and combined effects of different environmentally representative concentrations of glyphosate (0, 25, 50, 75, and 100 µg l−1) and aminomethylphosphonic acid (AMPA; 0, 12.5, 25, 37.5, and 50 µg l−1) on the physiology of Aedes aegypti larvae, as well as the capacity of the aquatic macrophyte Salvinia molesta to attenuate those compounds’ toxicological effects. Larvae of Ae. aegypti (between the third and fourth larval stages) were exposed for 48 h to glyphosate and/or AMPA in the presence or absence of S. molesta. Glyphosate and AMPA induced sublethal responses in Ae. aegypti larvae during acute exposures. Plants removed up to 49% of the glyphosate and 25% of AMPA from the water, resulting in the exposure of larvae to lower concentration of those compounds in relation to media without plants. As a result, lesser effects of glyphosate and/or AMPA were observed on larval acetylcholinesterase, P450 reductase, superoxide dismutase, mitochondrial electron transport chain enzymes, respiration rates, and lipid peroxidation. In addition to evidence of deleterious effects by media contamination with glyphosate and AMPA on aquatic invertebrates, our results attest to the ability of S. molesta plants to mitigate the toxicological impacts of those contaminants.
Graphical abstract





Similar content being viewed by others
Data availability
Data available on request from the authors.
References
A. Hawkes H (1998) Origin and development of the biological monitoring working party score system. Water Res 32:964–968. https://doi.org/10.1016/S0043-1354(97)00275-3
Ahmed AM (2012) Lipid peroxidation and oxidative protein products as biomarkers of oxidative stress in the autogenous mosquito, Aedes caspius, upon infection with the mosquitocidal bacterium, Bacillus thuringiensis kurstaki. Pak J Zool 44:525–536
Ahouangninou C, Martin T, Assogba-Komlan F et al (2013) Using Aedes aegypti larvae to assess pesticide contamination of soil, groundwater and vegetables. Br Biotechnol J 3:143–157
Angeli S, Adams E, Masceno GP et al (2021) Toxicity assays of commercial chlorpyrifos under UV/H2O2 and UVC photolysis treatments. Water, Air, Soil Pollut 232:353. https://doi.org/10.1007/s11270-021-05314-w
Azmi WA, Hussin NH, Amin NM (2018) Monitoring of water quality using aquatic insects as biological indicators in three streams of Terengganu. J Sustain Sci Manag 13:67–76
Baglan H, Lazzari CR, Guerrieri FJ (2018) Glyphosate impairs learning in mosquito larvae (Aedes aegypti) at field-realistic doses. J Exp Biol. https://doi.org/10.1242/jeb.187518
Barman B (2014) The importance of aquatic insects as biomonitors of freshwater ecosystems. Int J Environ Nat Sci 1:82–85
Borrero Landazabal MA, Carreño Otero AL, Kouznetsov VV et al (2018) Alterations of mitochondrial electron transport chain and oxidative stress induced by alkaloid-like α-aminonitriles on Aedes aegypti larvae. Pestic Biochem Physiol 144:64–70. https://doi.org/10.1016/j.pestbp.2017.11.006
Cain DJ, Luoma SN, Carter JL, Fend SV (1992) Aquatic insects as bioindicators of trace element contamination in cobble-bottom rivers and streams. Can J Fish Aquat Sci 49:2141–2154. https://doi.org/10.1139/f92-237
Cristofaro CDS, Branco CWC, de Rocha MIA, Portugal SDGM (2021) Assessing glyphosate concentrations in six reservoirs of Paraíba do Sul and Guandu River Basins in southeast Brazil. Ambient e Agua - An Interdiscip J Appl Sci 16:1. https://doi.org/10.4136/ambi-agua.2615
Da Silva MD, Peralba MCR, Mattos MLT (2003) Determination of gliphosate and aminomethylphosphonic acid in superficial waters of Arroi Passo do Pilão. Pestic Rev Ecotoxicologia e Meio Ambient 13:19–28. https://doi.org/10.5380/pes.v13i0.3161
Dizer H, da Silva de Assis HC, Hansen P-D (2001) Cholinesterase activity as a bioindicator for monitoring marine pollution in the Baltic Sea and the Mediterranean Sea. In: Biomarkers in marine organisms. Elsevier, pp 331–342. https://doi.org/10.1016/B978-044482913-9/50014-6
Duke SO (2011) Glyphosate degradation in glyphosate-resistant and -susceptible crops and weeds. J Agric Food Chem 59:5835–5841. https://doi.org/10.1021/jf102704x
Duke SO (2021) Glyphosate: uses other than in glyphosate-resistant crops, mode of action, degradation in plants, and effects on non-target plants and agricultural microbes. Rev Environ Contam Toxicol 255:1–65. https://doi.org/10.1007/398_2020_53
El-Shenawy NS (2009) Oxidative stress responses of rats exposed to Roundup and its active ingredient glyphosate. Environ Toxicol Pharmacol 28:379–385. https://doi.org/10.1016/j.etap.2009.06.001
Gao R, Yuan Z, Zhao Z, Gao X (1998) Mechanism of pyrogallol autoxidation and determination of superoxide dismutase enzyme activity. Bioelectrochem Bioenerg 45:41–45. https://doi.org/10.1016/S0302-4598(98)00072-5
Giroux I (2015) Présence de pesticides dans l’eau au Québec : Portrait et tendances dans les zones de maïs et de soya – 2011 à 2014, Québec, ministère du Développement durable, de l’Environnement et de la Lutte contre les changements climatiques, Di rection du suivi de l’état de l’environnement, 47 p. + 5 ann. http://www.mddelcc.gouv.qc.ca/eau/flrivlac/pesticides.htm
Gomes MP, Juneau P (2016) Oxidative stress in duckweed (Lemna minor L.) induced by glyphosate: Is the mitochondrial electron transport chain a target of this herbicide? Environ Pollut 218:402–409. https://doi.org/10.1016/j.envpol.2016.07.019
Gomes MP, Smedbol E, Chalifour A et al (2014) Alteration of plant physiology by glyphosate and its by-product aminomethylphosphonic acid (AMPA), an overview. J Exp Bot 65:4691–4703. https://doi.org/10.1093/jxb/eru269
Gomes GLGC, Carbonari CA, Velini ED et al (2015) Extraction and simultaneous determination of glyphosate, AMPA and compounds of the shikimic acid pathway in plants. Planta Daninha 33:295–304. https://doi.org/10.1590/0100-83582015000200015
Gomes MP, Cruz FVS, Borges FV et al (2016) Modulation of Dimorphandra wilsonii Rizz. seed germination through H2O2 production in response to Zn interference of the mitochondrial electron transport chain. Environ Exp Bot 128:51–58. https://doi.org/10.1016/j.envexpbot.2016.04.003
Guengerich FP, Martin MV, Sohl CD et al (2013) Measurement of cytochrome P450 and NADPH-cytochrome P450 reductase. Nat Protoc 4:345–349. https://doi.org/10.1038/nprot.2009.121.Measurement
Güngördü A (2013) Comparative toxicity of methidathion and glyphosate on early life stages of three amphibian species: Pelophylax ridibundus, Pseudepidalea viridis, and Xenopus laevis. Aquat Toxicol 140–141:220–228. https://doi.org/10.1016/j.aquatox.2013.06.012
Hénault-Ethier L, Lucotte M, Moingt M et al (2017) Herbaceous or Salix miyabeana ‘SX64’ narrow buffer strips as a means to minimize glyphosate and aminomethylphosphonic acid leaching from row crop fields. Sci Total Environ 598:1177–1186. https://doi.org/10.1016/j.scitotenv.2017.04.104
Hernández García E, Osnaya Brizuela N, Valenzuela Peraza A et al (2018) Biochemical and histological changes produced by sweeteners and cytarabine in the brain of young rats. Nutr Hosp 35:194. https://doi.org/10.20960/nh.1245
Hodges DM, Delong JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 1:604–611
Horth H, Blackmore K (2009) Survey of glyphosate and AMPA in groundwaters and surface waters in Europe. Final Report. WRc Ref: UC8073.02. Monsanto, Wiltshire, UK. https://docslib.org/doc/7120473/monsanto-survey-of-glyphosate-and-ampa-in-groundwaters-and-surface-waters-in-europe-final-report
Jaramillo Ramirez GI, Logan JG, Loza-Reyes E et al (2012) Repellents inhibit P450 enzymes in Stegomyia (Aedes) aegypti. PLoS One 7:e48698. https://doi.org/10.1371/journal.pone.0048698
Juliano SA (2009) Species interactions among larval mosquitoes: context dependence across habitat gradients. Annu Rev Entomol 54:37–56. https://doi.org/10.1146/annurev.ento.54.110807.090611
Knols BGJ (2021) Review of “mosquitoes of the world” by Richard C. Wilkerson, Yvonne-Marie Linton, and Daniel Strickman. Parasit Vectors 14:341. https://doi.org/10.1186/s13071-021-04848-6
Leong CT, D’Souza UJA, Iqbal M, Mustapha ZA (2013) Lipid peroxidation and decline in antioxidant status as one of the toxicity measures of diazinon in the testis. Redox Rep 18:155–164. https://doi.org/10.1179/1351000213Y.0000000054
Li N, Ragheb K, Lawler G et al (2003) Mitochondrial complex I inhibitor rotenone induces apoptosis through enhancing mitochondrial reactive oxygen species production. J Biol Chem 278:8516–8525. https://doi.org/10.1074/jbc.M210432200
Lumpkin WP, Stirek KR, Dyer LA (2020) Macrophyte diversity and complexity reduce larval mosquito abundance. J Med Entomol 57:1041–1048. https://doi.org/10.1093/jme/tjaa012
Mason TL, Poyton RO, Wharton DC, Schatz G (1973) Cytochrome c oxidase from Bakers’ yeast. J Biol Chem 248:1346–1354
Matozzo V, Munari M, Masiero L et al (2019) Ecotoxicological hazard of a mixture of glyphosate and aminomethylphosphonic acid to the mussel Mytilus galloprovincialis (Lamarck 1819). Sci Rep 9:14302. https://doi.org/10.1038/s41598-019-50607-0
Maxwell DM, Brecht KM, Koplovitz I, Sweeney RE (2006) Acetylcholinesterase inhibition: does it explain the toxicity of organophosphorus compounds? Arch Toxicol 80:756–760. https://doi.org/10.1007/s00204-006-0120-2
Mendes EJ, Malage L, Rocha DC, et al (2021) Isolated and combined effects of glyphosate and its by-product aminomethylphosphonic acid on the physiology and water remediation capacity of Salvinia molesta. J Hazard Mater 125694. https://doi.org/10.1016/j.jhazmat.2021.125694
Mendonça CFR (2018) Determinação de glifosato e AMPA nas águas superficiais da Bacia do Paraná 3. Dissertation, Universidade Estadual Paulista, 129 p
Metcalfe JL (1989) Biological water quality assessment of running waters based on macroinvertebrate communities: History and present status in Europe. Environ Pollut 60:101–139. https://doi.org/10.1016/0269-7491(89)90223-6
Murutaliev M, Guzov V, Walker F, Feyereisen R (2008) P450 reductase and cytochrome b5 interactions with cytochrome P450: Effects on house fly CYP6A1 catalysis. Insect Biochem Mol Biol 38:1008–1015. https://doi.org/10.1016/j.ibmb.2008.08.007
Nwani CD, Nagpure NS, Kumar R et al (2013) DNA damage and oxidative stress modulatory effects of glyphosate-based herbicide in freshwater fish, Channa punctatus. Environ Toxicol Pharmacol 36:539–547. https://doi.org/10.1016/j.etap.2013.06.001
Orr BK, Resh VH (1992) Influency of Myriophyllum aquaticum cover on Anopheles mosquito abundance oviposition, and larvla microhabitat. Oecologia 90:474–482
Overgaard HJ (2007) Effect of plant structure on oviposition behavior of Anopheles minimus s.l. J Vector Ecol 32:193–197. https://doi.org/10.3376/1081-1710(2007)32[193:EOPSOO]2.0.CO;2
Pascini T, Albeny D, Ramalho-Ortigão M et al (2011) Changes in the fat body during the post-embryonic development of the predator Toxorhynchites theobaldi (Dyar & Knab) (Diptera: Culicidae). Neotrop Entomol 40:456–461. https://doi.org/10.1590/S1519-566X2011000400008
Peixoto F (2005) Comparative effects of the Roundup and glyphosate on mitochondrial oxidative phosphorylation. Chemosphere 61:1115–1122. https://doi.org/10.1016/j.chemosphere.2005.03.044
Puértolas L, Damásio J, Barata C et al (2010) Evaluation of side-effects of glyphosate mediated control of giant reed (Arundo donax) on the structure and function of a nearby Mediterranean river ecosystem. Environ Res 110:556–564. https://doi.org/10.1016/j.envres.2010.05.004
Reisen WK, Meywe RP (1990) Attractiveness of selected oviposition substrates for gravid Culex tarsalis and Culex quinquefasciatus in California. J Am Mosq Control Assoc 6:244–250
Riaz MA, Poupardin R, Reynaud S et al (2009) Impact of glyphosate and benzo[a]pyrene on the tolerance of mosquito larvae to chemical insecticides. Role of detoxification genes in response to xenobiotics☆. Aquat Toxicol 93:61–69. https://doi.org/10.1016/j.aquatox.2009.03.005
Scribner EA, Battaglin WA, Gilliom RJ, Meyer MT (2007) Concentrations of glyphosate, its degradation product, aminomethylphosphonic acid, and glufosinate in ground- and surface-water, rainfall, and soil samples collected in the United States, 2001–06: U.S. Geological Survey Scientific Investigations Report 2007–5122, 111 p
Silva-Inacio CL, Paiva AAP de, Araújo JMG de, Ximenes M de FF de M (2020) Ecological relationships of Haemagogus spegazzinii (Diptera: Culicidae) in a semiarid area of Brazil. Rev Soc Bras Med Trop 53. https://doi.org/10.1590/0037-8682-0502-2020
Singer TP (1974) Determination of the activity of succinate, NADH, choline, and α-glycerophosphate dehydrogenases. In: Glick D (ed) Methods of biochememical analysis. John Wiley & Sons Inc, Hoboken, NJ, USA, pp 123–175
Smith DFQ, Camacho E, Thakur R et al (2021) Glyphosate inhibits melanization and increases susceptibility to infection in insects. PLOS Biol 19:e3001182. https://doi.org/10.1371/journal.pbio.3001182
Somlo M (1965) Induction des lactico-cytochrome c reductases (d-et l-) de la levure aerobie par les lactates (d- et l-). Biochim Biophys Acta - Gen Subj 97:183–201. https://doi.org/10.1016/0304-4165(65)90083-8
Souto RMG, Corbi JJ, Jacobucci GB (2019) Aquatic insects as bioindicators of heavy metals in sediments in Cerrado streams. Limnetica 38:575–586. https://doi.org/10.23818/limn.38.33
Uren Webster TM, Santos EM (2015) Global transcriptomic profiling demonstrates induction of oxidative stress and of compensatory cellular stress responses in brown trout exposed to glyphosate and Roundup. BMC Genomics 16:32. https://doi.org/10.1186/s12864-015-1254-5
Utzig LM, Lima RM, Gomes MF et al (2019) Ecotoxicity response of chlorpyrifos in Aedes aegypti larvae and <I>Lactuca sativa</i> seeds after UV/H2O2 and UVC oxidation. Ecotoxicol Environ Saf 169:449–456. https://doi.org/10.1016/j.ecoenv.2018.11.003
van Aardt WJ, du Plessis H (2018) The oxygen consumption rate of three Lepidopteran maize pest species measured by a clark polarographic oxygen electrode using an air-to-water respirometer chamber of novel design. African Entomol 26:550–554. https://doi.org/10.4001/003.026.0550
Varandas SG, Cortes RMV (2010) Evaluating macroinvertebrate biological metrics for ecological assessment of streams in northern Portugal. Environ Monit Assess 166:201–221. https://doi.org/10.1007/s10661-009-0996-4
WHO (1981) Instructions for determining the susceptibility or resistance of mosquito larvae to insecticides (No. WHO/VBC/81.807). World Health Organization
Worek F, Reiter G, Eyer P, Szinicz L (2002) Reactivation kinetics of acetylcholinesterase from different species inhibited by highly toxic organophosphates. Arch Toxicol 76:523–529. https://doi.org/10.1007/s00204-002-0375-1
Funding
This research was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior of Brazil (CAPES, Finance Code 001) and by the Universidade Federal do Paraná (UFPR). P.L.F received a scholarship from UFPR, and M.P.G and M.A.N-S received productivity fellowships from CNPq.
Author information
Authors and Affiliations
Contributions
Mariana Perez dos Santos, Patrícia Lawane de Freitas, Ana Carolina Felício Alves, Emily Nentwing de Barros, Rafael Shinji Akiyama Kitamura: methodology, investigation, formal analysis, preparing the original draft; Marcelo Pedrosa Gomes, Volnei Paulete and Mario Antônio Navarro-Silva: conceptualization, investigation, funding acquisition, preparing the review and editing. Marcelo Pedrosa Gomes: conceptualization, methodology, investigation, formal analysis, preparation of the original draft, and review and editing, supervision, funding acquisition, and project administration.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Committee of Ethics and Animal Experimentation from the Biological Science Sector of UFPR (CEAU 719).
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Bruno Nunes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gomes, M.P., dos Santos, M.P., de Freitas, P.L. et al. The aquatic macrophyte Salvinia molesta mitigates herbicides (glyphosate and aminomethylphosphonic acid) effects to aquatic invertebrates. Environ Sci Pollut Res 30, 12348–12361 (2023). https://doi.org/10.1007/s11356-022-23012-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-23012-w