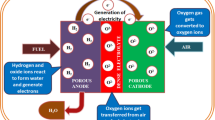
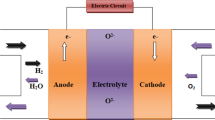
Abstract
Solid oxide fuel cells (SOFCs) are emerging as energy conversion devices for large-scale electrical power generation because of their high energy conversion efficiency, excellent ability to minimize air pollution, and high fuel flexibility. In this context, this critical review has focussed on the recent advancements in developing a suitable electrolyte for SOFCs which has been required for the commercialization of SOFC technology after emphasizing the literature from the prior studies. In particular, the significant developments in the field of solid oxide electrolytes for SOFCs, particularly zirconia- and ceria-based electrolytes, have been highlighted as important advancements toward the production of sustainable and clean energy. It has been reported that among various electrolyte materials, zirconia-based electrolytes have the potential to be utilized as the electrolyte in SOFC because of their high thermal stability, non-reducing nature, and high mechanical strength, along with acceptable oxygen ion conductivity. However, some studies have proved that the zirconia-based electrolytes are not suitable for low and intermediate-temperature working conditions because of their poor ionic conductivity to below 850 °C. On the other hand, ceria-based electrolytes are being investigated at a rapid pace as electrolytes for intermediate and low-temperature SOFCs due to their higher oxygen ion conductivity with good electrode compatibility, especially at lower temperatures than stabilized zirconia. In addition, the most emerging advancements in electrolyte materials have demonstrated that the intermediate temperature SOFCs as next-generation energy conversion technology have great potential for innumerable prospective applications.
Graphical abstract



Reproduced with permission from the ref. (Omar et al. 2006). Copyright 2006 Elsevier

Reproduced from the ref. (Zhigachev et al. 2021)

Reproduced from the ref. (Joh et al. 2017). Copyright 2017 ACS

Reproduced with permission from the ref. (Cho et al. 2019). Copyright 2019 Elsevier

Reproduced with permission from the ref. (Nie et al. 2021). Copyright 2021 Elsevier

Reproduced from the reference (Ji et al. 2015). Copyright 2015ACS

Copyright 2012 Elsevier

Reproduced with permission from the ref. (Gong et al. 2016). Copyright 2016 ACS

Reproduced from the reference (Gong et al. 2016). Copyright ACS 2020

Copyright RSC under common creative license
Similar content being viewed by others
Data availability
Not applicable.
References
Abdalla AM, Hossain S, Azad AT et al (2018) Nanomaterials for solid oxide fuel cells: a review. Renew Sustain Energy Rev 82:353–368. https://doi.org/10.1016/j.rser.2017.09.046
Accardo G, Dell’agli G, Frattini D et al (2017) Electrical behaviour and microstructural characterization of magnesia co-doped ScSZ nanopowders synthesized by urea co-precipitation. Chem Eng Trans 57:1345–1350. https://doi.org/10.3303/CET1757225
Afroze S, Karim AH, Cheok Q et al (2019) Latest development of double perovskite electrode materials for solid oxide fuel cells: a review. Front Energy 13:770–797. https://doi.org/10.1007/s11708-019-0651-x
Aguadero A, Fawcett L, Taub S et al (2012) Materials development for intermediate-temperature solid oxide electrochemical devices. J Mater Sci 47:3925–3948. https://doi.org/10.1007/s10853-011-6213-1
Andújar JM, Segura F (2009) Fuel cells: History and updating. A walk along two centuries. Renew Sustain Energy Rev 13:2309–2322. https://doi.org/10.1016/j.rser.2009.03.015
Anwar M, Muchtar A, Somalu MR (2016) Effects of various co-dopants and carbonates on the properties of doped ceria-based electrolytes: a brief review. Int J Appl Eng Res 11:9921–9928
Arachi Y, Asai T, Yamamoto O et al (2001) Electrical conductivity of ZrO[sub 2]-Sc[sub 2]O[sub 3] doped with HfO[sub 2], CeO[sub 2], and Ga[sub 2]O[sub 3]. J Electrochem Soc 148:A520. https://doi.org/10.1149/1.1366622
Arachi Y, Sakai H, Yamamoto O et al (1999) Electrical conductivity of the ZrO2-Ln2O3 (Ln = lanthanides) system. Solid State Ion 121:133–139. https://doi.org/10.1016/S0167-2738(98)00540-2
Arı M, Balcı M, Polat Y (2018) Synthesis and characterization of (Bi2O3)1–x−y−z(Gd2O3)x (Sm2O3)y(Eu2O3)z quaternary solid solutions for solid oxide fuel cell. Chinese J Phys 56:2958–2966. https://doi.org/10.1016/j.cjph.2018.10.001
Azim Jais A, Muhammed Ali SA, Anwar M et al (2017) Enhanced ionic conductivity of scandia-ceria-stabilized-zirconia (10Sc1CeSZ) electrolyte synthesized by the microwave-assisted glycine nitrate process. Ceram Int 43:8119–8125. https://doi.org/10.1016/j.ceramint.2017.03.135
Badwal SPS (1992) Zirconia-based solid electrolytes: microstructure, stability and ionic conductivity. Solid State Ion 52:23–32. https://doi.org/10.1016/0167-2738(92)90088-7
Badwal SPS, Ciacchi FT (2000) Oxygen-ion conducting electrolyte materials for solid oxide fuel cells. Ionics (kiel) 6:1–21. https://doi.org/10.1007/BF02375543
Badwal SPS, Ciacchi FT, Milosevic D (2000) Scandia-zirconia electrolytes for intermediate temperature solid oxide fuel cell operation. Solid State Ion 136–137:91–99. https://doi.org/10.1016/S0167-2738(00)00356-8
Bhattacharyya R, Omar S (2018) Electrical conductivity study of B-site Ga doped non-stoichiometric sodium bismuth titanate ceramics. J Alloys Compd 746:54–61. https://doi.org/10.1016/j.jallcom.2018.02.213
Cao J, Gong Z, Hou J et al (2015) Novel reduction-resistant Ba(Ce, Zr)1-xGdxO3-δ electron-blocking layer for Gd0.1Ce0.9O2-δ electrolyte in IT-SOFCs. Ceram Int 41:6824–6830. https://doi.org/10.1016/j.ceramint.2015.01.131
Cao J, Su C, Ji Y et al (2021) Recent advances and perspectives of fluorite and perovskite-based dual-ion conducting solid oxide fuel cells. J Energy Chem 57:406–427. https://doi.org/10.1016/j.jechem.2020.09.010
Chao CC, Hsu CM, Cui Y, Prinz FB (2011) Improved solid oxide fuel cell performance with nanostructured electrolytes. ACS Nano 5:5692–5696. https://doi.org/10.1021/nn201354p
Chen D, Xu Y, Tade MO, Shao Z (2017) General regulation of air flow distribution characteristics within planar solid oxide fuel cell stacks. ACS Energy Lett 2:319–326. https://doi.org/10.1021/acsenergylett.6b00548
Chen G, Wang Y, Sunarso J et al (2016a) A new scandium and niobium co-doped cobalt-free perovskite cathode for intermediate-temperature solid oxide fuel cells. Energy 95:137–143. https://doi.org/10.1016/j.energy.2015.11.061
Chen K, Li N, Ai N et al (2016b) Polarization-induced interface and Sr segregation of in situ assembled La0.6Sr0.4Co0.2Fe0.8O3-δ electrodes on Y2O3-ZrO2 electrolyte of solid oxide fuel cells. ACS Appl Mater Interfaces 8:31729–31737. https://doi.org/10.1021/acsami.6b11665
Chen Y, Gerdes K, Paredes Navia SA et al (2019) Conformal electrocatalytic surface nanoionics for accelerating higherature electrochemical reactions in solid oxide fuel cells. Nano Lett 19:8767–8773. https://doi.org/10.1021/acs.nanolett.9b03515
Cho GY, Lee YH, Yu W et al (2019) Optimization of Y2O3 dopant concentration of yttria stabilized zirconia thin film electrolyte prepared by plasma enhanced atomic layer deposition for high performance thin film solid oxide fuel cells. Energy 173:436–442. https://doi.org/10.1016/j.energy.2019.01.124
Dadashzadeh M, Ahmad A, Khan F (2016) Dispersion modelling and analysis of hydrogen fuel gas released in an enclosed area: a CFD-based approach. Fuel 184:192–201. https://doi.org/10.1016/j.fuel.2016.07.008
Danilov NA, Lyagaeva JG, Medvedev DA et al (2018) Transport properties of highly dense proton-conducting BaCe0.8–xZrxDy0.2O3–δ materials in low- and high-temperature ranges. Electrochim Acta 284:551–559. https://doi.org/10.1016/j.electacta.2018.07.179
Duan C, Tong J, Shang M, et al (2015) Readily processed protonic ceramic fuel cells with high performance at low temperatures. Science (80- ) 349:1321–1326. https://doi.org/10.1126/science.aab3987
Eigenbrodt BC, Pomfret MB, Steinhurst DA et al (2011) Direct, in situ optical studies of Ni-YSZ anodes in solid oxide fuel cells operating with methanol and methane. J Phys Chem C 115:2895–2903. https://doi.org/10.1021/jp109292r
Ermiş İ (2019) Fabrication of Bi0.95-xEr0.05MxO1.5-δ (M = Lu, Ho, and Gd) electrolyte for intermediate temperature solid oxide fuel cells. J Aust Ceram Soc 55:711–718. https://doi.org/10.1007/s41779-018-0282-4
Fini D, Badwal SPS, Giddey S et al (2018) Evaluation of Sc2O3–CeO2–ZrO2 electrolyte-based tubular fuel cells using activated charcoal and hydrogen fuels. Electrochim Acta 259:143–150. https://doi.org/10.1016/j.electacta.2017.10.140
Fleischhauer F, Terner M, Bermejo R et al (2015) Fracture toughness and strength distribution at room temperature of zirconia tapes used for electrolyte supported solid oxide fuel cells. J Power Sources 275:217–226. https://doi.org/10.1016/j.jpowsour.2014.10.083
Fu Q, Li Z, Wei W et al (2021) Performance degradation prediction of direct internal reforming solid oxide fuel cell due to Ni-particle coarsening in composite anode. Energy Convers Manag 233:113902. https://doi.org/10.1016/j.enconman.2021.113902
Fuentes RO, Baker RT (2009) Structural, morphological and electrical properties of Gd0.1Ce0.9O1.95 prepared by a citrate complexation method. J Power Sources 186:268–277. https://doi.org/10.1016/j.jpowsour.2008.09.119
Fujimori H, Yashima M, Kakihana M, Yoshimura M (1998) Structural changes of scandia-doped zirconia solid solutions: Rietveld analysis and Raman scattering. J Am Ceram Soc 81:2885–2893. https://doi.org/10.1111/j.1151-2916.1998.tb02710.x
Ghezel-Ayagh H, Jolly S, Patel D, Stauffer D (2013) Solid oxide fuel cell system utilizing syngas from coal gasifiers. Ind Eng Chem Res 52:3112–3120. https://doi.org/10.1021/ie300841m
Gong Z, Sun W, Shan D et al (2016) Tuning the thickness of Ba-containing “functional” Layer toward high-performance ceria-based solid oxide fuel cells. ACS Appl Mater Interfaces 8:10835–10840. https://doi.org/10.1021/acsami.6b01000
Grimes P (2000) Historical pathways for fuel cells. The new electric century. Proc Annu Batter Conf Appl Adv 2000-January:41–45. https://doi.org/10.1109/BCAA.2000.838369
Guan L, Le S, He S et al (2015) Densification behavior and space charge blocking effect of Bi2O3 and Gd2O3 Co-doped CeO2 as electrolyte for solid oxide fuel cells. Electrochim Acta 161:129–136. https://doi.org/10.1016/j.electacta.2015.02.090
Haering C, Roosen A, Schichl H, Schnöller M (2005) Degradation of the electrical conductivity in stabilised zirconia system Part II: scandia-stabilised zirconia. Solid State Ion 176:261–268. https://doi.org/10.1016/j.ssi.2004.07.039
Hanif MB, Rauf S, Motola M et al (2021) Recent progress of perovskite-based electrolyte materials for solid oxide fuel cells and performance optimizing strategies for energy storage applications. Mater Res Bull 146:111612. https://doi.org/10.1016/j.materresbull.2021.111612
Hao SJ, Wang C, Le LT et al (2017) Fabrication of nanoscale yttria stabilized zirconia for solid oxide fuel cell. Int J Hydrogen Energy 42:29949–29959. https://doi.org/10.1016/j.ijhydene.2017.08.143
Hassen D, El-Safty SA, Tsuchiya K et al (2016a) Longitudinal hierarchy Co 3 O 4 mesocrystals with high-dense exposure facets and anisotropic interfaces for direct-ethanol fuel cells. Sci Rep 6:1–12. https://doi.org/10.1038/srep24330
Hassen D, Shenashen MA, El-Safty SA et al (2016b) Nitrogen-doped carbon-embedded TiO2 nanofibers as promising oxygen reduction reaction electrocatalysts. J Power Sources 330:292–303. https://doi.org/10.1016/j.jpowsour.2016.08.140
He S, Zhang Q, Maurizio G et al (2018) In Situ Formation of Er0.4Bi1.6O3 Protective layer at cobaltite cathode/Y2O3-ZrO2 electrolyte interface under solid oxide fuel cell operation conditions. ACS Appl Mater Interfaces 10:40549–40559. https://doi.org/10.1021/acsami.8b14026
He W, Wu X, Yang G et al (2017) BaCo0.7Fe0.22Y0.08O3-δ as an active oxygen reduction electrocatalyst for low-temperature solid oxide fuel cells below 600 °c. ACS Energy Lett 2:301–305. https://doi.org/10.1021/acsenergylett.6b00617
Hibino T, Mizutani K, Yajima T, Iwahara H (1992) Evaluation of proton conductivity in SrCeO3, BaCeO3, CaZrO3 and SrZrO3 by temperature programmed desorption method. Solid State Ion 57:303–306. https://doi.org/10.1016/0167-2738(92)90162-I
Hou X, Zhao K, Marina OA et al (2019) NiMo-ceria-zirconia-based anode for solid oxide fuel cells operating on gasoline surrogate. Appl Catal B Environ 242:31–39. https://doi.org/10.1016/j.apcatb.2018.09.095
Hou Y, Wang L, Bian L et al (2021) Excellent electrochemical performance of La0.3Sr0.7Fe0.9Ti0.1O3-δas a symmetric electrode for solid oxide cells. ACS Appl Mater Interfaces 13:22381–22390. https://doi.org/10.1021/acsami.1c02856
Hua B, Li M, Zhang YQ et al (2016) Facile synthesis of highly active and robust Ni-Mo bimetallic electrocatalyst for hydrocarbon oxidation in solid oxide fuel cells. ACS Energy Lett 1:225–230. https://doi.org/10.1021/acsenergylett.6b00109
Huang C, Zhao G, Song Y et al (2022) Preparation of novel biodegradable cellulose nanocrystal proton exchange membranes for direct methanol fuel-cell applications. ACS Sustain Chem Eng 10:5559–5568. https://doi.org/10.1021/acssuschemeng.2c00221
Huang H, Nakamura M, Su P et al (2007) High-performance ultrathin solid oxide fuel cells for low-temperature operation. J Electrochem Soc 154:B20. https://doi.org/10.1149/1.2372592
Huang R, Antono E, Meredig B et al (2021) High-throughput characterization of Lu-doped zirconia. Solid State Ion 368:115698. https://doi.org/10.1016/j.ssi.2021.115698
Hwang KJ, Jang M, Kim MK et al (2021) Effective buffer layer thickness of La-doped CeO2 for high durability and performance on La0.9Sr0.1Ga0.8Mg0.2O3-δ electrolyte supported type solid oxide fuel cells. J Eur Ceram Soc 41:2674–2681. https://doi.org/10.1016/j.jeurceramsoc.2020.11.036
Irshad M, Siraj K, Raza R, et al (2016) A brief description of high temperature solid oxide fuel cell’s operation, materials, design, fabrication technologies and performance. Appl Sci 6. https://doi.org/10.3390/app6030075
Ishihara T, Yan J, Shinagawa M, Matsumoto H (2006) Ni-Fe bimetallic anode as an active anode for intermediate temperature SOFC using LaGaO3 based electrolyte film. Electrochim Acta 52:1645–1650. https://doi.org/10.1016/j.electacta.2006.03.103
Istomin SY, Lyskov NV, Mazo GN, Antipov EV (2021) Electrode materials based on complex d-metal oxides for symmetrical solid oxide fuel cells. Russ Chem Rev 90:644–676. https://doi.org/10.1070/rcr4979
Ito S, Mori T, Suzuki A et al (2019) Design of active site at heterointerface between brownmillerite type oxide promoter and fluorite cubic ZrO2 in anode of intermediate temperature SOFCs. ACS Appl Energy Mater 2:5183–5197. https://doi.org/10.1021/acsaem.9b00864
Iwahara H, Esaka T, Uchida H, Maeda N (1981) Proton conduction in sintered oxides and its application to steam electrolysis for hydrogen production. Solid State Ion 3–4:359–363. https://doi.org/10.1016/0167-2738(81)90113-2
Jamil A, Rafiq S, Iqbal T et al (2022) Current status and future perspectives of proton exchange membranes for hydrogen fuel cells. Chemosphere 303:135204. https://doi.org/10.1016/j.chemosphere.2022.135204
Jang I, Lee H, Tamarany R et al (2020) Tailoring the ratio of a-site cations in Pr1- xNd xBaCo1.6Fe0.4O5+δto promote the higher oxygen reduction reaction activity for low-temperature solid oxide fuel cells. Chem Mater 32:3841–3849. https://doi.org/10.1021/acs.chemmater.9b05378
Ji S, Cho GY, Yu W et al (2015) Plasma-enhanced atomic layer deposition of nanoscale yttria-stabilized zirconia electrolyte for solid oxide fuel cells with porous substrate. ACS Appl Mater Interfaces 7:2998–3002. https://doi.org/10.1021/am508710s
Jiang N, Wachsman ED, Jung SH (2002) A higher conductivity Bi2O3-based electrolyte. Solid State Ion 150:347–353. https://doi.org/10.1016/S0167-2738(02)00291-6
Joh DW, Park JH, Kim D et al (2017) Functionally graded bismuth oxide/zirconia bilayer electrolytes for high-performance intermediate-temperature solid oxide fuel cells (IT-SOFCs). ACS Appl Mater Interfaces 9:8443–8449. https://doi.org/10.1021/acsami.6b16660
Kang H, Kim E, Jung SP (2017) Influence of flowrates to a reverse electro-dialysis (RED) stack on performance and electrochemistry of a microbial reverse electrodialysis cell (MRC). Int J Hydrog Energy 42:27685–27692. https://doi.org/10.1016/j.ijhydene.2017.06.187
Kerman K, Ramanathan S (2014) Complex oxide nanomembranes for energy conversion and storage: a review. J Mater Res 29:320–337. https://doi.org/10.1557/jmr.2013.301
Khan MZ, Mehran MT, Song RH et al (2018) A simplified approach to predict performance degradation of a solid oxide fuel cell anode. J Power Sources 391:94–105. https://doi.org/10.1016/j.jpowsour.2018.04.080
Khan MZ, Song RH, Hussain A et al (2020) Effect of applied current density on the degradation behavior of anode-supported flat-tubular solid oxide fuel cells. J Eur Ceram Soc 40:1407–1417. https://doi.org/10.1016/j.jeurceramsoc.2019.11.017
Kharton VV, Marques FMB, Atkinson A (2004) Transport properties of solid oxide electrolyte ceramics: a brief review. Solid State Ion 174:135–149. https://doi.org/10.1016/j.ssi.2004.06.015
Kumar A, Jaiswal A, Sanbui M, Omar S (2016) Scandia stabilized zirconia-ceria solid electrolyte (xSc1CeSZ, 5 < x < 11) for IT-SOFCs: structure and conductivity studies. Scr Mater 121:10–13. https://doi.org/10.1016/j.scriptamat.2016.04.023
Kumar A, Jaiswal A, Sanbui M, Omar S (2017) Oxygen-ion conduction in scandia-stabilized zirconia-ceria solid electrolyte (xSc2O3–1CeO2–(99–x)ZrO2, 5 ≤ x ≤ 11). J Am Ceram Soc 100:659–668. https://doi.org/10.1111/jace.14595
Kumar CNS, Bauri R, Reddy GS (2020) Phase stability and conductivity of rare earth co-doped nanocrystalline zirconia electrolytes for solid oxide fuel cells. J Alloys Compd 833:155100. https://doi.org/10.1016/j.jallcom.2020.155100
Lee DS, Kim WS, Choi SH et al (2005) Characterization of ZrO2 co-doped with Sc2O 3 and CeO2 electrolyte for the application of intermediate temperature SOFCs. Solid State Ion 176:33–39. https://doi.org/10.1016/j.ssi.2004.07.013
Lee JG, Park JH, Shul YG (2014) Tailoring gadolinium-doped ceria-based solid oxide fuel cells to achieve 2Wcm-2 at 550 °c. Nat Commun 5. https://doi.org/10.1038/ncomms5045
Lee S, Zhang W, Khatkhatay F et al (2015) Ionic conductivity increased by two orders of magnitude in micrometer-thick vertical yttria-stabilized ZrO2 nanocomposite films. Nano Lett 15:7362–7369. https://doi.org/10.1021/acs.nanolett.5b02726
Li M, Wang Y, Wang Y et al (2014) Bismuth doped lanthanum ferrite perovskites as novel cathodes for intermediate-temperature solid oxide fuel cells. ACS Appl Mater Interfaces 6:11286–11294. https://doi.org/10.1021/am5017045
Li R, Yang J, Yan D et al (2021) Performance of Al2O3 particle reinforced glass-based seals in planar solid oxide fuel cells. Ceram Int 47:7504–7510. https://doi.org/10.1016/j.ceramint.2020.10.256
Liang YC, Chou YH (2020) Improved photoelectrode performance of chemical solution-derived Bi2O3crystals: via manipulation of crystal characterization. RSC Adv 10:45042–45058. https://doi.org/10.1039/d0ra08746g
Lim HT, Virkar AV (2009) Measurement of oxygen chemical potential in Gd2O3-doped ceria-Y2O3-stabilized zirconia bi-layer electrolyte, anode-supported solid oxide fuel cells. J Power Sources 192:267–278. https://doi.org/10.1016/j.jpowsour.2009.03.035
Liu M, Ding D, Bai Y et al (2012) An efficient SOFC based on samaria-doped ceria (SDC) electrolyte. J Electrochem Soc 159:B661–B665. https://doi.org/10.1149/2.032206jes
Liu T, Ren C, Fang S et al (2014) Microstructure tailoring of the nickel oxide-yttria-stabilized zirconia hollow fibers toward high-performance microtubular solid oxide fuel cells. ACS Appl Mater Interfaces 6:18853–18860. https://doi.org/10.1021/am5046907
Lu Y, Cai Y, Souamy L et al (2018) Solid oxide fuel cell technology for sustainable development in China: an over-view. Int J Hydrog Energy 43:12870–12891. https://doi.org/10.1016/j.ijhydene.2018.05.008
Lucia U (2014) Overview on fuel cells. Renew Sustain Energy Rev 30:164–169. https://doi.org/10.1016/j.rser.2013.09.025
Lunca Popa P, Sønderby S, Kerdsongpanya S et al (2017) Structural, morphological, and optical properties of Bi2O3 thin films grown by reactive sputtering. Thin Solid Films 624:41–48. https://doi.org/10.1016/j.tsf.2017.01.013
Ma G, Matsumoto H, Iwahara H (1999) Ionic conduction and nonstoichiometry in non-doped BaxCeO3-α. Solid State Ion 122:237–247. https://doi.org/10.1016/S0167-2738(99)00074-0
Mahato N, Banerjee A, Gupta A et al (2015) Progress in material selection for solid oxide fuel cell technology: a review. Prog Mater Sci 72:141–337. https://doi.org/10.1016/j.pmatsci.2015.01.001
Malavasi L, Fisher CAJ, Islam MS (2010) Oxide-ion and proton conducting electrolyte materials for clean energy applications: structural and mechanistic features. Chem Soc Rev 39:4370–4387. https://doi.org/10.1039/b915141a
Masciandaro S, Torrell M, Leone P, Tarancón A (2019) Three-dimensional printed yttria-stabilized zirconia self-supported electrolytes for solid oxide fuel cell applications. J Eur Ceram Soc 39:9–16. https://doi.org/10.1016/j.jeurceramsoc.2017.11.033
Masina SM, Billing C, Erasmus RM, Billing DG (2021) Insights on the phase transitions, stability and conductivity in the Bi2O3-WO3 system. J Electroceramics 1. https://doi.org/10.1007/s10832-021-00243-w
Matsuda M, Hosomi T, Murata K et al (2007) Fabrication of bilayered YSZ/SDC electrolyte film by electrophoretic deposition for reduced-temperature operating anode-supported SOFC. J Power Sources 165:102–107. https://doi.org/10.1016/j.jpowsour.2006.11.087
Medvedev D, Murashkina A, Pikalova E et al (2014) BaCeO3: Materials development, properties and application. Prog Mater Sci 60:72–129. https://doi.org/10.1016/j.pmatsci.2013.08.001
Morales M, Pesce A, Slodczyk A et al (2018) Enhanced performance of gadolinia-doped ceria diffusion barrier layers fabricated by pulsed laser deposition for large-area solid oxide fuel cells. ACS Appl Energy Mater 1:1955–1964. https://doi.org/10.1021/acsaem.8b00039
Morikawa H, Kikuchi H, Saito N (2009) Development and advances of a V-flow FC stack for FCX clarity. SAE Tech Pap 2:955–959. https://doi.org/10.4271/2009-01-1010
Muñoz-García AB, Ritzmann AM, Pavone M et al (2014) Oxygen transport in perovskite-type solid oxide fuel cell materials: insights from quantum mechanics. Acc Chem Res 47:3340–3348. https://doi.org/10.1021/ar4003174
Ng CK, Ramesh S, Tan CY et al (2016) Microwave sintering of ceria-doped scandia stabilized zirconia as electrolyte for solid oxide fuel cell. Int J Hydrog Energy 41:14184–14190. https://doi.org/10.1016/j.ijhydene.2016.06.146
Ng KH, Rahman HA, Somalu MR (2019) Review: enhancement of composite anode materials for low-temperature solid oxide fuels. Int J Hydrog Energy 44:30692–30704. https://doi.org/10.1016/j.ijhydene.2018.11.137
Nie J, Zheng D, Ganesh KS et al (2021) Efficient strategy to boost the electrochemical performance of yttrium stabilized zirconia electrolyte solid oxide fuel cell for low-temperature applications. Ceram Int 47:3462–3472. https://doi.org/10.1016/j.ceramint.2020.09.190
Nielsen J, Persson ÅH, Muhl TT, Brodersen K (2018) Towards high power density metal supported solid oxide fuel cell for mobile applications. J Electrochem Soc 165:F90–F96. https://doi.org/10.1149/2.0741802jes
Nikonov AV, Kaygorodov AS, Kuterbekov KA, Bekmyrza KZ (2017) Microhardness and fracture toughness of ZrO2–Sc2O3 solid electrolyte, doped with rare-earth and transition metals. Inorg Mater 53:937–943. https://doi.org/10.1134/S002016851709014X
Oh EO, Whang CM, Lee YR et al (2012) Extremely thin bilayer electrolyte for solid oxide fuel cells (SOFCs) fabricated by chemical solution deposition (CSD). Adv Mater 24:3373–3377. https://doi.org/10.1002/adma.201200505
Omar S, Bin Najib W, Chen W, Bonanos N (2012) Electrical conductivity of 10 mol% Sc 2 O 3–1 mol% M 2 O 3- ZrO 2 ceramics. J Am Ceram Soc 95:1965–1972. https://doi.org/10.1111/j.1551-2916.2012.05126.x
Omar S, Wachsman ED, Nino JC (2008) Higher conductivity Sm3+ and Nd3+ co-doped ceria-based electrolyte materials. Solid State Ion 178:1890–1897. https://doi.org/10.1016/j.ssi.2007.12.069
Omar S, Wachsman ED, Nino JC (2006) A co-doping approach towards enhanced ionic conductivity in fluorite-based electrolytes. Solid State Ion 177:3199–3203. https://doi.org/10.1016/j.ssi.2006.08.014
Orozco-Hernández G, Olaya-Flórez J, Pineda-Vargas C et al (2020) Structural, chemical and electrochemical studies of bismuth oxide thin films growth via unbalanced magnetron sputtering. Surfaces and Interfaces 21:100627. https://doi.org/10.1016/j.surfin.2020.100627
Pan KJ, Hussain AM, Huang YL et al (2019) Evolution of solid oxide fuel cells via fast interfacial oxygen crossover. ACS Appl Energy Mater 2:4069–4074. https://doi.org/10.1021/acsaem.9b00185
Parbin A, Rafiuddin, (2022) The fabrication of a highly conductive ceria-embedded gadolinium-stabilized bismuth oxide nanocomposite solid electrolyte for low-temperature solid oxide fuel cells. Mater Adv 3:3316–3325. https://doi.org/10.1039/d1ma01254a
Park CO, Akbar SA, Weppner W (2003) Ceramic electrolytes and electrochemical sensors. J Mater Sci 38:4639–4660. https://doi.org/10.1023/A:1027454414224
Perry ML, Fuller TF (2002) A historical perspective of fuel cell technology in the 20th century. J Electrochem Soc 149:S59. https://doi.org/10.1149/1.1488651
Politova TI, Irvine JTS (2004) Investigation of scandia-yttria-zirconia system as an electrolyte material for intermediate temperature fuel cells - influence of yttria content in system (Y2O3)x(Sc2O3) (11–x)(ZrO2)89. Solid State Ion 168:153–165. https://doi.org/10.1016/j.ssi.2004.02.007
Price M, Dong J, Gu X et al (2005) Formation of YSZ-SDC solid solution in a nanocrystalline heterophase system and its effect on the electrical conductivity. J Am Ceram Soc 88:1812–1818. https://doi.org/10.1111/j.1551-2916.2005.00389.x
Qian J, Zhu Z, Dang J et al (2013) Improved performance of solid oxide fuel cell with pulsed laser deposited thin film ceria-zirconia bilayer electrolytes on modified anode substrate. Electrochim Acta 92:243–247. https://doi.org/10.1016/j.electacta.2013.01.017
Qiao J, Chen H, Wang Z et al (2020) Enhancing the catalytic activity of Y0.08Sr0.92TiO3-δAnodes through in Situ Cu exsolution for direct carbon solid oxide fuel cells. Ind Eng Chem Res 59:13105–13112. https://doi.org/10.1021/acs.iecr.0c02203
Qiao Z, Xia C, Cai Y et al (2018) Electrochemical and electrical properties of doped CeO2-ZnO composite for low-temperature solid oxide fuel cell applications. J Power Sources 392:33–40. https://doi.org/10.1016/j.jpowsour.2018.04.096
Rajendran S, Thangavel NK, Alkatie S et al (2021) Y, Gd, and Pr tri-doped perovskite-type proton conducting electrolytes with improved sinterability and chemical stability. J Alloys Compd 870:159431. https://doi.org/10.1016/j.jallcom.2021.159431
Rashid NLRM, Samat AA, Jais AA et al (2019) Review on zirconate-cerate-based electrolytes for proton-conducting solid oxide fuel cell. Ceram Int 45:6605–6615. https://doi.org/10.1016/j.ceramint.2019.01.045
Ren R, Wang Z, Meng X et al (2020a) Boosting the electrochemical performance of Fe-based layered double perovskite cathodes by Zn2+ doping for solid oxide fuel cells. ACS Appl Mater Interfaces 12:23959–23967. https://doi.org/10.1021/acsami.0c04605
Ren R, Wang Z, Meng X et al (2020b) Tailoring the oxygen vacancy to achieve fast intrinsic proton transport in a perovskite cathode for protonic ceramic fuel cells. ACS Appl Energy Mater 3:4914–4922. https://doi.org/10.1021/acsaem.0c00486
Riegraf M, Amaya-Dueñas DM, Sata N et al (2021) Performance and limitations of nickel-doped chromite anodes in electrolyte-supported solid oxide fuel cells. Chemsuschem 14:2401–2413. https://doi.org/10.1002/cssc.202100330
Ruh R, Garrett HJ, Domagala RF, Patel VA (1977) The system zirconia-scandia. J Am Ceram Soc 60:399–403. https://doi.org/10.1111/j.1151-2916.1977.tb15521.x
Sacanell J, Hernández Sánchez J, Rubio López AE et al (2017) Oxygen reduction mechanisms in nanostructured La0.8Sr0.2MnO3 cathodes for solid oxide fuel cells. J Phys Chem C 121:6533–6539. https://doi.org/10.1021/acs.jpcc.7b00627
Sandhu NK, Hanifi AR, Woldnik A et al (2016) Electrochemical performance of a short tubular solid oxide fuel cell stack at intermediate temperatures. Appl Energy 183:358–368. https://doi.org/10.1016/j.apenergy.2016.08.179
Shah MAKY, Rauf S, Mushtaq N et al (2020) Semiconductor Fe-doped SrTiO3-δ perovskite electrolyte for low-temperature solid oxide fuel cell (LT-SOFC) operating below 520 °C. Int J Hydrog Energy 45:14470–14479. https://doi.org/10.1016/j.ijhydene.2020.03.147
Shim JH, Chao CC, Huango H, Prinz FB (2007) Atomic layer deposition of yttria-stabilized zirconia for solid oxide fuel cells. Chem Mater 19:3850–3854. https://doi.org/10.1021/cm070913t
Shimada H, Yamaguchi T, Sumi H et al (2017) Extremely fine structured cathode for solid oxide fuel cells using Sr-doped LaMnO3 and Y2O3-stabilized ZrO2 nano-composite powder synthesized by spray pyrolysis. J Power Sources 341:280–284. https://doi.org/10.1016/j.jpowsour.2016.12.002
Shimada H, Yamaguchi T, Sumi H et al (2019) A key for achieving higher open-circuit voltage in protonic ceramic fuel cells: lowering interfacial electrode polarization. ACS Appl Energy Mater 2:587–597. https://doi.org/10.1021/acsaem.8b01617
Shin JS, Park H, Park K et al (2021) Activity of layered swedenborgite structured Y0.8Er0.2BaCo3.2Ga0.8O7+: δfor oxygen electrode reactions in at intermediate temperature reversible ceramic cells. J Mater Chem A 9:607–621. https://doi.org/10.1039/d0ta11000k
Shin JW, Oh S, Lee S et al (2019) Ultrathin atomic layer-deposited CeO2 overlayer for high-performance fuel cell electrodes. ACS Appl Mater Interfaces 11:46651–46657. https://doi.org/10.1021/acsami.9b10572
Singh R, Chavan SB (2019) Processing and Properties of scandia-doped zirconia electrolyte for intermediate temperature SOFC. ECS Trans 7:2207–2212. https://doi.org/10.1149/1.2729336
Smeacetto F, Salvo M, Ajitdoss LC et al (2010) Yttria-stabilized zirconia thin film electrolyte produced by RF sputtering for solid oxide fuel cell applications. Mater Lett 64:2450–2453. https://doi.org/10.1016/j.matlet.2010.08.016
Son S, Koo B, Chai H et al (2021) Comparison of hydrogen production and system performance in a microbial electrolysis cell containing cathodes made of non-platinum catalysts and binders. J Water Process Eng 40:101844. https://doi.org/10.1016/j.jwpe.2020.101844
Spirin A, Ivanov V, Nikonov A et al (2012) Scandia-stabilized zirconia doped with yttria: synthesis, properties, and ageing behavior. Solid State Ion 225:448–452. https://doi.org/10.1016/j.ssi.2012.02.022
Steele BCH (2001) Material science and engineering: the enabling technology for the commercialisation of fuel cell systems. J Mater Sci 36:1053–1068. https://doi.org/10.1023/A:1004853019349
Sudarsan P, Krishnamoorthy SB (2018) Grain boundary scavenging through reactive sintering of strontium and iron in samarium doped ceria electrolyte for ITSOFC applications. Mater Res Bull 100:446–457. https://doi.org/10.1016/j.materresbull.2017.12.047
Sun W, Liu W (2012) A novel ceria-based solid oxide fuel cell free from internal short circuit. J Power Sources 217:114–119. https://doi.org/10.1016/j.jpowsour.2012.05.065
Sun W, Shi Z, Qian J et al (2014) In-situ formed Ce0.8Sm0.2O2-δat Ba(Ce, Zr)1–x(Sm, Y)xO3-δ core/shell electron-blocking layer towards Ce0.8Sm0.2O2-δ-based solid oxide fuel cells with high open cir. Nano Energy 8:305–311. https://doi.org/10.1016/j.nanoen.2014.06.018
Sundmacher K (2010) Fuel cell engineering: toward the design of efficient electrochemical power plants. Ind Eng Chem Res 49:10159–10182. https://doi.org/10.1021/ie100902t
Takahashi T, Esaka T, Iwahara H (1977) Conduction in Bi2O3-based oxide ion conductors under low oxygen pressure. I. Current blackening of the Bi2O3-Y2O3 electrolyte. J Appl Electrochem 7:299–302. https://doi.org/10.1007/BF01059169
Tran VHH, Kim E, Jung SP (2022) Anode biofilm maturation time, stable cell performance time, and time-course electrochemistry in a single-chamber microbial fuel cell with a brush-anode. J Ind Eng Chem 106:269–278. https://doi.org/10.1016/j.jiec.2021.11.001
Virkar AV (1991) Theoretical analysis of solid oxide fuel cells with two-layer, composite electrolytes: electrolyte stability. J Electrochem Soc 138:1481–1487. https://doi.org/10.1149/1.2085811
Vostakola MF, Horri BA (2021) Progress in material development for low-temperature solid oxide fuel cells: a review. Energies 14:1–55. https://doi.org/10.3390/en14051280
Wang F, Lyu Y, Chu D et al (2019) The electrolyte materials for SOFCs of low-intermediate temperature: review. Mater Sci Technol (united Kingdom) 35:1551–1562. https://doi.org/10.1080/02670836.2019.1639008
Wang Q, Fan H, Xiao Y, Zhang Y (2022) Applications and recent advances of rare earth in solid oxide fuel cells. J Rare Earths. https://doi.org/10.1016/j.jre.2021.09.003
Wang Z, Cheng M, Bi Z et al (2005) Structure and impedance of ZrO2 doped with Sc2O 3 and CeO2. Mater Lett 59:2579–2582. https://doi.org/10.1016/j.matlet.2004.07.065
Wei B, Schroeder M, Martin M (2018) Surface cation segregation and chromium deposition on the double-perovskite oxide PrBaCo2O5+δ. ACS Appl Mater Interfaces 10:8621–8629. https://doi.org/10.1021/acsami.7b17881
Wei CC, Li K (2008) Yttria-stabilized zirconia (YSZ)-based hollow fiber solid oxide fuel cells. Ind Eng Chem Res 47:1506–1512. https://doi.org/10.1021/ie070960v
Wei L, Zhang J, Yu F et al (2019) A novel fabrication of yttria-stabilized-zirconia dense electrolyte for solid oxide fuel cells by 3D printing technique. Int J Hydrogen Energy 44:6182–6191. https://doi.org/10.1016/j.ijhydene.2019.01.071
Wei T, Zhang LA, Chen Y et al (2017) Promising proton conductor for intermediate-temperature fuel cells: Li13.9Sr0.1Zn(GeO4)4. Chem Mater 29:1490–1495. https://doi.org/10.1021/acs.chemmater.6b03471
Wilberforce T, Alaswad A, Palumbo A et al (2016) Advances in stationary and portable fuel cell applications. Int J Hydrogen Energy 41:16509–16522. https://doi.org/10.1016/j.ijhydene.2016.02.057
Williams KR (1994) Francis Thomas Bacon, 21 December 1904–24 May 1992. Biogr Mem Fellows R Soc 39:1–18. https://doi.org/10.1098/rsbm.1994.0001
Xia C, Cai Y, Ma Y et al (2016) Natural mineral-based solid oxide fuel cell with heterogeneous nanocomposite derived from hematite and rare-earth minerals. ACS Appl Mater Interfaces 8:20748–20755. https://doi.org/10.1021/acsami.6b05694
Xia C, Liu M (2001) Low-temperature SOFCs based on Gd0.1Ce0.9O1.95 fabricated by dry pressing. Solid State Ion 144:249–255. https://doi.org/10.1016/S0167-2738(01)00980-8
Xia C, Qiao Z, Shen L et al (2018) Semiconductor electrolyte for low-operating-temperature solid oxide fuel cell: Li-doped ZnO. Int J Hydrog Energy 43:12825–12834. https://doi.org/10.1016/j.ijhydene.2018.04.121
Xu M, Yu J, Song Y et al (2020) Advances in Ceramic thin films fabricated by pulsed laser deposition for intermediate-temperature solid oxide fuel cells. Energy Fuels 34:10568–10582. https://doi.org/10.1021/acs.energyfuels.0c02338
Yahiro H, Ohuchi T, Eguchi K, Arai H (1988) Electrical properties and microstructure in the system ceria-alkaline earth oxide. J Mater Sci 23:1036–1041. https://doi.org/10.1007/BF01154008
Yamamoto O, Arati Y, Takeda Y et al (1995) Electrical conductivity of stabilized zirconia with ytterbia and scandia. Solid State Ion 79:137–142. https://doi.org/10.1016/0167-2738(95)00044-7
Yang G, Su C, Shi H et al (2020) Toward reducing the operation temperature of solid oxide fuel cells: our past 15 years of efforts in cathode development. Energy Fuels 34:15169–15194. https://doi.org/10.1021/acs.energyfuels.0c01887
Yang J, Molouk AFS, Okanishi T et al (2015) Electrochemical and catalytic properties of Ni/BaCe0.75Y0.25O3-δ anode for direct ammonia-fueled solid oxide fuel cells. ACS Appl Mater Interfaces 7:7406–7412. https://doi.org/10.1021/acsami.5b01048
Yasuda K, Uemura K, Shiota T (2012) Sintering and mechanical properties of gadolinium-doped ceria ceramics. J Phys Conf Ser 339:2–6. https://doi.org/10.1088/1742-6596/339/1/012006
Yoshida T, Kojima K (2015) Toyota MIRAI fuel cell vehicle and progress toward a future hydrogen society. Electrochem Soc Interface 24:45–49. https://doi.org/10.1149/2.F03152if
Zahid M, Savla N, Pandit S et al (2022) Microbial desalination cell: desalination through conserving energy. Desalination 521:115381. https://doi.org/10.1016/j.desal.2021.115381
Zhang W, Wang H, Guan K et al (2019) La0.6Sr0.4Co0.2Fe0.8O3-δ/CeO2 heterostructured composite nanofibers as a highly active and robust cathode catalyst for solid oxide fuel cells. ACS Appl Mater Interfaces 11:26830–26841. https://doi.org/10.1021/acsami.9b06668
Zhang W, Wang H, Guan K et al (2020) Enhanced anode performance and coking resistance by in situ exsolved multiple-twinned Co-Fe nanoparticles for solid oxide Fuel Cells. ACS Appl Mater Interfaces 12:461–473. https://doi.org/10.1021/acsami.9b14655
Zhang X, Robertson M, Deĉes-Petit C et al (2007) Internal shorting and fuel loss of a low temperature solid oxide fuel cell with SDC electrolyte. J Power Sources 164:668–677. https://doi.org/10.1016/j.jpowsour.2006.10.087
Zhang X, Robertson M, Yick S et al (2006) Sm0.5Sr0.5CoO3 + Sm0.2Ce0.8O1.9 composite cathode for cermet supported thin Sm0.2Ce0.8O1.9 electrolyte SOFC operating below 600 °C. J Power Sources 160:1211–1216. https://doi.org/10.1016/j.jpowsour.2006.03.035
Zhao J, Xu X, Zhou W et al (2017) Proton-conducting la-doped ceria-based internal reforming layer for direct methane solid oxide fuel cells. ACS Appl Mater Interfaces 9:33758–33765. https://doi.org/10.1021/acsami.7b07938
Zhigachev AO, Rodaev VV, Zhigacheva DV et al (2021) Doping of scandia-stabilized zirconia electrolytes for intermediate-temperature solid oxide fuel cell: a review. Ceram Int 47:32490–32504. https://doi.org/10.1016/j.ceramint.2021.08.285
Zhuiykov S (2000) An investigation of conductivity, microstructure and stability of HfO2-ZrO2-Y2O3-Al2O 3 electrolyte compositions for high-temperature oxygen measurement. J Eur Ceram Soc 20:967–976. https://doi.org/10.1016/s0955-2219(99)00233-2
Acknowledgements
We acknowledge Mahatma Gandhi Central Library, IIT Roorkee, for providing all literature.
Funding
This work was financially supported by the Ministry of Human Resources Development, India, and IIT Roorkee, India.
Author information
Authors and Affiliations
Contributions
Tushar Kanti Maiti: conceptualization, writing, reviewing, editing, visualization, data analysis. Jagannath Majhi: Editing, visualization. Subrata Kumar Maiti: conceptualization, reviewing. Jitendra Singh: conceptualization, editing. Prakhar Dixit: editing, visualization. Tushita Rohilla: reviewing, editing. Samaresh Ghosh: conceptualization, data analysis. Sakchi Bhushan: reviewing, visualization. Sujay Chattopadhyay: conceptualization, supervision.
Corresponding author
Ethics declarations
Ethical approval
We have obtained all ethical approval.
Consent to participate
All authors voluntarily agreed to participate in this study.
Consent for publication
All authors have agreed to publish.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maiti, T.K., Majhi, J., Maiti, S.K. et al. Zirconia- and ceria-based electrolytes for fuel cell applications: critical advancements toward sustainable and clean energy production. Environ Sci Pollut Res 29, 64489–64512 (2022). https://doi.org/10.1007/s11356-022-22087-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-22087-9