Abstract
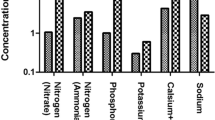
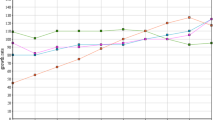
The world needs to adapt to recycling and reusing water due to limited resources. So, decision-makers and policy leaders should use sustainable practices to improve protection and pollution remediation. Aluminum sulfate is used for surface water treatment, which leads to waste sludge being disposed into water bodies, causing environmental pollution. Coagulants’ regeneration from sludge improves water quality and reuse options. Organics accumulation is the primary concern regarding coagulant regeneration, using acidification. Our study investigated the raw water quality, aluminum sulfate, and sludge and evaluated its influence on coagulant recovery, using acidification, from eight water treatment plants (WTPs) in Cairo, Egypt. The significant elements in the tested sludge were aluminum with a concentration range of 86.65–688.85 mg/g sludge in El-Rawda and Embaba and iron with a concentration range of 9.45–7.45 mg/g in Shamal Helwan and El-Fostat. Recovery percentages of aluminum, iron, manganese, and strontium recorded the highest values 97%, 89%, 89%, and 92% for Embaba, Rod El-Farag, Embaba, El-Rawda, respectively. The correlation between metal concentration and recovery was insignificant in the studied matrix and conditions for the four metals. Total organic carbon (TOC) transfer into recovered solutions was maximum in El-Fostat (82.6%) and minimum in Embaba (36.7%). The TOC transfer percentage depends on the matrix of the sludge. The best location for coagulant recovery is at the Embaba WTP, where there were minimum organics transfer and maximum Al recovery.









Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Baird RB, Eaton AD, Clesceri LS (2012) In: Rice EW (ed) Standard methods for the examination of water and wastewater, vol 10. American Public Health Association, Washington
Benjamin MM, Lawler DF (2013) Water quality engineering: physical/chemical treatment processes. John Wiley and Sons
Bertsch PM, Bloom PR (1996) Aluminum. Methods Soil Anal: Part 3. Chem Methods 5:517–550. https://doi.org/10.2136/sssabookser5.3.c18
Elarabawy M, Tosswell P, Attia B (2000) Integrated water resources management for Egypt. J Water Supply Res Technol AQUA 49(3):111–125. https://doi.org/10.2166/aqua.2000.0010
Fouad MM, El-Gendy AS, Razek TM (2017a) Evaluation of leached metals in recovered aluminum coagulants from water treatment slurry. Water Sci Technol 75(3-4):998–1006. https://doi.org/10.2166/wst.2016.582
Fouad MM, Razek T, Elgendy AS (2017b) Utilization of drinking water treatment slurry to produce aluminum sulfate coagulant. Water Environ Res 89(2):186–191. https://doi.org/10.2175/106143016X14504669769056
Fouad MM, Razek T, El-Gendy AS (2018) Repeated aluminum sulfate recovery from waterworks sludge: a case study in El-Sheikh Zaid WTP. Water Environ Res 90(12):2030–2035. https://doi.org/10.2175/106143017X15131012188150
Geriesh MH, Balke KD, El-Rayes AE (2008) Problems of drinking water treatment along Ismailia Canal Province, Egypt. Egypt J Zhejiang Univ Sci B 9(3):232–242. https://doi.org/10.1631/jzus.b0710634
Hagedorn B (2008) Acid digestion of sediments, sludge, and soil (EPA 3050). Applied Science, Engineering and Technology Laboratory, University of Alaska, Anchorage
Hirsch L (1960) Inventor; Harvey Machine Co Inc, assignee. Process for the production of aluminum sulfate. United States patent US 2,958,580, Washington, DC
Hosseini SA, Niaei A, Salari D (2011) Production of γ-Al2O3 from Kaolin Seyed. Open J Phys Chem 1(2):23–27. https://doi.org/10.4236/ojpc.2011.12004
Hussein MMF, Khalil MM, El-Gendy AS, Razik T (2020) Tracing TOC concentration and SUVA254 in surface water, treated water and wasted aluminum sludge. Egypt J Chem 64(11):6–8. https://doi.org/10.21608/ejchem.2020.34664.2724
Keeley J, Jarvis P, Judd SJ (2014) Coagulant recovery from water treatment residuals: a review of applicable technologies. Crit Rev Environ Sci Technol 44(24):2675–2719. https://doi.org/10.1080/10643389.2013.829766
Lu Y, Nakicenovic N, Visbeck M, Stevance AS (2015) Policy: five priorities for the UN sustainable development goals. Nat News 520(7548):432–433. https://doi.org/10.1038/520432a
Maxfield R, Mindak B (1985) EPA (Environmental Protection Agency) Method Study 27, Method 200. 7 trace metals by ICP (inductively coupled plasma) (No. PB-85-248656/XAB). Versar, Inc., Springfield, VA (USA)
O’Donnell AJ (2014) Strontium in drinking water: Occurrence, distribution, and removal. (Electronic thesis or dissertation). Retrieved from https://etd.ohiolink.edu/
Potter BB, Wimsatt JC (2005) METHOD 415.3 - a measurement of total organic carbon, dissolved organic carbon, and specific UV absorbance at 254 nm in source water and drinking water. U.S. Environmental Protection Agency, Washington, DC
Ramadan H, El Sayed AEA (2019) Optimization of alum recovery from water treatment sludge-case study: Samannoud water treatment plant, Egypt. Water Environ J 34(3):464–473. https://doi.org/10.1111/wej.12481
Shamrukh M, Abdel-Wahab A (2008) Riverbank filtration for sustainable water supply: application to a large-scale facility on the Nile River. Clean Technol Environ 10(4):351–358. https://doi.org/10.1007/s10098-007-0143-2
Acknowledgements
The authors proudly acknowledge the reference lab for water and the reference lab for wastewater and the general department for quality and environmental affairs in the holding company for water and wastewater, Egypt, and Professor Peter Jarvis, Professor of Water Science & Technology Cranfield Water Science Institute, the UK, for their thankful support and efforts introduced to our research to reach its final form.
Author information
Authors and Affiliations
Contributions
ASE, TMAR, and MMHK formulated the research idea and initialized the first steps to start. ASE and MMF provided the research plan and allocated available resources. MMF performed the practical part of sampling, sludge analysis, coagulant recovery, and analysis. ASE and MMF reviewed the generated raw data and started writing the manuscript. TMAR, ASE, and MMHK reviewed the research article several times to ensure the best quality. MMF applied the required modification for the research article. Finally, all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ta Yeong Wu
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fouad, M.M., El-Gendy, A.S., Khalil, M.M.H. et al. Aluminum sulfate regeneration from surface water treatment waste in Cairo, Egypt. Environ Sci Pollut Res 28, 61450–61459 (2021). https://doi.org/10.1007/s11356-021-15140-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-15140-6