Abstract
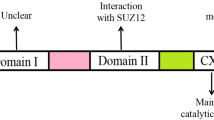
Liver diseases are particularly severe health problems, but the options available for preventing and treating them remain limited. Accumulating evidence has shown that there is altered expression of individual histone deacetylase (HDAC) family members in hepatocellular carcinoma cells. In a previous study, we have identified a set of proteins which interact with histone deacetylase 1 and 3 (HDAC1/3) in hepatocellular carcinoma cell lines HepG2 by proteomic approach. This study was designed to investigate the therapeutic potential and expression of HDAC1/3-interacting genes in a human hepatocellular carcinoma cell line (HepG2). Pharmacological and transcriptional inhibition of HDAC1/3 resulted in the suppression of cancer cell proliferation, change of cell morphology, and downregulation of HDAC1/3 genes in HepG2 cells. The pharmacological inhibition also resulted in inhibition of liver cancer cell migration by wound scratch assay. Taken together, the results from this study show that the upregulation of HDAC1/3 in hepatocellular carcinoma resulted in the overexpression of CNOT1, PFDN2/6, and HMG20B, and that these genes could serve as novel molecular targets in liver cancer.









Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Ahmad M, Hamid A, Hussain A, Majeed R, Qurishi Y, Bhat JA, Najar RA, Qazi AK, Zargar MA, Singh SK, Saxena AK (2012) Understanding histone deacetylases in the cancer development and treatment: an epigenetic perspective of cancer chemotherapy. DNA Cell Biol 31:S62–S71
Alghamdi IG, Alghamdi MS (2020) The incidence rate of liver cancer in Saudi Arabia: an observational descriptive epidemiological analysis of data from the Saudi Cancer Registry (2004-2014). Cancer Manag Res 12:1101–1111
Andoorfar S, Hosseini Tafreshi SA, Rezvani Z (2019) Assessment of the expression level of miRNA molecules using a semi-quantitative RT-PCR approach. Mol Biol Rep 46:5057–5062
Barneda-Zahonero B, Parra M (2012) Histone deacetylases and cancer. Mol Oncol 6:579–589
Buurman R, Sandbothe M, Schlegelberger B, Skawran B (2016) HDAC inhibition activates the apoptosome via Apaf1 upregulation in hepatocellular carcinoma. Eur J Med Res 21:26
Cappiello F, Casciaro B, Mangoni ML (2018): A novel in vitro wound healing assay to evaluate cell migration. J Vis Exp
Chen R, Zhu S, Fan XG, Wang H, Lotze MT, Zeh HJ 3rd, Billiar TR, Kang R, Tang D (2018) High mobility group protein B1 controls liver cancer initiation through yes-associated protein -dependent aerobic glycolysis. Hepatology 67:1823–1841
Chen Z, Xu K (2009) Effect of fluorouracil combined with FK228 on the proliferation, apoptosis and Fas mRNA level in HepG2 hepatoma cell lines. Zhong Nan Da Xue Xue Bao Yi Xue Ban 34:124–129
Cheng DD, Li J, Li SJ, Yang QC, Fan CY (2017) CNOT1 cooperates with LMNA to aggravate osteosarcoma tumorigenesis through the Hedgehog signaling pathway. Mol Oncol 11:388–404
Dehghan-Nayeri N, Rezaei-Tavirani M, Omrani MD, Gharehbaghian A, Pour KG, Eshghi P (2017) Identification of potential predictive markers of dexamethasone resistance in childhood acute lymphoblastic leukemia. J Cell Commun Signal 11:137–145
Emanuele S, Lauricella M, Carlisi D, Vassallo B, D'Anneo A, Di Fazio P, Vento R, Tesoriere G (2007) SAHA induces apoptosis in hepatoma cells and synergistically interacts with the proteasome inhibitor Bortezomib. Apoptosis 12:1327–1338
Farooq M, Sulochana KN, Pan X, To J, Sheng D, Gong Z, Ge R (2008) Histone deacetylase 3 (hdac3) is specifically required for liver development in zebrafish. Dev Biol 317:336–353
Farooq M, Hozzein WN, Elsayed EA, Taha NA, Wadaan MA (2013) Identification of histone deacetylase 1 protein complexes in liver cancer cells. Asian Pac J Cancer Prev 14:915–921
Farooq M, Wadaan MA (2013) Epigenetic targets in hepatocellular carcinoma cells: identification of chaperone protein complexes with histone deacetylases. Epigenomics 5:501–512
Freese K, Seitz T, Dietrich P, Lee SML, Thasler WE, Bosserhoff A, Hellerbrand C (2019) Histone deacetylase expressions in hepatocellular carcinoma and functional effects of histone deacetylase inhibitors on liver cancer cells in vitro. Cancers (Basel) 11
Ganslmayer M, Konturek P, Herold C, Neurath MF, Zopf S (2012) Antitumoral efficacy of four histone deacetylase inhibitors in hepatoma in vitro and in vivo. Anticancer Res 32:5263–5269
Gong P, Li K, Li Y, Liu D, Zhao L, Jing Y (2018) HDAC and Ku70 axis- an effective target for apoptosis induction by a new 2-cyano-3-oxo-1,9-dien glycyrrhetinic acid analogue. Cell Death Dis 9:623
Hampsey J, Karnsakul W (2013) Liver disorders: nutritional management. In: Caballero B (ed) Encyclopedia of human nutrition (third edition). Academic Press, Waltham, pp 87–99
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Kim HJ, Bae SC (2011) Histone deacetylase inhibitors: molecular mechanisms of action and clinical trials as anti-cancer drugs. Am J Transl Res 3:166–179
Ko S, Russell JO, Tian J, Gao C, Kobayashi M, Feng R, Yuan X, Shao C, Ding H, Poddar M, Singh S, Locker J, Weng HL, Monga SP, Shin D (2019) Hdac1 regulates differentiation of bipotent liver progenitor cells during regeneration via Sox9b and Cdk8. Gastroenterology 156(187-202):e14
Lachenmayer A, Toffanin S, Cabellos L, Alsinet C, Hoshida Y, Villanueva A, Minguez B, Tsai HW, Ward SC, Thung S, Friedman SL, Llovet JM (2012) Combination therapy for hepatocellular carcinoma: additive preclinical efficacy of the HDAC inhibitor panobinostat with sorafenib. J Hepatol 56:1343–1350
Lee M, Daniels MJ, Garnett MJ, Venkitaraman AR (2011) A mitotic function for the high-mobility group protein HMG20b regulated by its interaction with the BRC repeats of the BRCA2 tumor suppressor. Oncogene 30:3360–3369
Lee M, Venkitaraman AR (2014) A cancer-associated mutation inactivates a region of the high-mobility group protein HMG20b essential for cytokinesis. Cell Cycle 13:2554–2563
Li Y, Seto E (2016) HDACs and HDAC inhibitors in cancer development and therapy. Cold Spring Harb Perspect Med 6
Liang J, Xia L, Oyang L, Lin J, Tan S, Yi P, Han Y, Luo X, Wang H, Tang L, Pan Q, Tian Y, Rao S, Su M, Shi Y, Cao D, Zhou Y, Liao Q (2020) The functions and mechanisms of prefoldin complex and prefoldin-subunits. Cell Biosci 10:87
Liu M, Jiang L, Guan XY (2014) The genetic and epigenetic alterations in human hepatocellular carcinoma: a recent update. Protein Cell 5:673–691
Lu XF, Cao XY, Zhu YJ, Wu ZR, Zhuang X, Shao MY, Xu Q, Zhou YJ, Ji HJ, Lu QR, Shi YJ, Zeng Y, Bu H (2018) Histone deacetylase 3 promotes liver regeneration and liver cancer cells proliferation through signal transducer and activator of transcription 3 signaling pathway. Cell Death Dis 9:398
Marks PA (2007) Discovery and development of SAHA as an anticancer agent. Oncogene 26:1351–1356
Meier K, Brehm A (2014) Chromatin regulation: how complex does it get? Epigenetics 9:1485–1495
Nami B, Wang Z (2018) Genetics and expression profile of the tubulin gene superfamily in breast cancer subtypes and its relation to taxane resistance. Cancers (Basel) 10
Noel ES, Casal-Sueiro A, Busch-Nentwich E, Verkade H, Dong PD, Stemple DL, Ober EA (2008) Organ-specific requirements for Hdac1 in liver and pancreas formation. Dev Biol 322:237–250
Pavanello L, Hall B, Airhihen B, Winkler GS (2018) The central region of CNOT1 and CNOT9 stimulates deadenylation by the Ccr4-Not nuclease module. Biochem J 475:3437–3450
Petit AP, Wohlbold L, Bawankar P, Huntzinger E, Schmidt S, Izaurralde E, Weichenrieder O (2012) The structural basis for the interaction between the CAF1 nuclease and the NOT1 scaffold of the human CCR4-NOT deadenylase complex. Nucleic Acids Res 40:11058–11072
Prabst K, Engelhardt H, Ringgeler S, Hubner H (2017) Basic colorimetric proliferation assays: MTT, WST, and resazurin. Methods Mol Biol 1601:1–17
Rikimaru T, Taketomi A, Yamashita Y, Shirabe K, Hamatsu T, Shimada M, Maehara Y (2007) Clinical significance of histone deacetylase 1 expression in patients with hepatocellular carcinoma. Oncology 72:69–74
Rodriguez-Paredes M, Esteller M (2011) Cancer epigenetics reaches mainstream oncology. Nat Med 17:330–339
Souza ACMF, Souza DRV, Sanabani SS, Giorgi RR, Bendit I (2009) The performance of semi-quantitative differential PCR is similar to that of real-time PCR for the detection of the MYCN gene in neuroblastomas. Braz J Med Biol Res 42:791–795
Sumoy L, Carim L, Escarceller M, Nadal M, Gratacos M, Pujana MA, Estivill X, Peral B (2000) HMG20A and HMG20B map to human chromosomes 15q24 and 19p13.3 and constitute a distinct class of HMG-box genes with ubiquitous expression. Cytogenet Cell Genet 88:62–67
Vainberg IE, Lewis SA, Rommelaere H, Ampe C, Vandekerckhove J, Klein HL, Cowan NJ (1998) Prefoldin, a chaperone that delivers unfolded proteins to cytosolic chaperonin. Cell 93:863–873
Wang YC, Yang X, Xing LH, Kong WZ (2013) Effects of SAHA on proliferation and apoptosis of hepatocellular carcinoma cells and hepatitis B virus replication. World J Gastroenterol 19:5159–5164
Winkler GS, Mulder KW, Bardwell VJ, Kalkhoven E, Timmers HT (2006) Human Ccr4-Not complex is a ligand-dependent repressor of nuclear receptor-mediated transcription. EMBO J 25:3089–3099
Yang W, Zhao X, Pei F, Ji M, Ma W, Wang Y, Jiang G (2015) Activation of the intrinsic apoptosis pathway contributes to the induction of apoptosis in hepatocellular carcinoma cells by valproic acid. Oncol Lett 9:881–886
Zhou H, Cai Y, Liu D, Li M, Sha Y, Zhang W, Wang K, Gong J, Tang N, Huang A, Xia J (2018) Pharmacological or transcriptional inhibition of both HDAC1 and 2 leads to cell cycle blockage and apoptosis via p21(Waf1/Cip1) and p19(INK4d) upregulation in hepatocellular carcinoma. Cell Prolif 51:e12447
Zhou H, Xu J, Zhang C, Wen Y (2019) Aberrant histone deacetylase 1 expression upregulates vimentin expression via an NF-kappaB-dependent pathway in hepatocellular carcinoma. Oncol Lett 18:339–347
Acknowledgements
The authors thank the Deanship of Scientific Research and RSSU at King Saud University for the technical support. The authors also acknowledge Najla bin Shwish, Mary Grace Vigilla, and Rhodanne Lambarte for their assistance and contributions to this report, and the Molecular and Cell Biology Laboratory, a core research facility of the King Saud University College of Dentistry, in collaboration with the Prince Naif bin AbdulAziz Health Research Center, where part of this study was performed.
Funding
This study was funded by the Deanship of Scientific Research, King Saud University, through the Vice Deanship of Scientific Research Chairs. The funding body had no role in the design, collection, analysis, and interpretation of data, or writing of the manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization: M.F.K., R.S.A., and M.A.W. Methodology: M.F.K., N.A.Y., and M.M.A. Resources: M.F.K., M.S.A., and M.A.W. Data curation: N.A.Y., M.F.K., M.M.A., and M.S.A. Writing—original draft preparation: M.F.K., N.A.Y., M.M.A. Writing—review and editing: M.F.K., M.M.A., M.S.A., R.S.A., and M.A.W. Supervision: M.F.K. Funding acquisition: M.F.K. and M.A.W. All the authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Al-yhya, N., Khan, M.F., Almeer, R.S. et al. Pharmacological inhibition of HDAC1/3-interacting proteins induced morphological changes, and hindered the cell proliferation and migration of hepatocellular carcinoma cells. Environ Sci Pollut Res 28, 49000–49013 (2021). https://doi.org/10.1007/s11356-021-13668-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-13668-1