Abstract
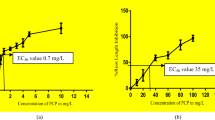
The organic toxicants formed in chlorinated water cause potential harm to human beings, and it is extensively concentrated all over the world. Various disinfection by-products (DBPs) occur in chlorinated water are genotoxic and carcinogenic. The toxicity is major concern for chlorinated DBPs which has been present more in potable water. The purpose of the work was to evaluate genotoxic properties of DBPs in Allium cepa as a plant model system. The chromosomal aberration and DNA laddering assays were performed to examine the genotoxic effect of trichloroacetic acid (TCAA), trichloromethane (TCM), and tribromomethane (TBM) in a plant system with distinct concentrations, using ethyl methanesulfonate (EMS) as positive control and tap water as negative control. In Allium cepa root growth inhibition test, the inhibition was concentration dependent, and EC50 values for trichloroacetic acid (TCAA), trichloromethane (TCM), and tribromomethane (TBM) were 100 mg/L, 160 mg/L, and 120 mg/L respectively. In the chromosome aberration assay, root tip cells were investigated after 120 h exposure. The bridge formation, sticky chromosomes, vagrant chromosomes, fragmented chromosome, c-anaphase, and multipolarity chromosomal aberrations were seen in anaphase–telophase cells. It was noticed that with enhanced concentrations of DBPs, the total chromosomal aberrations were more frequent. The DNA damage was analyzed in roots of Allium cepa exposed with DBPs (TCAA, TCM, TBM) by DNA laddering. The biochemical assays such as lipid peroxidation, H2O2 content, ascorbate peroxidase, guaiacol peroxidase, and catalase were concentration dependent. The DNA interaction studies were performed to examine binding mode of TCAA, TCM, and TBM with DNAs. The DNA interaction was evaluated by spectrophotometric and spectrofluorometric studies which revealed that TCAA, TCM, and TBM might interact with Calf thymus DNA (CT- DNA) by non-traditional intercalation manner.






Similar content being viewed by others
References
Ammar WB, Nouairi I, Zarrouk M, Ghorrbel M, Jemal F (2008) Antioxidative response to cadmium in roots and leaves of tomato plants. Biol Plant 52:727–731
Arjmand F, Jamsheera A (2011) DNA binding studies of new valine derived chiral complexes of tin (iv) and zirconium (iv). Spectrochim Acta A 78:45–51
Arya SK, Mukherjee A (2014) Sensitivity of Allium cepa and Vicia faba towards cadmium toxicity. Jour Soil Sci Plant Nutri 14(2):447–458
Asada K, Takahashi M (1987) Production and scavenging of active oxygen in photosynthesis. In: Photoinhibition, pp 227–287
Bela K, Horvath E, Galle A, Szabados L, Tari I, Csiszar J (2015) Plant glutathione peroxidases: emerging role of the antioxidant enzymes in plant development and stress responses. Jour Plant Physio 176:192–197
Butour JL, Macquet JP (1977) Differentiation of DNA-platinum complexes by fluorescence. The use of an intercalating dye as a probe. Eur J Biochem 78:455–463
Canistro D, Melega S, Ranieri D, Sapone A, Gustavino B, Monfrinotti M, Rizzoni M, Paolini M (2012) Modulation of cytochrome P450 and induction of DNA damage in Cyprinus carpio exposed in situ to surface water treated with chlorine or alternative disinfectants in different seasons. Mutat Res 729(1–2):81–89
Çelik A, Ünyayar S, Çekiç FO, Güzel A (2008) Micronucleus frequency and lipid peroxidation in Allium sativum root tip cells treated with gibberellic acid and cadmium. Cell Biol Toxicol 24:159–164
Chowdhury S, Alhooshani K, Karanfil T (2014) Disinfection byproducts in swimming pool: occurrences, implications and future needs. Water Res 53:68–109
Chowdhury SR, Mukherjee KK, Bhattacharyya R (2005) Biophisical and biochemical investigation on the binding of the manganese - cyanonitosyl complex with DNA. Trans Met Chem 30:601-604
Cirillo S, Canistro D, Vivarelli F, Paolini M (2016) Effects of chlorinated drinking water on the xenobiotic metabolism in Cyprinus carpio treated with samples from two Italian municipal networks. Environ Sci Pollut Res 23:18777–18788
Collins A, Harrington V (2002) Repair of oxidative DNA damage: assessing its contribution to cancer prevention. Mutagenesis 17:489–493
Dat J (2000) Dual action of the active oxygen species during plant stress responses. Cell Mol Life Sci 57:779–795
Drotar A, Phelps P, Fall R (1985) Evidence for glutathione peroxidase activities in cultured plant cells. Plant Sci 42:35–40
Fantuzzi G, Righi E, Predieri G, Giacobazzi P, Mastroianni K, Aggazzotti G (2010) Prevalence of ocular, respiratory and cutaneous symptoms in indoor swimming pool workers and exposure to disinfection by-products (DBPs). Int J Environ Res Public Health 7:1379–1391
Farre´ MJ, Day S, Neale PA, Stalter D, Tang JYM, Escher BI (2013) Bioanalytical and chemical assessment of the disinfection by-product formation potential: role of organic matter. Water Res 47(14):5409–5542
Fiskesjö G (1987) The Allium test as a standard in environmental monitoring. Hereditas 102:99–112
Fiskesjo G, Levan A (1993) Evaluation of the first ten MEIC chemicals in the Allium test. ATLA 21:139–149
Foyer CH, Noctor G (2005) Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. Plant Cell 17:1866–1875
Ghoshal S, Bandyopadhyay A, Mukherjee A (2010) Genotoxicity of titanium dioxide (TiO2) nanoparticles at two tropic levels: plant and human lymphocytes. Chemosphere 81:253–1262
Grata¨o PL, Polle A, Lea PJ, Azevedo RA (2005) Making the life of heavy metal-stressed plants a little easier. Funct Plant Biol 32:481–494
Hammond-Kosack KE, Jones JDG (1996) Resistance gene-dependent plant defense responses. Plant Cell 8:1773–1791
Hartley-Whitaker J, Ainsworth G, Meharg AA (2001) Copper and arsenate induced oxidative stress in Holcus lanatus L. clones with differential sensitivity. Plant Cell Environ 24:13–22
Hinckley AF, Bachand AM, Reif JS (2005) Late pregnancy exposures to disinfection byproducts and growth-related birth outcomes. Environ Health Perspect 113:1808–1813
Hu Y, Tan L, Zhang S, Zuo Y, Han X, Liu N, Lu W, Liu A (2017) Detection of genotoxic effects of drinking water disinfection by- products using Vicia faba bioassay. Environ Sci Pollut Res 24:1509–1517
Kapustka LA, Lipton J, Galbraith H, Cacela D, Lejeune K (1995) Metallic and arsenic impacts to soils, vegetation communities and wildlife habitat in southwest Montana uplands contained by smelter emissions: II. Laboratory phytotoxicity studies. Environ Toxicol Chem 14:1905–1912
Kelly TM, Tossi AB, McConnel DJ, Streakas TC (1985) A study of the interactions of some polypyridylruthenium (II) complexes with DNA using fluorescence spectroscopy, topoisomerisation and thermal denaturation. Nucleic Acids Res 13:6017–6034
Kogevinas M, Villanueva CM, Font-Ribera L, Liviac D, Bustamante M, Espinoza F, Nieuwenhuijsen MJ, Espinosa A, Fernandez P, DeMarini DM, Grimalt JO, Grummt T, Marcos R (2010) Genotoxic effects in swimmers exposed to disinfection by-products in indoor swimming pools. Environ Health Perspect 118(11):1531–1537
Kumar A, Majeti NVP (2014) Proteomic responses to lead-induced oxidative stress in Talinum triangulare Jacq. (Willd.) roots: identification of key biomarkers related to glutathione metabolisms. Environ Sci Pollut Res 21:8750–8764
Kumari M, Mukherjee A, Chandrasekaran N (2009) Genotoxicity of silver nanoparticles in Allium cepa. Sci Total Environ 407:5243–5246
Leme DM, Marin-Morales MA (2009) Allium cepa test in environmental monitoring: a review on its application. Mutat Res 682:71–81
Lutterbeck CA, Kem DI, Machado ÊL, Kümmerer K (2015) Evaluation of the toxic effects of four anti-cancer drugs in plant bioassays and its potency for screening in the context of waste water reuse for irrigation. Chemosphere 135:403–410
Malar S, Manikandan R, Favas PJC, Vikram Sahi S, Venkatachalam P (2014) Effect of lead on phytotoxicity, growth, biochemical alterations and its role on genomic template stability in Sesbania grandiflora: a potential plant for phytoremediation. Ecotoxicol Environ Saf 108:249–257
Manasfi T, De Méo M, Coulomb B, Di Giorgio C, Boudenne JL (2016) Identification of disinfection by-products in freshwater and seawater swimming pools and evaluation of genotoxicity. Environ Int 88:94–102
Mascher R, Lippmann B, Holzinger S, Bergmann H (2002) Arsenate toxicity: effects on oxidative stress response molecules and enzymes in red clover plants. Plant Sci 163:961–969
Michałowicz J, Posmyk M, Duda W (2009) Chlorophenols induce lipid peroxidation and change antioxidant parameters in the leaves of wheat (Triticum aestivum L.). J Plant Physiol 166:559–568
Michalowicz J, Urbanek H, Bukowska, Duda W (2010) The effect of 2,4-dichlorophenol and pentachlorophenol on antioxidant system in the leaves of Phalaris arudinacea. Biol Plant 54(3):597–600
Mittler R (2002) Oxidative stress, antioxidant and stress tolerance. Trends Plant Sci 7:405–410
Mogren LM, Olssen ME, Gertsson UE (2007) Effects of cultivar, lifting time and nitrogen fertilizer level on quercetin content in onion (Allium cepa L.) at lifting. J Sci Food Agri 87:470–476
Montillet J-L, Chamnongpol S, Ruste’rucci C, Dat J, Van de Cotte B, Agnel J-P, Battesti C, Inze’ D, Van Breusegem F, Triantaphylide’s C (2005) Fatty acid hydroperoxides and H2O2 in the execution of hypersensitive cell death in tobacco leaves. Plant Physiol 138:1516–1526
Neale Pa, Antony A, Bartkow ME, Farré MJ, Heitz A, Kristiana I, Tang JYM, Escher BI (2012) Bioanalytical assessment of the formation of disinfection byproducts in a drinking water treatment plant. Environ Sci Technol 46:10317–10325
Oberdorster G, Stone V, Donaldson K (2007) Toxicology of nanoparticles: a historical perspective. Nanotoxicology 1:2–25
Pasqualini V, Robles C, Garcino S, Greff S, Bousquet- Melou A, Bonin G (2003) Phenolic compounds content in Pinus halepensis mill. Needles: a bioindicator of air pollution. Chemosphere 52:239–248
Pentamwa P, Sukton B, Wongklom T, Pentamwa S (2013) Cancer risk assessment from trihalomethanes in community water supply at northeastern Thailand. Int Jour of Environ Sci Develop 5:538–544
Plewa MJ, Wagner ED, Mitch WA (2011) Comparative mammalian cell cytotoxicity of water concentrates from disinfected recreational pools. Environ. Sci. Technol. 45(9):4159–4165
Plewa MJ, Wagner ED, Metz DH, Kashinkunti R, Jamriska K, Meyer M (2012) Differential toxicity of drinking water disinfected with combinations of ultraviolet radiation and chlorine. Environ. Sci. Technol. 46(14):7811–7817
Rahban M, Divsalar A, Saboury AA, Golestani A (2010) Nanotoxicity and spectroscopy studies of silver nanoparticle: calf thymus DNA and K562 as targets. J Phys Chem C 114:5798–5803
Reeves JF, Davies SJ, Dodd NJF, Jha AN (2007) Hydroxyl radicals (-OH) are associated with titanium dioxide (TiO2) nanoparticle-induced cytoxicity and oxidative DNA damage in fish cells. Mutat Res 640:113–122
Rhizsky L, Hallak-Herr E, Van Breusegen F, Rachmilevich S, Barr J, Rodermel S, Inze S, Mittler R (2002) Double antisense plants lacking ascorbate peroxidase and catalase are less sensitive to oxidative stress than single antisense plants lacking ascorbate peroxidase and catalase. Planta 32:329–342
Richardson SD, Plewa MJ, Wagner ED, Schoeny R, DeMarini DM (2007) Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection byproducts in drinking water: a review and roadmap for research. Mutat Res Rev Mutat Res 636(1–3):178–242
Richardson SD, DeMarini DM, Kogevinas M, Fernandez P, Marco E, Lourencetti C, Balleste C, Heederik D, Meliefste K, McKague AB, Marcos R, Font-Ribera L, Grimalt JO, Villanueva CM (2010) What’s in the pool? A comprehensive identification of disinfection by-products and assessment of mutagenicity of chlorinated and brominated swimming pool water. Environ Health Perspect 118(11):1523–1530
Rook JJ (1974) Formation of haloforms during chlorination of natural waters. J Water Treat Examin 23:234–243
Ross PD, Subramanian S (1981) Thermodynamics of protein association reactions: forces contributing to stability. J Biochem 20:3096–3102
Roy S, Ihantola R, Hanninen O (1992) Peroxidase activity in lake macrophytes and its relation to pollution tolerance. Environ Exp Bot 32:457–464
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, vol 9, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, Chapter, pp 14–23
Sapone A, Canistro D, Vivarelli F, Paolini M (2016) Perturbation of xenobiotic metabolism in Dreissena polymorpha model exposed in situ to surface water (Lake Trasimene) purified with various disinfectants. Chemosphere 144:548–554
Schroder P, Gotzberger C (1997) Partial purification and characterization of glutathione S- trasferase isozymes from the leaves of Juniperus communis, Larix deciduas and Taxus baccata. Appl Botany 71:31–37
Shahabadi N, Fatahi A (2010) Multispectroscopic DNA-binding studies of a tris-chelate nickel (II) complex containing 4,7-diphenyl 1,10-phenanthroline ligands. J Mol Struct 970(1–3):90–95
Shahabadi N, Hadidi S (2012) Spectroscopic studies on the interaction of calf thymus DNA with the drug levetiracetam. Spectrochim Acta A Mol Biomol Spectrosc 96:278–2833
Sharma AK, Sharma A (1980) Chromosome Techniques – Theory and Practice, third edn. Butterworths, London, p 711
Singh N, Ma LQ, Srivastava M, Rathinasabapathi B (2006) Metabolic adaptations to arsenic-induced oxidative stress in Pteris vittata L. and Pteris ensiformis L. Plant Sci 170:274–282
Singh H, Batish D, Kohli R, Arora K (2007) Arsenic-induced root growth inhibition in mung bean ( Phaseolus aureus Roxb.) is due to oxidative stress resulting from enhance lipid peroxidation. Plant Growth Regul 53:65–73
Son GS, Yeo JA, Kim JM, Kim SK, Moon HR, Nam W (1998) Base specific complex formation of norfloxacin with DNA. Biophys Chem 74:225–236
Srivastava M, Ma LQ, Singh N, Singh S (2005) Antioxidant responses of hyper-accumulator and sensitive fern species to arsenic. J Exp Bot 56:1335–1342
Stoeva N, Berova M, Zlatev Z (2005) Effect of arsenic on some physiological parameters in bean plants. Biol Plant 49:293–296
Teo TLL, Coleman HM, Khan SJ (2015) Chemical contaminants in swimming pools: occurrence, implications and control. Environ Int 76:16–31
Villanueva CM, Cantor KP, Grimalt JO, Malats N, Silverman D, Tardon A, Garcia-Closas R, Serra C, Carrato A, Castaño-Vinyals G, Marcos R, Rothman N, Real FX, Dosemeci M, Kogevinas M (2007) Bladder cancer and exposure to water disinfection by-products through ingestion, bathing, showering, and swimming in pools. Am J Epidemiol 165:148–156
Zhang H, Jiang Y, He Z, Ma M (2005) Cadmium accumulation and oxidative burst in garlic (Allium sativum). J Plant Physiol 162:977–984
Zhu R, Wang S, Chao J, Sun X, Yao S (2008) Oxidative and binding effect of nano- TiO2 on plasmid DNA and pepsin. In: the 2nd international conference on bioinformatics and Biomed Eng, pp. 1013–1016
Acknowledgments
The authors would like to thank TEQIP II and the National Institute of Technology, Durgapur, for providing good research lab facilities.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Gangrong Shi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ranjan, J., Mandal, T. & Mandal, D.D. Environmental risk appraisement of disinfection by-products (DBPs) in plant model system: Allium cepa. Environ Sci Pollut Res 26, 8609–8622 (2019). https://doi.org/10.1007/s11356-019-04262-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-04262-7