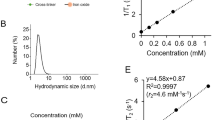
Abstract
Purpose
Gadolinium (Gd)-based contrast agents are primarily used for contrast-enhanced magnetic resonance lymphangiography (MRL). However, overcoming venous contamination issues remains challenging. This study aims to assess the MRL efficacy of the newly developed iron-based contrast agent (INV-001) that is specially designed to mitigate venous contamination issues. The study further explores the optimal dosage, including both injection volume and concentration, required to achieve successful visualization of the popliteal lymph nodes and surrounding lymphatic vessels.
Procedures.
All animals utilized in this study were male Sprague–Dawley (SD) rats weighing between 250 and 300 g. The contrast agents prepared were injected intradermally in the fourth phalanx of both hind limbs using a 30-gauge syringe in SD rats. MRL was performed every 16 min on a coronal 3D time-of-flight sequence with saturation bands using a 9.4-T animal machine.
Results
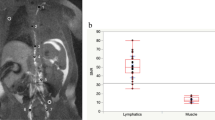
Contrary to Gd-DOTA, which exhibited venous contamination in most animals irrespective of injection dosages and conditions, INV-001 showed no venous contamination. For Gd-DOTA, the popliteal lymph nodes and lymphatic vessels reached peak enhancement 16 min after injection from the injection site and then rapidly washed out. However, with INV-001, they reached peak enhancement between 16 and 32 min after injection, with prolonged visualization of the popliteal lymph node and lymphatic vessels. INV-001 at 0.45 μmol (15 mM, 30 μL) and 0.75 μmol (15 mM, 50 μL) achieved high scores for qualitative image analysis, providing good visualization of the popliteal lymph nodes and lymphatic vessels without issues of venous contamination, interstitial space enhancement, or lymph node enlargement.
Conclusion
In MRL, INV-001, a novel T1 contrast agent based on iron, enables prolonged enhancement of popliteal lymph nodes and lymphatic vessels without venous contamination.






Similar content being viewed by others
References
Notohamiprodjo M, Baumeister RGH, Jakobs TF et al (2009) MR-lymphangiography at 3.0T-a feasibility study. Eur Radiol 19:2771–2778
Notohamiprodjo M, Weiss M, Baumeister RG et al (2012) MR lymphangiography at 3.0 T: correlation with lymphoscintigraphy. Radiology 264:78–87
White RD, Weir-McCall JR, Budak MJ, Waugh SA, Munnoch DA, Sudarshan TAP (2014) Contrast-enhanced magnetic resonance lymphography in the assessment of lower limb lymphoedema. Clin Radiol 69:E435–E444
Mitsumori LM, McDonald ES, Wilson GJ, Neligan PC, Minoshima S, Maki JH (2015) MR lymphangiography: how i do it. J Magn Reson Imaging 42:1465–1477
Kim EY, Hwang HS, Lee HY et al (2016) Anatomic and functional evaluation of central lymphatics with noninvasive magnetic resonance lymphangiography. Medicine 95:e3109
Salehi BP, Sibley RC, Friedman R et al (2023) MRI of lymphedema. J Magn Reson Imaging 57:977–991
Xiong L, Engel H, Gazyakan E et al (2014) Current techniques for lymphatic imaging: State of the art and future perspectives. Ejso-Eur J Surg Onc 40:270–276
Guerrini S, Gentili F, Mazzei FG, Gennaro P, Volterrani L, Mazzei MA (2020) Magnetic resonance lymphangiography: with or without contrast? Diagn Interv Radiol 26:587–595
Mills M, van Zanten M, Borri M et al (2021) Systematic review of magnetic resonance lymphangiography from a technical perspective. J Magn Reson Imaging 53:1766–1790
Ruehm SG, Schroeder T, Debatin JF (2001) Interstitial MR lymphography with gadoterate meglumine: Initial experience in humans. Radiology 220:816–821
Lohrmann C, Foeldi E, Speck O, Langer M (2006) High-resolution MR lymphangiography in patients with primary and secondary lymphedema. AJR Am J Roentgenol 187:556–561
Lohrmann C, Foeldi E, Langer M (2006) Indirect magnetic resonance lymphangiography in patients with lymphedema preliminary results in humans. Eur J Radiol 59:401–406
Felmerer G, Sattler T, Lohrmann C, Tobbia D (2012) Treatment of various secondary lymphedemas by microsurgical lymph vessel transplantation. Microsurg 32:171–177
Mitsumori LM, McDonald ES, Neligan PC, Maki JH (2016) Peripheral magnetic resonance lymphangiography: techniques and applications. Tech Vasc Interv Radiol 19:262–272
Mitsumori LM (2016) Response: Magnetic resonance lymphangiography: How to prove it? J Magn Reson Imaging 44:1368–1369
Maki JH, Neligan PC, Briller N, Mitsumori LM, Wilson GJ (2016) Dark blood magnetic resonance lymphangiography using dual-agent relaxivity contrast (DARC-MRL): a novel method combining gadolinium and iron contrast agents. Curr Probl Diagn Radiol 45:174–179
Kobayashi H, Kawamoto S, Bernardo M, Brechbiel MW, Knopp MV, Choyke PL (2006) Delivery of gadolinium-labeled nanoparticles to the sentinel lymph node: Comparison of the sentinel node visualization and estimations of intra-nodal gadolinium concentration by the magnetic resonance imaging. J Control Release 111:343–351
Kobayashi H, Kawamoto S, Choyke PL et al (2003) Comparison of dendrimer-based macromolecular contrast agents for dynamic micro-magnetic resonance lymphangiography. Magn Reson Med 50:758–766
Müller A, Fries P, Jelvani B et al (2017) Magnetic Resonance lymphography at 9.4 T using a gadolinium-based nanoparticle in rats. Invest Radiol 52:725–733
Kuo PH, Kanal E, Abu-Alfa AK, Cowper SE (2007) Gadolinium-based MR contrast agents and nephrogenic systemic fibrosis. Radiology 242:647–649
Weinreb JC, Rodby RA, Yee J et al (2021) Use of intravenous gadolinium-based contrast media in patients with kidney disease: consensus statements from the American College of Radiology and the National Kidney Foundation. Radiology 298:28–35
Cheong BYC, Wilson JM, Preventza OA, Muthupillai R (2022) Gadolinium-based contrast agents: updates and answers to typical questions regarding gadolinium use. Tex Heart I J 49(3):e217680
Shin TH, Kim PK, Kang S et al (2021) High-resolution T(1) MRI via renally clearable dextran nanoparticles with an iron oxide shell. Nat Biomed Eng 5:252–263
Mounzer R, Shkarin P, Papademetris X, Constable T, Ruddle NH, Fahmy TM (2007) Dynamic imaging of lymphatic vessels and lymph nodes using a bimodal nanoparticulate contrast agent. Lymphat Res Biol 5:151–158
Malhotra N, Lee JS, Liman RAD et al (2020) Potential toxicity of iron oxide magnetic nanoparticles: a review. Molecules 25:3159
Turkbey B, Czarniecki M, Shih JH et al (2020) Ferumoxytol-enhanced MR lymphography for detection of metastatic lymph nodes in genitourinary malignancies: a prospective study. AJR Am J Roentgenol 214:105–113
Hamilton BE (2020) Ferumoxytol-enhanced MRI is not inferior to gadolinium-enhanced MRI in detecting intracranial metastatic disease and metastasis size. Am J Roentgenol 215:1551–1551
Daldrup-Link HE, Theruvath AJ, Rashidi A et al (2022) How to stop using gadolinium chelates for magnetic resonance imaging: clinical-translational experiences with ferumoxytol. Pediatr Radiol 52:354–366
Werner P, Taupitz M, Schröder L, Schuenke P (2021) An NMR relaxometry approach for quantitative investigation of the transchelation of gadolinium ions from GBCAs to a competing macromolecular chelator. Sci Rep-Uk 11
Acknowledgements
We thank the core facilities of the Magnetic Resonance Core and the Comparative Pathology Core at the ConveRgence mEDIcine research cenTer (CREDIT), Asan Medical Center, for enabling the use of their equipment, services, and expertise.
Funding
This research was supported by the Bio&Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean government (MSIT) (No. 2022M3A9G1014476); the National Research Foundation of Korea (NRF) (Grant No. 2019R1C1C1010887/ 2022R1Z1A1A01066589) funded by the Korean government (MIST); the R&D project linked to Biohealth Investment Infrastructure (Grant No. HI21C1317) funded by the Ministry of Health & Welfare/Biohealth Innovation Start-up Center, Republic of Korea; and Asan Institute of Life Sciences, Asan Medical Center, Republic of Korea (2021IL0030).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosure of Conflicts of Interest
K.W.K. & M.H.K. are employed by Trial Informatics. J.W.K. & T.H.S. are employed by Inventera Inc., a startup focused on developing nanoimaging agents. There are no other financial interests or potential conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chae, Y.J., Kim, K.W., Kim, Mh. et al. Comparison of the Pharmacokinetics of Gadolinium-Based and Iron Oxide-Based Contrast Agents inside the Lymphatic Structure using Magnetic Resonance Lymphangiography. Mol Imaging Biol (2024). https://doi.org/10.1007/s11307-024-01918-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11307-024-01918-w