Abstract
Introduction
Studies of gastrointestinal physiology and the gut microbiome often consider the influence of intestinal region on experimental endpoints. However, this same consideration is not often applied to the gut metabolome. Understanding the contribution of gut regionality may be critically important to the rapidly changing metabolic environments, such as during pregnancy.
Objectives
We sought to characterize the difference in the gut metabolome in pregnant mice stratified by region—comparing the small intestine, cecum, and feces. Pre-pregnancy feces were collected to understand the influence of pregnancy on the fecal metabolome.
Methods
Feces were collected from CD-1 female mice before breeding. On gestation day (GD) 18, gut contents were collected from the small intestine, cecum, and descending colon. Metabolites were analyzed with LC–MS/MS using the Biocrates MetaboINDICATOR™ MxP® Quant 500 kit.
Results
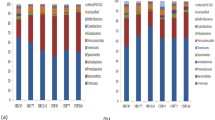
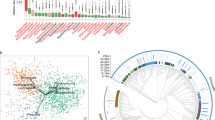
Of the 104 small molecule metabolites meeting analysis criteria, we found that 84 (81%) were differentially abundant based on gut region. The most significant regional comparison observed was between the cecum and small intestines, with 52 (50%) differentially abundant metabolites. Pregnancy itself altered 41 (39.4%) fecal small molecule metabolites.
Conclusions
The regional variation observed in the gut metabolome are likely due to the microbial and physiological differences between the different parts of the intestines. Additionally, pregnancy impacts the fecal metabolome, which may be due to evolving needs of both the dam and fetus.



Similar content being viewed by others
Data availability
Data used for analysis can be found at https://data.gov.
6. References
Ahmad, T. R., & Haeusler, R. A. (2019). Bile acids in glucose metabolism and insulin signalling – Mechanisms and research needs. Nature Reviews Endocrinology, 15, 701–712. https://doi.org/10.1038/s41574-019-0266-7
Bardócz, S., Grant, G., Brown, D. S., & Pusztai, A. (1998). Putrescine as a source of instant energy in the small intestine of the rat. Gut, 42(1), 24–28. https://doi.org/10.1136/gut.42.1.24
Bernstein, H., Bernstein, C., Payne, C. M., Dvorakova, K., & Garewal, H. (2005). Bile acids as carcinogens in human gastrointestinal cancers. Mutation Research, 589(1), 47–65. https://doi.org/10.1016/j.mrrev.2004.08.001
Berstad, A., Raa, J., & Valeur, J. (2015). Indole – the scent of a healthy ‘inner soil.’ Microbial Ecology in Health and Disease, 14(26), 27997. https://doi.org/10.3402/mehd.v26.27997
Brooks, M. E., Kristensen, K., van Benthem, K. J., Magnusson, A., Berg, C. W., Nielsen, A., Skaug, H. J., Maechler, M., & Bolker, B. M. (2017). glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. The R Journal, 9(2), 378–400. https://doi.org/10.32614/RJ-2017-066
Brown, K., Thomson, C. A., Wacker, S., Drikic, M., Groves, R., Fan, V., Lewis, I. A., & McCoy, K. D. (2023). Microbiota alters the metabolome in an age- and sex-dependent manner in mice. Nature Communications, 14, 1348. https://doi.org/10.1038/s41467-023-37055-1
Cai, H., Wen, Z., Meng, K., & Yang, P. (2021). Metabolomic signatures for liver tissue and cecum contents in high-fat diet-induced obese mice based on UHPLC-Q-TOF/MS. Nutrition and Metabolism, 18(1), 69. https://doi.org/10.1186/s12986-021-00595-8
Carmody, R. N., Gerber, G. K., Luevano, J. M., Jr., Gatti, D. M., Somes, L., Svenson, K. L., & Turnbaugh, P. J. (2015). Diet dominates host genotype in shaping the murine gut microbiota. Cell Host & Microbe, 17(1), 72–84. https://doi.org/10.1016/j.chom.2014.11.010
Chen, L., Zhong, Y., Ouyang, X., Wang, C., Yin, L., Huang, J., Li, Y., Wang, Q., Xie, J., Huang, P., Yang, H., & Yin, Y. (2022). Effects of β-alanine on intestinal development and immune performance of weaned piglets. Animal Nutrition (zhongguo Xu Mu Shou Yi Xue Hui), 12, 398–408. https://doi.org/10.1016/j.aninu.2022.10.008
Collins JT, Nguyen A, & Badireddy M. Anatomy, abdomen and pelvis, small intestine. [Updated 2022 Aug 8]. In: StatPearls [Internet]. StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459366/
Creamer, B. (1967). The turnover of the epithelium of the small intestine. British Medical Bulletin, 23(3), 226–230. https://doi.org/10.1093/oxfordjournals.bmb.a070561
David, L. A., Maurice, C. F., Carmody, R. N., Gootenberg, D. B., Button, J. E., Wolfe, B. E., Ling, A. V., Devlin, A. S., Varma, Y., Fischbach, M. A., Biddinger, S. B., Dutton, R. J., & Turnbaugh, P. J. (2014). Diet rapidly and reproducibly alters the human gut microbiome. Nature, 505(7484), 559–563. https://doi.org/10.1038/nature12820
Diaz Heijtz, R., Wang, S., Anuar, F., Qian, Y., Björkholm, B., Samuelsson, A., Hibberd, M. L., Forssberg, H., & Pettersson, S. (2011). Normal gut microbiota modulates brain development and behavior. Proceedings of the National Academy of Sciences of the United States of America, 108(7), 3047–3052. https://doi.org/10.1073/pnas.1010529108
Duca, F. A., Waise, T. M. Z., Peppler, W. T., & Lam, T. K. T. (2021). The metabolic impact of small intestinal nutrient sensing. Nature Communications, 12(1), 903. https://doi.org/10.1038/s41467-021-21235-y
El Aidy, S., Merrifield, C. A., Derrien, M., van Baarlen, P., Hooiveld, G., Levenez, F., Doré, J., Dekker, J., Holmes, E., Claus, S. P., Reijngoud, D. J., & Kleerebezem, M. (2013). The gut microbiota elicits a profound metabolic reorientation in the mouse jejunal mucosa during conventionalisation. Gut, 62(9), 1306–1314. https://doi.org/10.1136/gutjnl-2011-301955
Elango, R., & Ball, R. O. (2016). Protein and amino acid requirements during pregnancy. Advances in Nutrition (Bethesda, Md.), 7(4), 839S–844S. https://doi.org/10.3945/an.115.011817
Fan, Y., & Pedersen, O. (2021). Gut microbiota in human metabolic health and disease. Nature Reviews. Microbiology, 19(1), 55–71. https://doi.org/10.1038/s41579-020-0433-9
Flint, H. J., Scott, K. P., Louis, P., & Duncan, S. H. (2012). The role of the gut microbiota in nutrition and health. Nature Reviews. Gastroenterology & Hepatology, 9(10), 577–589. https://doi.org/10.1038/nrgastro.2012.156
Friedman, E. S., Bittinger, K., Esipova, T. V., Hou, L., Chau, L., Jiang, J., Mesaros, C., Lund, P. J., Liang, X., FitzGerald, G. A., Goulian, M., Lee, D., Garcia, B. A., Blair, I. A., Vinogradov, S. A., & Wu, G. D. (2018). Microbes vs. chemistry in the origin of the anaerobic gut lumen. Proceedings of the National Academy of Sciences of the United States of America, 115(16), 4170–4175. https://doi.org/10.1073/pnas.1718635115
Geboes, K., Geboes, K. P., & Maleux, G. (2001). Vascular anatomy of the gastrointestinal tract. Best Practice & Research. Clinical Gastroenterology, 15(1), 1–14. https://doi.org/10.1053/bega.2000.0152
Gremel, G., Wanders, A., Cedernaes, J., Fagerberg, L., Hallstrom, B., Edlund, K., Sjostedt, E., Uhlen, M., & Ponten, F. (2015). The human gastrointestinal tract-specific transcriptome and proteome as defined by RNA sequencing and antibody-based profiling. Journal of Gastroenterology, 50(1), 46–57. https://doi.org/10.1007/s00535-014-0958-7
Gruner, N., & Mattner, J. (2021). Bile acids and microbiota: Multifaceted and versatile regulators of the liver-gut axis. International Journal of Molecular Sciences, 22(3), 1397. https://doi.org/10.3390/ijms22031397
Gu, S., Chen, D., Zhang, J. N., Lv, X., Wang, K., Duan, L. P., Nie, Y., & Wu, X. L. (2013). Bacterial community mapping of the mouse gastrointestinal tract. PLoS ONE, 8(10), e74957. https://doi.org/10.1371/journal.pone.0074957
Hamamah, S., Aghazarian, A., Nazaryan, A., Hajnal, A., & Covasa, M. (2022). Role of microbiota-gut-brain axis in regulating dopaminergic signaling. Biomedicines, 10(2), 436. https://doi.org/10.3390/biomedicines10020436
Hashash, J. G., & Kane, S. (2015). Pregnancy and inflammatory bowel disease. Gastroenterology and Hepatology, 11(2), 96–102.
Hendrikx, T., & Schnabl, B. (2019). Indoles: Metabolites produced by intestinal bacteria capable of controlling liver disease manifestation. Journal of Internal Medicine, 286(1), 32–40. https://doi.org/10.1111/joim.12892
Herrera, E. (2002). Lipid metabolism in pregnancy and its consequences in the fetus and newborn. Endocrine, 19(1), 43–55. https://doi.org/10.1385/ENDO:19:1:43
Hugenholtz, F., & de Vos, W. M. (2018). Mouse models for human intestinal microbiota research: A critical evaluation. Cellular and Molecular Life Sciences: CMLS, 75(1), 149–160. https://doi.org/10.1007/s00018-017-2693-8
Jääskeläinen, T., Kärkkäinen, O., Jokkala, J., et al. (2021). A non-targeted LC–MS metabolic profiling of pregnancy: Longitudinal evidence from healthy and pre-eclamptic pregnancies. Metabolomics, 17, 20. https://doi.org/10.1007/s11306-020-01752-5
Johansson, M. E., Ambort, D., Pelaseyed, T., Schütte, A., Gustafsson, J. K., Ermund, A., Subramani, D. B., Holmén-Larsson, J. M., Thomsson, K. A., Bergström, J. H., van der Post, S., Rodriguez-Piñeiro, A. M., Sjövall, H., Bäckström, M., & Hansson, G. C. (2011). Composition and functional role of the mucus layers in the intestine. Cellular and Molecular Life Sciences: CMLS, 68(22), 3635–3641. https://doi.org/10.1007/s00018-011-0822-3
Kiela, P. R., & Ghishan, F. K. (2016). Physiology of intestinal absorption and secretion. Best Practice & Research. Clinical Gastroenterology, 30(2), 145–159. https://doi.org/10.1016/j.bpg.2016.02.007
Kimura, I., Miyamoto, J., Ohue-Kitano, R., Watanabe, K., Yamada, T., Onuki, M., Aoki, R., Isobe, Y., Kashihara, D., Inoue, D., Inaba, A., Takamura, Y., Taira, S., Kumaki, S., Watanabe, M., Ito, M., Nakagawa, F., Irie, J., Kakuta, H., Shinohara, M., et al. (2020). Maternal gut microbiota in pregnancy influences offspring metabolic phenotype in mice. Science (New York, N.Y.), 367(6481), eaaw8429. https://doi.org/10.1126/science.aaw8429
Koren, O., Goodrich, J. K., Cullender, T. C., Spor, A., Laitinen, K., Bäckhed, H. K., Gonzalez, A., Werner, J. J., Angenent, L. T., Knight, R., Bäckhed, F., Isolauri, E., Salminen, S., & Ley, R. E. (2012). Host remodeling of the gut microbiome and metabolic changes during pregnancy. Cell, 150(3), 470–480. https://doi.org/10.1016/j.cell.2012.07.008
Kyozuka, H., Fukuda, T., Murata, T., Endo, Y., Kanno, A., Yasuda, S., Yamaguchi, A., Ono, M., Sato, A., Hashimoto, K., & Fujimori, K. (2020). Comprehensive metabolomic analysis of first-trimester serum identifies biomarkers of early-onset hypertensive disorder of pregnancy. Scientific Reports, 10(1), 13857. https://doi.org/10.1038/s41598-020-70974-3
Lain, K. Y., & Catalano, P. M. (2007). Metabolic changes in pregnancy. Clinical Obstetrics and Gynecology, 50(4), 938–948. https://doi.org/10.1097/GRF.0b013e31815a5494
Lenth, R. V. (2023). emmeans: Estimated Marginal Means, aka Least-Squares Means. R package version 1.8.5. https://CRAN.R-project.org/package=emmeans
Liang, S., Hou, Z., Li, X., Wang, J., Cai, L., Zhang, R., & Li, J. (2019). The fecal metabolome is associated with gestational diabetes mellitus. RSC Advances, 9(51), 29973–29979. https://doi.org/10.1039/c9ra05569j
Liang, X., Wang, R., Luo, H., Liao, Y., Chen, X., Xiao, X., & Li, L. (2022). The interplay between the gut microbiota and metabolism during the third trimester of pregnancy. Frontiers in Microbiology, 13, 1059227. https://doi.org/10.3389/fmicb.2022.1059227
Lin, K., Zhu, L., Yang, L. (2022). Gut and obesity/metabolic disease: Focus on microbiota metabolites. MedComm, 3(3), e171. 10.1002%2Fmco2.171
Liu, B., Jiang, X., Cai, L., Zhao, X., Dai, Z., Wu, G., & Li, X. (2019). Putrescine mitigates intestinal atrophy through suppressing inflammatory response in weanling piglets. Journal of Animal Science and Biotechnology, 10, 69. https://doi.org/10.1186/s40104-019-0379-9
Liu, C., Wang, Y., Zheng, W., Wang, J., Zhang, Y., Song, W., Wang, A., Ma, X., & Li, G. (2021). Putrescine as a novel biomarker of maternal serum in first trimester for the prediction of gestational diabetes mellitus: A nested case-control study. Frontiers in Endocrinology, 12, 759893. https://doi.org/10.3389/fendo.2021.759893
Löser, C. (2000). Polyamines in human and animal milk. The British Journal of Nutrition, 84(Suppl 1), S55–S58. https://doi.org/10.1017/s0007114500002257
Martinez-Guryn, K., Leone, V., & Chang, E. B. (2019). Regional diversity of the gastrointestinal microbiome. Cell Host & Microbe, 26(3), 314–324. https://doi.org/10.1016/j.chom.2019.08.011
Mawe, G. M., & Hoffman, J. M. (2013). Serotonin signalling in the gut–functions, dysfunctions and therapeutic targets. Nature Reviews. Gastroenterology & Hepatology, 10(8), 473–486. https://doi.org/10.1038/nrgastro.2013.105
McConnell, E. L., Basit, A. W., & Murdan, S. (2008). Measurements of rat and mouse gastrointestinal pH, fluid and lymphoid tissue, and implications for in-vivo experiments. The Journal of Pharmacy and Pharmacology, 60(1), 63–70. https://doi.org/10.1211/jpp.60.1.0008
Meier, K. H. U., Trouillon, J., Li, H., Lang, M., Fuhrer, T., Zamboni, N., Sunagawa, S., Macpherson, A. J., & Sauer, U. (2023). Nature Metabolism, 5, 968–980. https://doi.org/10.1038/s42255-023-00802-1
Mueller, N. T., Bakacs, E., Combellick, J., Grigoryan, Z., & Dominguez-Bello, M. G. (2015). The infant microbiome development: Mom matters. Trends in Molecular Medicine, 21(2), 109–117. https://doi.org/10.1016/j.molmed.2014.12.002
Nguyen, T. L., Vieira-Silva, S., Liston, A., & Raes, J. (2015). How informative is the mouse for human gut microbiota research? Disease Models & Mechanisms, 8(1), 1–16. https://doi.org/10.1242/dmm.017400
Noah, T. K., Donahue, B., & Shroyer, N. F. (2011). Intestinal development and differentiation. Experimental Cell Research, 317(19), 2702–2710. https://doi.org/10.1016/j.yexcr.2011.09.006
O’Hara, A. M., & Shanahan, F. (2006). The gut flora as a forgotten organ. EMBO Reports, 7(7), 688–693. https://doi.org/10.1038/sj.embor.7400731
Overmyer, K. A., Thonusin, C., Qi, N. R., Burant, C. F., & Evans, C. R. (2015). Impact of anesthesia and euthanasia on metabolomics of mammalian tissues: Studies in a C57BL/6J mouse model. PLoS ONE, 10(2), e0117232. 10.1371%2Fjournal.pone.0117232
Palau-Rodriguez, M., Tulipani, S., Isabel Queipo-Ortuño, M., Urpi-Sarda, M., Tinahones, F. J., & Andres- Lacueva, C. (2015). Metabolomic insights into the intricate gut microbial-host interaction in the development of obesity and type 2 diabetes. Frontiers in Microbiology, 6, 1151. https://doi.org/10.3389/fmicb.2015.01151
Parrettini, S., Caroli, A., & Torlone, E. (2020). Nutrition and metabolic adaptations in physiological and complicated pregnancy: Focus on obesity and gestational diabetes. Frontiers in Endocrinology, 11, 611929. 10.3389%2Ffendo.2020.611929
Pessa-Morikawa, T., Husso, A., Karkkainen, O., Koistinen, V., Hanhineva, K., Iivananinen, A., & Niku, M. (2022). Maternal microbiota-derived metabolic profile in fetal murine intestine, brain and placenta. BMC Microbiology, 22(1), 46. https://doi.org/10.1186/s12866-022-02457-6
Russell, D. H., Giles, H. R., Christian, C. D., & Campbell, J. L. (1978). Polyamines in amniotic fluid, plasma, and urine during normal pregnancy. American Journal of Obstetrics and Gynecology, 132(6), 649–652. https://doi.org/10.1016/0002-9378(78)90858-x
Scholtens, P. A., Oozeer, R., Martin, R., Amor, K. B., & Knol, J. (2012). The early settlers: Intestinal microbiology in early life. Annual Review of Food Science and Technology, 3, 425–447. https://doi.org/10.1146/annurev-food-022811-101120
Sommersberger, S., Gunawan, S., Elger, T., Fererberger, T., Loibl, J., Huss, M., Kandulski, A., Krautbauer, S., Muller, M., Liebisch, G., Buechler, C., & Tews, H. C. (2023). Altered fecal bile acid composition in active ulcerative colitis. Lipids in Health and Disease, 22(1), 199. https://doi.org/10.1186/s12944-023-01971-4
Takahashi, H., Suzuki, Y., Nishimura, J., Haraguchi, N., Ohtuska, M., Miyazaki, S., Uemura, M., Hata, T., Takemasa, I., Mizushima, T., Yamamoto, H., Doki, Y., & Mori, M. (2016). Characteristics of carbonic anhydrase 9 expression cells in the human intestinal crypt base. International Journal of Oncology, 48(1), 115–122. https://doi.org/10.3892/ijo.2015.3260
Thaiss, C. A., Levy, M., Korem, T., Dohnalová, L., Shapiro, H., Jaitin, D. A., David, E., Winter, D. R., Gury- BenAri, M., Tatirovsky, E., Tuganbaev, T., Federici, S., Zmora, N., Zeevi, D., Dori-Bachash, M., Pevsner- Fischer, M., Kartvelishvily, E., Brandis, A., Harmelin, A., Shibolet, O., et al. (2016). Microbiota diurnal rhythmicity programs host transcriptome oscillations. Cell, 167(6), 1495–1510. https://doi.org/10.1016/j.cell.2016.11.003
Thaiss, C. A., Zeevi, D., Levy, M., Segal, E., & Elinav, E. (2015). A day in the life of the meta-organism: Diurnal rhythms of the intestinal microbiome and its host. Gut Microbes, 6(2), 137–142. https://doi.org/10.1080/19490976.2015.1016690
Turnbaugh, P. J., Ley, R. E., Mahowald, M. A., Magrini, V., Mardis, E. R., & Gordon, J. I. (2006). An obesity- associated gut microbiome with increased capacity for energy harvest. Nature, 444(7122), 1027–1031. https://doi.org/10.1038/nature05414
van Buuren, S., & Groothuis-Oudshoorn, K. (2011). mice: multivariate imputation by chained equations in R. Journal of Statistical Software, 45(3), 1–67. https://doi.org/10.18637/jss.v045.i03
Warnes, G.R., Bolker, B., Bonebakker, L., Gentleman, R., Huber, W., Liaw, A., Lumley, T., Maechler, M., Magnusson, A., Moeller, S., Schwartz, M., & Venables, B. (2022). gplots: Various R Programming Tools for Plotting Data. R package version 3.1.3. https://CRAN.R-project.org/package=gplots
Weger, B. D., Gobet, C., Yeung, J., Martin, E., Jimenez, S., Betrisey, B., Foata, F., Berger, B., Balvay, A., Foussier, A., Charpagne, A., Boizet-Bonhoure, B., Chou, C. J., Naef, F., & Gachon, F. (2019). The mouse microbiome is required for sex-specific diurnal rhythms of gene expression and metabolism. Cell Metabolism, 29(2), 362–382. https://doi.org/10.1016/j.cmet.2018.09.023
Wu, G. D., Chen, J., Hoffmann, C., Bittinger, K., Chen, Y. Y., Keilbaugh, S. A., Bewtra, M., Knights, D., Walters, W. A., Knight, R., Sinha, R., Gilroy, E., Gupta, K., Baldassano, R., Nessel, L., Li, H., Bushman, F. D., & Lewis, J. D. (2011). Linking long-term dietary patterns with gut microbial enterotypes. Science (New York, N.Y.), 334(6052), 105–108. https://doi.org/10.1126/science.1208344
Wu, G., Bazer, F. W., Satterfield, M. C., Li, X., Wang, X., Johnson, G. A., Burghardt, R. C., Dai, Z., Wang, J., & Wu, Z. (2013). Impacts of arginine nutrition on embryonic and fetal development in mammals. Amino Acids, 45(2), 241–256. https://doi.org/10.1007/s00726-013-1515-z
Xu, T. T., Li, H., Dai, Z., Lau, G. K., Li, B. Y., Zhu, W. L., Liu, X. Q., Liu, H. F., Cai, W. W., Huang, S. Q., Wang, Q., & Zhang, S. J. (2020). Spermidine and spermine delay brain aging by inducing autophagy in SAMP8 mice. Aging, 12(7), 6401–6414. https://doi.org/10.18632/aging.103035
Zeisel, S.H. (2008). Choline: Critical role during fetal development and dietary requirements in adults. Annual Review of Nutrition, 26, 229–250. 10.1146%2Fannurev.nutr.26.061505.111156
Zeng, H., Umar, S., Rust, B., Lazarova, D., & Bordonaro, M. (2019). Secondary bile acids and short chain fatty acids in the colon: A focus on colonic microbiome, cell proliferation, inflammation, and cancer. International Journal of Molecular Sciences, 20(5), 1214. https://doi.org/10.3390/ijms20051214
Zhai, L., Xiao, H., Lin, C., Wong, H. L. X., Lam, Y. Y., Gong, M., Wu, G., Ning, Z., Huang, C., Zhang, Y., Yang, C., Luo, J., Zhang, L., Zhao, L., Zhang, C., Lau, J. Y., Lu, A., Lau, L. T., Jia, W., Zhao, L., et al. (2023). Gut microbiota-derived tryptamine and phenethylamine impair insulin sensitivity in metabolic syndrome and irritable bowel syndrome. Nature Communications, 14(1), 4986. https://doi.org/10.1038/s41467-023-40552-y
Zhang, K., Liu, G., Wu, Y., Zhang, T., Guo, M., Lei, Y., Cao, X., Suo, X., Brugger, D., Wang, X., Yang, Y., & Chen, Y. (2023). Gut microbial succession patterns and metabolic profiling during pregnancy and lactation in a goat model. Microbiology Spectrum, 11(1). https://doi.org/10.1128/spectrum.02955-22
Zhao, M., Xiong, X., Ren, K., Xu, B., Cheng, M., Sahu, C., Wu, K., Nie, Y., Huang, Z., Blumberg, R. S., Han, X., & Ruan, H. B. (2018). Deficiency in intestinal epithelial O-GlcNAcylation predisposes to gut inflammation. EMBO Molecular Medicine, 10(8), e8736. https://doi.org/10.15252/emmm.201708736
Zhao, C., Ge, J., Li, X., Jiao, R., Li, Y., Quan, H., Li, J., Guo, Q., & Wang, W. (2020). Integrated metabolome analysis reveals novel connections between maternal fecal metabolome and the neonatal blood metabolome in women with gestational diabetes mellitus. Scientific Reports, 10, 3660. https://doi.org/10.1038/s41598-020-60540-2
Zhao, M., Ren, K., Xiong, X., Cheng, M., Zhang, Z., Huang, Z., Han, X., Yang, X., Alejandro, E. U., & Ruan, H. B. (2020). Protein O-GlcNac modification links dietary and gut microbial cues to the differentiation of enteroendocrine L cells. Cell Reports, 32(6), 108013. https://doi.org/10.1016/j.celrep.2020.108013
Zheng, D., Liwinski, T., & Elinav, E. (2020). Interaction between microbiota and immunity in health and disease. Cell Research, 30(6), 492–506. https://doi.org/10.1038/s41422-020-0332-7
Acknowledgements
The authors thank Drs. Justin Conley, Ian Gilmour, Christopher Lau, and Katie O’Shaughnessy for their careful review of this manuscript.
Funding
This study was funded by the Sustainable and Healthy Communities Research Program in the Office of Research and Development at the U.S. Environmental Protection Agency. This research was also supported in part by an appointment to the Research Participation Program at the Center for Public Health and Environmental Assessment at the U.S. EPA administered by the Oak Ridge Institute for Science and Education through an interagency agreement between the U.S. Department of Energy and the U.S. EPA.
Author information
Authors and Affiliations
Contributions
MM, JD, MS, and CM conceived and contributed to the design of the experiment. MM prepared the samples and JF conducted the metabolomics. TJ and MM analyzed data. MM wrote the manuscript. All authors participated in the review and revision of the manuscript and approved the final version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Disclaimer
The views expressed in this article are those of the authors and do not necessarily represent the views or policies of the U.S. EPA nor the U.S. government. The mention of trade names or commercial products does not constitute endorsement or recommendation for use.
Animal usage/safety statement
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Moore, M.L., Ford, J.L., Schladweiler, M.C. et al. Gut metabolic changes during pregnancy reveal the importance of gastrointestinal region in sample collection. Metabolomics 20, 40 (2024). https://doi.org/10.1007/s11306-024-02099-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-024-02099-x