Abstract
Introduction
Acute ischemic stroke (AIS) accounts for the majority of all stroke, globally the second leading cause of death. Due to its rapid development after onset, its early diagnosis is crucial.
Objectives
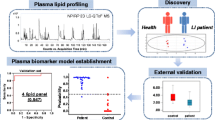
We aim to identify potential highly reliable blood-based biomarkers for early diagnosis of AIS using quantitative plasma lipid profiling via a machine learning approach.
Methods
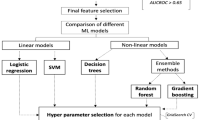
Lipidomics was used for quantitative plasma lipid profiling, based on ultra-performance liquid chromatography tandem mass spectrometry. Our samples were divided into a discovery and a validation set, each containing 30 AIS patients and 30 health controls (HC). Differentially expressed lipid metabolites were screened based on the criteria VIP > 1, p < 0.05, and fold change > 1.5 or < 0.67. The least absolute shrinkage and selection operator (LASSO) and random forest algorithms in machine learning were used to select differential lipid metabolites as potential biomarkers.
Results
Three key differential lipid metabolites, CarnitineC10:1, CarnitineC10:1-OH and Cer(d18:0/16:0), were identified as potential biomarkers for early diagnosis of AIS. The former two, associated with thermogenesis, were down-regulated, whereas the latter, associated with necroptosis and sphingolipd metabolism, was upregulated. Univariate and multivariate logistic regressions showed that these three lipid metabolites and the resulting diagnostic model exhibited a strong ability in discriminating between AIS patients and HCs in both the discovery and validation sets, with an area under the curve above 0.9.
Conclusions
Our work provides valuable information on the pathophysiology of AIS and constitutes an important step toward clinical application of blood-based biomarkers for diagnosing AIS.




Similar content being viewed by others
Data Availability
The original contributions presented in the study are included in the article. Data not provided in the article because of space limitations may be shared (anonymized) at the request of any qualified investigator for purposes of replicating procedures and results. Further inquiries can be directed to the corresponding author.
Abbreviations
- AIS:
-
Acute ischemic stroke
- AMI:
-
Acute myocardial infarction
- AUC:
-
Area under the curve
- CAR:
-
Carnitine
- Cer:
-
Ceramide
- CerP:
-
Ceramide 1-phosphate
- DA:
-
Differential abundance
- ESI:
-
Electrospray ionization
- FC:
-
Fold change
- FFA:
-
Free fatty acid
- GPI:
-
Glycosylphosphatidylinositol
- HC:
-
Healthy control
- HDL-C:
-
High-density lipoprotein cholesterol
- ICH:
-
Intracerebral haemorrhage
- KEGG:
-
Kyoto Encyclopedia of genes and genomes
- LASSO:
-
Least absolute shrinkage and selection operator
- LDL-C:
-
Low density lipoprotein cholesterol
- MS/MS:
-
Tandem mass spectrometry
- OPLS-DA:
-
Orthogonal partial least squares discriminant analysis
- PA:
-
Phosphatidic acid
- PC:
-
Phosphatidylcholine
- PCA:
-
Principal component analysis
- PE:
-
Phosphatidyl ethanolamine
- PE-O:
-
Phosphatidylethanolamine-ethers
- RF:
-
Random forest
- ROC:
-
Receiver operating characteristic
- TC:
-
Total cholesterol
- TG:
-
Triglycerides
- UPLC:
-
Ultra-performance liquid chromatography
- VIP:
-
Variable importance in projection
References
American_Stroke_Association. (2017). ACUTE ISCHEMIC STROKE -- Current Treatment Approaches for Acute Ischemic Stroke. https://www.stroke.org/-/media/Stroke-Files/Ischemic-Stroke-Professional-Materials/AIS-Toolkit/AIS-Professional-Education-Presentation-ucm_485538
Au, A. (2018). Metabolomics and Lipidomics of Ischemic Stroke. Advances in clinical chemistry (Vol. 85, pp. 31–69). Elsevier.
Benjamin, E. J., Muntner, P., Alonso, A., Bittencourt, M. S., Callaway, C. W., Carson, A. P., et al. (2019). Heart disease and stroke statistics—2019 update: A report from the american heart association. Circulation, 139(10), e56–e528. https://doi.org/10.1161/CIR.0000000000000659
Breiman, L. (2001). Random forests. Machine Learning, 45(1), 5–32. https://doi.org/10.1023/A:1010933404324
Bujak, R., Struck-Lewicka, W., Markuszewski, M. J., & Kaliszan, R. (2015). Metabolomics for laboratory diagnostics. Journal of Pharmaceutical and Biomedical Analysis, 113, 108–120. https://doi.org/10.1016/j.jpba.2014.12.017
Bustamante, A., López-Cancio, E., Pich, S., Penalba, A., Giralt, D., García-Berrocoso, T., et al. (2017). Blood biomarkers for the early diagnosis of stroke. Stroke, 48(9), 2419–2425. https://doi.org/10.1161/STROKEAHA.117.017076
Cao, B., Chen, Y., McIntyre, R. S., & Yan, L. (2020). Acyl-Carnitine plasma levels and their association with metabolic syndrome in individuals with schizophrenia. Psychiatry Research, 293, 113458. https://doi.org/10.1016/j.psychres.2020.113458
Datorre, S. (1991). Amphipathic lipid metabolites and their relation to arrhythmogenesis in the ischemic heart. Journal of Molecular and Cellular Cardiology, 23(SUPPL. 1), 11–22. https://doi.org/10.1016/0022-2828(91)90019-I
Dong, M.-X., Hu, L., Wei, Y.-D., & Chen, G.-H. (2021). Metabolomics profiling reveals altered lipid metabolism and identifies a panel of lipid metabolites as biomarkers for Parkinson’s disease related anxiety disorder. Neuroscience Letters, 745, 135626. https://doi.org/10.1016/j.neulet.2021.135626
Fiedorowicz, A., Kozak-Sykała, A., Bobak, Ł, Kałas, W., & Strządała, L. (2019). Ceramides and sphingosine-1-phosphate as potential markers in diagnosis of ischaemic stroke. Neurologia i Neurochirurgia Polska, 53(6), 484–491. https://doi.org/10.5603/PJNNS.a2019.0063
Giles, C., Takechi, R., Lam, V., Dhaliwal, S. S., & Mamo, J. C. L. (2018). Contemporary lipidomic analytics: Opportunities and pitfalls. Progress in Lipid Research, 71, 86–100. https://doi.org/10.1016/j.plipres.2018.06.003
Huynh, K., Barlow, C. K., Jayawardana, K. S., Weir, J. M., Mellett, N. A., Cinel, M., et al. (2019). High-Throughput plasma lipidomics: Detailed mapping of the associations with cardiometabolic risk factors. Cell Chemical Biology, 26(1), 71-84.e4. https://doi.org/10.1016/j.chembiol.2018.10.008
Ighodaro, E. T., Graff-Radford, J., Syrjanen, J. A., Bui, H. H., Petersen, R. C., Knopman, D. S., et al. (2020). Associations between plasma ceramides and cerebral microbleeds or lacunes. Arteriosclerosis, Thrombosis, and Vascular Biology, 40, 2785–2793. https://doi.org/10.1161/ATVBAHA.120.314796
Ke, C., Pan, C.-W., Zhang, Y., Zhu, X., & Zhang, Y. (2019). Metabolomics facilitates the discovery of metabolic biomarkers and pathways for ischemic stroke: A systematic review. Metabolomics, 15(12), 152. https://doi.org/10.1007/s11306-019-1615-1
Kurz, J., Parnham, M. J., Geisslinger, G., & Schiffmann, S. (2019). Ceramides as novel disease biomarkers. Trends in Molecular Medicine, 25(1), 20–32. https://doi.org/10.1016/j.molmed.2018.10.009
Liepinsh, E., Makrecka-Kuka, M., Volska, K., Kuka, J., Makarova, E., Antone, U., et al. (2016). Long-chain acylcarnitines determine ischaemia/reperfusion-induced damage in heart mitochondria. Biochemical Journal, 473(9), 1191–1202. https://doi.org/10.1042/BCJ20160164
Liu, J., Tang, L., Lu, Q., Yu, Y., Xu, Q.-G., Zhang, S., et al. (2022). Plasma quantitative lipid profiles: Identification of Carnitine C18:1-OH, CarnitineC18:2-OH and FFA (20:1) as novel biomarkers for pre-warning and prognosis in acute myocardial infarction. Frontiers in Cardiovascular Medicine, 9, 734. https://doi.org/10.3389/fcvm.2022.848840
Liu, P., Li, R., Antonov, A. A., Wang, L., Li, W., Hua, Y., et al. (2017). Discovery of metabolite biomarkers for acute ischemic stroke progression. Journal of Proteome Research, 16(2), 773–779. https://doi.org/10.1021/acs.jproteome.6b00779
Montaner, J., Ramiro, L., Simats, A., Tiedt, S., Makris, K., Jickling, G. C., et al. (2020). Multilevel omics for the discovery of biomarkers and therapeutic targets for stroke. Nature Reviews Neurology, 16(5), 247–264. https://doi.org/10.1038/s41582-020-0350-6
Ouro, A., Correa-Paz, C., Maqueda, E., Custodia, A., Aramburu-Núñez, M., Romaus-Sanjurjo, D., et al. (2022). Involvement of ceramide metabolism in cerebral ischemia. Frontiers in Molecular Biosciences, 9, 309. https://doi.org/10.3389/fmolb.2022.864618
Park, J. Y., Lee, S.-H., Shin, M.-J., & Hwang, G.-S. (2015). Alteration in metabolic signature and lipid metabolism in patients with angina pectoris and myocardial infarction. PLoS ONE, 10(8), e0135228. https://doi.org/10.1371/journal.pone.0135228
Seo, W.-K., Jo, G., Shin, M.-J., & Oh, K. (2018). Medium-chain acylcarnitines are associated with cardioembolic stroke and stroke recurrence. Arteriosclerosis, Thrombosis, and Vascular Biology, 38(9), 2245–2253. https://doi.org/10.1161/ATVBAHA.118.311373
Sheth, S. A., Iavarone, A. T., Liebeskind, D. S., Won, S. J., & Swanson, R. A. (2015). Targeted lipid profiling discovers plasma biomarkers of acute brain injury. PLoS ONE, 10(6), e0129735. https://doi.org/10.1371/journal.pone.0129735
Shin, T. H., Lee, D. Y., Basith, S., Manavalan, B., Paik, M. J., Rybinnik, I., et al. (2020). Metabolome changes in cerebral ischemia. Cells, 9(7), 1630. https://doi.org/10.3390/cells9071630
Sun, D., Tiedt, S., Yu, B., Jian, X., Gottesman, R. F., Mosley, T. H., et al. (2019a). A prospective study of serum metabolites and risk of ischemic stroke. Neurology, 92(16), e1890–e1898. https://doi.org/10.1212/WNL.0000000000007279
Sun, L., Clarke, R., Bennett, D., Guo, Y., Walters, R. G., Hill, M., et al. (2019b). Causal associations of blood lipids with risk of ischemic stroke and intracerebral hemorrhage in Chinese adults. Nature Medicine, 25(4), 569–574. https://doi.org/10.1038/s41591-019-0366-x
Sun, N., Keep, R. F., Hua, Y., & Xi, G. (2016). Critical role of the sphingolipid pathway in stroke: A review of current utility and potential therapeutic targets. Translational Stroke Research, 7(5), 420–438. https://doi.org/10.1007/S12975-016-0477-3
Sun, R., Li, Y., Cai, M., Cao, Y., & Piao, X. (2019c). Discovery of a new biomarker pattern for differential diagnosis of acute ischemic stroke using targeted metabolomics. Frontiers in Neurology, 10, 1–7. https://doi.org/10.3389/fneur.2019.01011
Takagi, A., Hawke, P., Tokuda, S., Toda, T., Higashizono, K., Nagai, E., et al. (2022). Serum carnitine as a biomarker of sarcopenia and nutritional status in preoperative gastrointestinal cancer patients. Journal of Cachexia, Sarcopenia and Muscle, 13(1), 287–295. https://doi.org/10.1002/jcsm.12906
Tibshirani, R. (1997). The LASSO method for variable selection in the Cox model. Statistics in Medicine, 16(4), 385–395. https://doi.org/10.1002/(SICI)1097-0258(19970228)16:4%3c385::AID-SIM380%3e3.0.CO;2-3
Tsugawa, H., Ikeda, K., Takahashi, M., Satoh, A., Mori, Y., Uchino, H., et al. (2020). A lipidome atlas in MS-DIAL 4. Nature Biotechnology, 38(10), 1159–1163. https://doi.org/10.1038/s41587-020-0531-2
Vasile, V. C., Meeusen, J. W., Medina Inojosa, J. R., Donato, L. J., Scott, C. G., Hyun, M. S., et al. (2021). Ceramide scores predict cardiovascular risk in the community. Arteriosclerosis, Thrombosis, and Vascular Biology, 41, 1558–1569. https://doi.org/10.1161/ATVBAHA.120.315530
Wang, Y.-J., Li, Z.-X., Gu, H.-Q., Zhai, Y., Jiang, Y., Zhao, X.-Q., et al. (2020). China stroke statistics 2019: A report from the national center for healthcare quality management in neurological diseases, china national clinical research center for neurological diseases, the chinese stroke association, national center for chronic and Stroke Collaborations. Stroke and Vascular Neurology, 5(3), 211–239. https://doi.org/10.1136/svn-2020-000457
WHO. (2020). The top 10 causes of death. https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death. Accessed 14 April 2022
Zafarullah, M., Palczewski, G., Rivera, S. M., Hessl, D. R., & Tassone, F. (2020). Metabolic profiling reveals dysregulated lipid metabolism and potential biomarkers associated with the development and progression of Fragile X-Associated Tremor/Ataxia Syndrome (FXTAS). The FASEB Journal, 34(12), 16676–16692. https://doi.org/10.1096/fj.202001880R
Acknowledgements
We thank Qiang Wang at Yangjiang People’s Hospital for his help in sample collection.
Funding
This work was financially supported in part by the Key Projects of Natural Science Foundation of Yangjiang People’s Hospital (Grant Nos. 10, 11, and 14).
Author information
Authors and Affiliations
Contributions
JCL conceived the study and oversaw the project, YY, XW, and JL drafted the manuscript. YY, JL, XW, HFL, SWL, and QGX participated in sample collection, data analysis and discussions. JCL and YY proofread and polished the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
All procedures followed in this study were in accordance with the ethical standards of the committee on human experimentation (Yangjiang People’s Hospital, China) and with the Helsinki Declaration of 1975, as revised in 2000(5). Written informed consent was obtained from all participants for being included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, Y., Wen, X., Lin, JG. et al. Identification of three potential novel biomarkers for early diagnosis of acute ischemic stroke via plasma lipidomics. Metabolomics 19, 32 (2023). https://doi.org/10.1007/s11306-023-01990-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-023-01990-3