Abstract
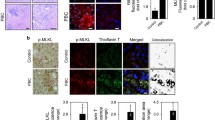
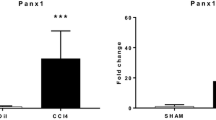
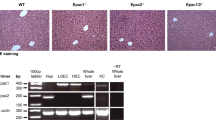
Ecto-nucleoside triphosphate diphosphohydrolases (E-NTPDases) are cell surface-located transmembrane ecto-enzymes of the CD39 superfamily which regulate inflammation and tissue repair by catalyzing the phosphohydrolysis of extracellular nucleotides and modulating purinergic signaling. In the liver, NTPDase2 is reportedly expressed on portal fibroblasts, but its functional role in regulating tissue regeneration and fibrosis is incompletely understood. Here, we studied the role of NTPDase2 in several models of liver injury using global knockout mice. Liver regeneration and severity of fibrosis were analyzed at different time points after exposure to carbon tetrachloride (CCl4) or 3,5-diethoxycarbonyl-1,4-dihydrocollidine (DDC) or partial hepatectomy in C57BL/6 wild-type and globally NTPDase2-deficient (Entpd2 null) mice. After chronic CCl4 intoxication, Entpd2 null mice exhibit significantly more severe liver fibrosis, as assessed by collagen content and histology. In contrast, deletion of NTPDase2 does not have a substantial effect on biliary-type fibrosis in the setting of DDC feeding. In injured livers, NTPDase2 expression extends from the portal areas to fibrotic septae in pan-lobular (CCl4-induced) liver fibrosis; the same pattern was observed, albeit to a lesser extent in biliary-type (DDC-induced) fibrosis. Liver regeneration after partial hepatectomy is not substantively impaired in global Entpd2 null mice. NTPDase2 protects from liver fibrosis resulting from hepatocellular injury induced by CCl4. In contrast, Entpd2 deletion does not significantly impact fibrosis secondary to DDC injury or liver regeneration after partial hepatectomy. Our observations highlight mechanisms relating to purinergic signaling in the liver and indicate possible therapeutic avenues and new cellular targets to test in the management of hepatic fibrosis.






Similar content being viewed by others
References
Dranoff JA, Wells RG (2010) Portal fibroblasts: underappreciated mediators of biliary fibrosis. Hepatology 51:1438–1444. https://doi.org/10.1002/hep.23405
Lu D, Insel PA (2014) Cellular mechanisms of tissue fibrosis. 6. Purinergic signaling and response in fibroblasts and tissue fibrosis. Am J Physiol Cell Physiol 306:88. https://doi.org/10.1152/ajpcell.00381.2013
Enjyoji K, Sévigny J, Lin Y, Frenette PS, Christie PD, Esch JS et al (1999) Targeted disruption of cd39/ATP diphosphohydrolase results in disordered hemostasis and thromboregulation. Nat Med 5:1010–1017. https://doi.org/10.1038/12447.
Deaglio S, Dwyer KM, Gao W, Friedman D, Usheva A, Erat A et al (2007) Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med 204:1257–1265. https://doi.org/10.1084/jem.20062512
Mascanfroni ID, Yeste A, Vieira SM, Burns EJ, Patel B, Sloma I et al (2013) IL-27 acts on DCs to suppress the T cell response and autoimmunity by inducing expression of the immunoregulatory molecule CD39. Nat Immunol 14:1054–1063. https://doi.org/10.1038/ni.2695
Beldi G, Wu Y, Sun X, Imai M, Enjyoji K, Csizmadia E et al (2008) Regulated catalysis of extracellular nucleotides by vascular CD39/ENTPD1 is required for liver regeneration. Gastroenterology 135:1751–1760. https://doi.org/10.1053/j.gastro.2008.07.025
Dranoff JA, Kruglov EA, Robson SC, Braun N, Zimmermann H, Sévigny J (2002) The ecto-nucleoside triphosphate diphosphohydrolase NTPDase2/CD39L1 is expressed in a novel functional compartment within the liver. Hepatology 36:1135–1144. https://doi.org/10.1053/jhep.2002.36823
Wink MR, Braganhol E, Tamajusuku ASK, Lenz G, Zerbini LF, Libermann TA et al (2006) Nucleoside triphosphate diphosphohydrolase-2 (NTPDase2/CD39L1) is the dominant ectonucleotidase expressed by rat astrocytes. Neuroscience 138:421–432. https://doi.org/10.1016/j.neuroscience.2005.11.039
Sévigny J, Sundberg C, Braun N, Guckelberger O, Csizmadia E, Qawi I et al (2002) Differential catalytic properties and vascular topography of murine nucleoside triphosphate diphosphohydrolase 1 (NTPDase1) and NTPDase2 have implications for thromboregulation. Blood 99:2801–2809
Heine P, Braun N, Heilbronn A, Zimmermann H (1999) Functional characterization of rat ecto-ATPase and ecto-ATP diphosphohydrolase after heterologous expression in CHO cells. Eur J Biochem 262:102–107
Jhandier MN, Kruglov EA, Lavoie EG, Sévigny J, Dranoff JA (2005) Portal fibroblasts regulate the proliferation of bile duct epithelia via expression of NTPDase2. J Biol Chem 280:22986–22992. https://doi.org/10.1074/jbc.M412371200
Iwaisako K, Jiang C, Zhang M, Cong M, Moore-Morris TJ, Park TJ et al (2014) Origin of myofibroblasts in the fibrotic liver in mice. Proc Natl Acad Sci U S A 111:E3297–E3305. https://doi.org/10.1073/pnas.1400062111.
Lua I, Li Y, Zagory JA, Wang KS, French SW, Sévigny J et al (2016) Characterization of hepatic stellate cells, portal fibroblasts, and mesothelial cells in normal and fibrotic livers. J Hepatol. https://doi.org/10.1016/j.jhep.2016.01.010
Lemoinne S, Cadoret A, Rautou P-E, El Mourabit H, Ratziu V, Corpechot C et al (2015) Portal myofibroblasts promote vascular remodeling underlying cirrhosis formation through the release of microparticles. Hepatology 61:1041–1055. https://doi.org/10.1002/hep.27318
Gampe K, Stefani J, Hammer K, Brendel P, Pötzsch A, Enikolopov G et al (2014) NTPDase2 and purinergic signaling control progenitor cell proliferation in neurogenic niches of the adult mouse brain. Stem Cells. https://doi.org/10.1002/stem.1846
Mitchell C, Willenbring H (2008) A reproducible and well-tolerated method for 2/3 partial hepatectomy in mice. Nat Protoc 3:1167–1170. https://doi.org/10.1038/nprot.2008.80
Popov Y, Sverdlov DY, Sharma AK, Bhaskar KR, Li S, Freitag TL et al (2011) Tissue transglutaminase does not affect fibrotic matrix stability or regression of liver fibrosis in mice. Gastroenterology 140:1642–1652. https://doi.org/10.1053/j.gastro.2011.01.040
Fickert P, Stöger U, Fuchsbichler A, Moustafa T, Marschall H-U, Weiglein AH et al (2007) A new xenobiotic-induced mouse model of sclerosing cholangitis and biliary fibrosis. Am J Pathol 171:525–536. https://doi.org/10.2353/ajpath.2007.061133
Popov Y, Patsenker E, Bauer M, Niedobitek E, Schulze-Krebs A, Schuppan D (2006) Halofuginone induces matrix metalloproteinases in rat hepatic stellate cells via activation of p38 and NFkappaB. J Biol Chem 281:15090–15098. https://doi.org/10.1074/jbc.M600030200.
Bartel DL, Sullivan SL, Lavoie EG, Sevigny J, Finger TE (2006) Nucleoside triphosphate diphosphohydrolase-2 is the ecto-ATPase of type I cells in taste buds. J Comp Neurol 497:1–12. https://doi.org/10.1002/cne.20954.
Burnstock G, Vaughn B, Robson SC (2014) Purinergic signalling in the liver in health and disease. Purinergic Signal 10:51–70. https://doi.org/10.1007/s11302-013-9398-8
Ayata CK, Ganal SC, Hockenjos B, Willim K, Vieira RP, Grimm M et al (2012) Purinergic P2Y2 receptors promote neutrophil infiltration and hepatocyte death in mice with acute liver injury. Gastroenterology 143:1620. https://doi.org/10.1053/j.gastro.2012.08.049
Grant CR, Liberal R, Holder BS, Cardone J, Ma Y, Robson SC et al (2013) Dysfunctional CD39 regulatory T cells and aberrant control of T-helper type 17 cells in autoimmune hepatitis. Hepatology. https://doi.org/10.1002/hep.26583
Schmelzle M, Duhme C, Junger W, Salhanick SD, Chen Y, Wu Y et al (2013) CD39 modulates hematopoietic stem cell recruitment and promotes liver regeneration in mice and humans after partial hepatectomy. Ann Surg 257:693–701. https://doi.org/10.1097/SLA.0b013e31826c3ec2
Fausto N, Campbell JS, Riehle KJ (2006) Liver regeneration. Hepatology 43:S45–S53. https://doi.org/10.1002/hep.20969
Dranoff JA, Ogawa M, Kruglov EA, Gaça MDA, Sévigny J, Robson SC et al (2004) Expression of P2Y nucleotide receptors and ectonucleotidases in quiescent and activated rat hepatic stellate cells. Am J Physiol Gastrointest Liver Physiol 287:24. https://doi.org/10.1152/ajpgi.00294.2003
Dranoff JA, Kruglov EA, Abreu-Lanfranco O, Nguyen T, Arora G, Aurora G et al (2007) Prevention of liver fibrosis by the purinoceptor antagonist pyridoxal-phosphate-6-azophenyl-2′,4′-disulfonate (PPADS). In Vivo 21:957–965
Chiang DJ, Roychowdhury S, Bush K, McMullen MR, Pisano S, Niese K et al (2013) Adenosine 2A receptor antagonist prevented and reversed liver fibrosis in a mouse model of ethanol-exacerbated liver fibrosis. PLoS One 8:e69114. https://doi.org/10.1371/journal.pone.0069114
Yoshida S, Ikenaga N, Liu SB, Peng Z-W, Chung J, Sverdlov DY et al (2014) Extra-hepatic PDGFB, delivered by platelets, promotes activation of hepatic stellate cells and biliary fibrosis in mice. Gastroenterology. https://doi.org/10.1053/j.gastro.2014.08.038
Flores RV, Hernández-Pérez MG, Aquino E, Garrad RC, Weisman GA, Gonzalez FA (2005) Agonist-induced phosphorylation and desensitization of the P2Y2 nucleotide receptor. Mol Cell Biochem 280:35–45. https://doi.org/10.1007/s11010-005-8050-5
Kauffenstein G, Pelletier J, Lavoie EG, Kukulski F, Martin-Satue M, Dufresne SS et al (2014) Nucleoside triphosphate diphosphohydrolase-1 ectonucleotidase is required for normal vas deferens contraction and male fertility through maintaining P2X1 receptor function. J. Biol. Chem. https://doi.org/10.1074/jbc.M114.604082
Dranoff JA, Kruglov EA, Toure J, Braun N, Zimmermann H, Jain D et al (2004) Ectonucleotidase NTPDase2 is selectively down-regulated in biliary cirrhosis. J Investig Med 52:475–482
Yu J, Lavoie EG, Sheung N, Tremblay JJ, Sévigny J, Dranoff JA (2008) IL-6 downregulates transcription of NTPDase2 via specific promoter elements. Am J Physiol Gastrointest Liver Physiol 294:56. https://doi.org/10.1152/ajpgi.00208.2007
Aoki H, Aoki M, Yang J, Katsuta E, Mukhopadhyay P, Ramanathan R et al (2016) Murine model of long-term obstructive jaundice. J Surg Res 206:118–125. https://doi.org/10.1016/j.jss.2016.07.020
Riteau N, Gasse P, Fauconnier L, Gombault A, Couegnat M, Fick L et al (2010) Extracellular ATP is a danger signal activating P2X7 receptor in lung inflammation and fibrosis. Am J Respir Crit Care Med 182:774–783. https://doi.org/10.1164/rccm.201003-0359OC
Funding
LF received a research fellowship from the Deutsche Forschungsgemeinschaft (DFG; FE1434/1-1), ZGJ was granted a clinical research award from ACG and a clinical translational research award from AASLD, JS received a “Chercheur National” scholarship award from the Fonds de recherche du Québec-Santé (FRQS). This work was also, in part, supported by the National Institutes of Health grants to SCR (NIH; P01HL107152, R21CA164970) and Canadian Institute of Health Research to JS (CIHR; MOP-102472).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
Linda Feldbrügge declares that she has no conflict of interest.
Z. Gordon Jiang declares that he has no conflict of interest.
Eva Csizmadia declares that she has no conflict of interest.
Shuji Mitsuhashi declares that he has no conflict of interest.
Stephanie Tran declares that she has no conflict of interest.
Eric U. Yee declares that he has no conflict of interest.
Sonja Rothweiler declares that she has no conflict of interest.
Kahini A. Vaid declares that she has no conflict of interest.
Jean Sévigny declares that he has no conflict of interest.
Moritz Schmelzle declares that he has no conflict of interest.
Yury V. Popov declares that he has no conflict of interest.
Simon C. Robson declares that he has no conflict of interest.
Ethical approval
The animal experiment protocol was reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Beth Israel Deaconess Medical Center.
Electronic supplementary material
ESM 1
(PDF 883 kb)
Rights and permissions
About this article
Cite this article
Feldbrügge, L., Jiang, Z.G., Csizmadia, E. et al. Distinct roles of ecto-nucleoside triphosphate diphosphohydrolase-2 (NTPDase2) in liver regeneration and fibrosis. Purinergic Signalling 14, 37–46 (2018). https://doi.org/10.1007/s11302-017-9590-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11302-017-9590-3