Abstract
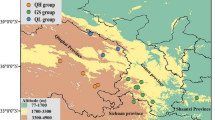
Test of the relationship of genetic and particularly epigenetic variation with geographic isolation and environment is important to reveal potential environmental drivers for selection. Rhododendron oldhamii is widespread but inhabits fragmented subtropical forest landscapes, and populations across its range may exhibit different levels of genetic and epigenetic structuring correlated to their environmental conditions. Here, we investigated the genetic and epigenetic variations and their ecological correlates in R. oldhamii. Genetic and epigenetic variations were surveyed using amplified fragment length polymorphism (AFLP) and methylation-sensitive amplification polymorphism (MSAP), respectively. Using variation partitioning by redundant analysis (RDA), we examined the pure and spatially structured environmental effects and pure spatial factors on genetic and epigenetic variations among individuals collected from 18 localities across R. oldhamii distribution range in Taiwan. We found that environments compared to geographic isolation among sites explained more genetic and epigenetic variations. Patchy distribution of the contemporary R. oldhamii populations was revealed by correlograms with patch size of approximately around 20–30 km based on the total genetic and epigenetic data. Spatial variables derived from the method of principal coordinates of neighbor matrices (PCNM), including PCNM3, PCNM5, PCNM7, and PCNM8 representing biotic processes, such as individual dispersal, were found to be important influencing potentially adaptive genetic and epigenetic variations. Annual mean temperature, annual precipitation, precipitation of the warmest quarter, aspect, slope, and soil moisture were the most important environmental variables influencing potentially adaptive genetic and epigenetic variations and could be particularly important for the evolution of local adaptation in R. oldhamii.



Similar content being viewed by others
References
Batke SP, Jocque M, Kelly DL (2014) Modelling hurricane exposure and wind speed on a mesoclimate scale: a case study from Cusuco NP, Honduras. PLoS One 9:e91306
Bartoń K (2013) MuMIn: multi-model inference. R package version 1(9):13, Available at: http://CRAN.R-project.org/package=MuMin. Accessed 29 October 2013
Beaumont MA, Balding DJ (2004) Identifying adaptive genetic divergence among populations from genome scans. Mol Ecol 13:969–980
Blanchet FG, Legendre P, Borcard D (2008) Forward selection of explanatory variables. Ecology 89:2623–2632
Borcard D, Legendre P (2002) All-scale spatial analysis of ecological data by means of principal coordinates of neighbor matrices. Ecol Model 153:51–68
Borcard D, Legendre P, Drapeau P (1992) Partialling out the spatial component of ecological variation. Ecology 73:1045–1055
Borcard D, Gillet F, Legnedre P (2011) Numerical ecology with R. Springer, New York
Bossdorf O, Richards CL, Pigliucci M (2008) Epigenetics for ecologists. Ecol Lett 11:106–115
Bothwell H, Bisbing S, Therkildsen NO, Crawford L, Alvarez N, Holderegger R, Manel S (2013) Identifying genetic signatures of selection in a non-model species, alpine gentian (Gentiana nivalis L.), using a landscape genetic approach. Conserv Genet 14:467–481
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Caccamo G, Chisholm LA, Bradstock RA, Puotinen ML (2011) Assessing the sensitivity of MODIS to monitor drought in high biomass ecosystems. Rem Sens Environ 115:2626–2639
Carrera-Hernández JJ, Gaskin SJ (2007) Spatio temporal analysis of daily precipitation and temperature in the basin of Mexico. J Hydrol 336:231–249
Chang L-W, Zelený D, Li C-F, Chiu S-T, Hsieh C-F (2013) Better environmental data may reverse conclusions about niche- and dispersal-based processes in community assembly. Ecology 94:2145–2151
Chen C-Y, Liang B-K, Chung J-D, Chang C-T, Hsieh Y-C, Lin T-C, Hwang S-Y (2014) Demography of the upward-shifting temperate woody species of the Rhododendron pseudochrysanthum complex and ecologically relevant adaptive divergence in its trailing edge populations. Tree Genet Gen 10:111–126
Chiou C-R, Song G-Z M, Chien J-H, Hsieh C-F, Wang J-C, Chen M-Y, Liu H-Y, Yeh C-L, Hsia Y-J, Chen T-Y (2010) Altitudinal distribution patterns of plant species in Taiwan are mainly determined by the northeast monsoon rather than the heat retention mechanism of Massenerhebung. Bot Stud 51:89–97
Cottenie K (2005) Integrating environmental and spatial processes in ecological community dynamics. Ecol Lett 8:1175–1182
Dray S (2013) Packfor: forward selection with permutation (Canoco p.46), R package version 0.0-8. Available at: http://r-forge.r-project.org/R/?group_id=195. Accessed 25 October 2013
Dray S, Legendre P, Peres-Neto PR (2006) Spatial-modelling: a comprehensive framework for principal coordinate analysis of neighbor matrices (PCNM). Ecol Model 196:483–493
Dray S, Pélissier R, Couteron P, Fortin M-J, Legendre P, Peres-Neto PR, Bellier E, Bivand R, Blanchet FG, De Cáceres M, Dufour A-B, Heegaard E, Jombart T, Nunoz F, Oksanen J, Thioulouse J, Wagner HH (2012) Community ecology in the age of multivariate multiscale spatial analysis. Ecol Monogr 82:257–275
Eckert CG, Samis KE, Lougheed SC (2008) Genetic variation across species’ geographical ranges: the central-marginal hypothesis and beyond. Mol Ecol 17:1170–1188
Epperson BK (1990) Spatial autocorrelation of genotypes under directional selection. Genetics 124:757–771
Epperson BK (1993) Spatial and space-time correlations in systems of subpopulations with genetic drift and migration. Genetics 133:711–727
Escaravage N, Wagner J (2004) Pollination effectiveness and pollen dispersal in a Rhododendron ferrugineum (Ericaceae) population. Pl Biol 6:606–615
Escudero A, Iriondo JM, Torres ME (2003) Spatial analysis of genetic diversity as a tool for plant conservation. Biol Conserv 113:351–365
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620
Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Res 10:564–567
Falush D, Stephens M, Pritchard JK (2007) Inference of population structure using multilocus genotype data: dominant markers and null alleles. Mol Ecol Notes 7:574–578
Fang J-Y, Chung J-D, Chiang Y-C, Chang C-T, Chen C-Y, Hwang S-Y (2013) Divergent selection and local adaptation in disjunct populations of an endangered conifer, Keteleeria davidiana var. formosana (Pinaceae). PLoS One 8:e70162
Foll M, Gaggiotti O (2008) A genome scan method to identify selected loci appropriate for both dominant and codominant markers: a Bayesain perspective. Genetics 180:977–993
Herrera CM, Bazaga P (2010) Epigenetic differentiation and relationship to adaptive genetic divergence in discrete populations of the violet Viola cazorlensis. New Phytol 187:867–876
Herrera CM, Bazaga P (2011) Untangling individual variation in natural populations: ecological, genetic and epigenetic correlates of long-term inequality in herbivory. Mol Ecol 20:1675–1688
Herrera CM, Medrano M, Bazaga P (2014) Variation in DNA methylation transmissibility, genetic heterogeneity and fecundity-related traits in natural populations of the perennial herb Helleborus foetidus. Mol Ecol 23:10851095
Heywood JS (1991) Spatial analysis of genetic variation in plant populations. Ann Rev Ecol System 22:335–355
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Internal J Climat 25:1965–1978
Hirao AS (2010) Kinship between parents reduces offspring fitness in a natural population of Rhododendron brachycarpum. Ann Bot 105:637–646
Hsieh Y-C, Chung J-D, Wang C-N, Chang C-T, Chen C-Y, Hwang S-Y (2013) Historical connectivity, contemporary isolation and local adaptation in a widespread but discontinuously distributed species endemic to Taiwan, Rhododendron oldhamii (Ericaceae). Heredity 111:147–156
Huete AR, Didan K, Miura T, Rodriguez EP, Gao X, Ferreira LG (2002) Overview of the radiometric and biophysical performance of the MODIS vegetation indices. Rem Sens Environ 83:195–213
Hufford KM, Mazer SJ (2003) Plant ecotypes: genetic differentiation in the age of ecological restoration. Trends Ecol Evol 18:147–155
Jeffreys H (1961) Theory of probability, 3rd edn. Oxford University Press, Oxford
Jombart T (2008) adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24:1403–1405
Jombart T, Devillard S, Balloux F (2010) Discriminant analysis of principal components: a new method for the analysis of genetically structured populations. BMC Genet 11:94
Johnson LJ, Tricker PJ (2010) Epigenomic plasticity within populations: its evolutionary significance and potential. Heredity 105:113–121
Kameyama Y, Isagi Y, Nakagoshi N (2000) Microsatellite analysis of pollen flow in Rhododendron metternichii var. hondoense. Ecol Res 15:263–269
Kozak KH, Wiens JJ (2006) Does niche conservatism promote speciation: a case study in North American salamanders. Evolution 60:2604–2621
Latzel V, Allan E, Silveira AB, Colot V, Fischer M, Bossdorf O (2013) Epigenetic diversity increases the productivity and stability of plant populations. Nat Commu 4:2875
Legendre P, Fortin M-J (1989) Spatial pattern and ecological analysis. Vegetatio 80:107–138
Legendre P, Mi XC, Ren HB, Ma KP, Yu MJ, Sun I-F, He FL (2009) Partitioning beta diversity in a subtropical broad-leaved forest of China. Ecology 90:663–674
Liew P-M, Chung N-J (2001) Vertical migration of forests during the last glacial period in subtropical Taiwan. West Pac Earth Sci 1:405–414
Lira-Medeiros CF, Parisod C, Fernandes RA, Mata CS, Cardoso MA, Ferreira PCG (2010) Epigenetic variation in mangrove plants occurring in contrasting natural environments. PLoS One 5:e10326
Manel S, Schwartz MK, Luikart G, Taberlet P (2003) Landscape genetics: combining landscape ecology and population genetics. Trends Ecol Evol 18:189–197
Manel S, Poncet BN, Legendre P, Gugerli F, Holderegger R (2010) Common factors drive adaptive genetic variation at different spatial scales in Arabis alpina. Mol Ecol 19:3824–3835
Manel S, Gugerli F, Thuiller W, Alvarez N, Legendre P, Holderegger R, Gielly L, Taberlet P, IntraBioDiv Consortium (2012) Broad-scale adaptive genetic variation in alpine plants is driven by temperature and precipitation. Mol Ecol 21:3729–2738
Mikita T, Klimánek M (2010) Topographic exposure and its practical applications. J Landscape Ecol 3:42–51
Nei M (1973) Analysis of gene diversity in subdivided populations. Proc Natl Acad Sci U S A 70:3321–3323
Nei M, Li W-H (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci U S A 76:5269–5273
Nybom H (2004) Comparison of different nuclear DNA markers for estimating intraspecific genetic diversity in plants. Mol Ecol 13:1143–1155
Oden N, Sokal RR (1986) Directional autocorrelation: an extension of spatial correlograms to two dimensions. Syst Zool 35:608–617
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens HH, Wagner H (2011) vegan: community ecology package. R package version 2.0-1. Available at: http://CRAN.R-project.org/package=vegan. Accessed 31 December 2013.
Peakall R, Smouse PE (2012) GenAlEx 6.5: genetic analysis in excel. Population genetic software for teaching and research—an update. Bioinformatics 28:2537–2539
Poncet BN, Herrmann D, Gugerli F, Taberlet P, Holderegger R, Gielly L, Rious D, Thuiller W, Aubert S, Manel S (2010) Tracking genes of ecological relevance using a genome scan in two independent regional population samples of Arabis alpina. Mol Ecol 19:2896–2907
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
R Development Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, http://www.R-project.org/
Radeloff VC, Miller TF, He HS, Mladenoff DJ (2000) Periodicity in spatial data and geostatistical models: autocorrelation between patches. Ecography 23:81–92
Rapp RA, Wendel JF (2005) Epigenetics and plant evolution. New Phytol 168:81–91
Richards EJ (2006) Inherited epigenetic variation—revisiting soft inheritance. Nat Rev Genet 7:395–401
Richards EJ (2011) Natural epigenetic variation in plant species: a view from the field. Curr Opin Pl Biol 14:204–209
Sokal RR, Oden NL (1978) Spatial autocorrelation in biology. 2. Some biological implications and four applications of evolutionary and ecological interest. Biol J Linn Soc 10:229–249
Sokal RR, Jacquez GM, Wooten MC (1989) Spatial autocorrelation analysis of migration and selection. Genetics 121:845–855
Storfer A, Murphy MA, Evans JS, Goldberg CS, Robinson S, Spear SF, Dezzani R, Delmelle E, Vierling L, Waits LP (2007) Putting the “landscape” in landscape genetics. Heredity 98:128–142
Su H-J (1985) Studies on the climate and vegetation types of the natural forests in Taiwan (III): a scheme of geographic regions. Quar J Chin For 18:33–44
Taiwan Forestry Bureau (1995) The third survey of forest resources and land use in Taiwan. Taiwan Forestry Bureau, Department of Agriculture, Taipei, Taiwan.
Vekemans X, Beauwens T, Lemaire M, Roldan-Ruiz I (2002) Data from amplified fragment length polymorphism (AFLP) markers show indication of size homoplasy and of a relationship between degree of homoplasy and fragment size. Mol Ecol 11:139–151
Vos P, Hogers R, Bleeker M, Reijans M, van der Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M, Zabeau M (1995) AFLP: a new technique for DNA fingerprinting. Nucl Acid Res 23:4407–4414
Wagner HH, Fortin M-J (2005) Spatial analysis of landscapes: concepts and statistics. Ecology 86:1975–1987
Wang IJ, Glor RE, Losos JB (2013) Quantifying the roles of ecology and geography in spatial genetic divergence. Ecol Lett 16:175–182
Xiong LZ, Xu CG, Maroof MAS, Zhang Q (1999) Patterns of cytosine methylation in an elite rice hybrid and its parental lines, detected by a methylation-sensitive amplification polymorphism technique. Mol Gen Genet 261:439–446
Yilmaz MT, Hunt ER, Jackson TJ (2008) Remote sensing of vegetation water content from equivalent water thickness using satellite imagery. Rem Sens Environ 112:2514–2522
Zhivotovsky LA (1999) Estimating population structure in diploids with multilocus dominant DNA markers. Mol Ecol 8:907–913
Acknowledgments
We thank Ji-Sheng Wu for the assistance with field collections. This research was supported by the National Science Council of Taiwan (grant numbers NSC100-2621-B-003-001 and NSC101-2313-B-003-001-MY3 to SYH). We thank Yen-Heng Lin for his laboratory work. Postdoctoral fellowships awarded to JHC and to CTC from the National Science Council of Taiwan are also acknowledged.
Data archiving statement
AFLP and MSAP fingerprinting datasets used in this study deposited at Dryad: http://dx.doi.org/10.5061/dryad.fm278
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Y. Tsumura
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 3195 kb)
Rights and permissions
About this article
Cite this article
Huang, CL., Chen, JH., Tsang, MH. et al. Influences of environmental and spatial factors on genetic and epigenetic variations in Rhododendron oldhamii (Ericaceae). Tree Genetics & Genomes 11, 823 (2015). https://doi.org/10.1007/s11295-014-0823-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11295-014-0823-0