Abstract
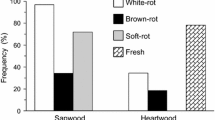
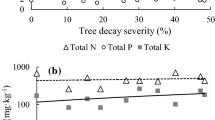
Fungal decomposition of coarse woody debris has an essential role in the biodiversity and carbon and nutrient dynamics in forest ecosystems. However, our knowledge of the effects of fungal species and within-stem diversity on wood decay is limited. In this paper, I described the process of wood decay and fungal succession of pine (Pinus densiflora) snags using chronosequence method. The results showed that the decay process differed between sapwood and heartwood. Sapwood decayed faster than heartwood, despite their initial densities being the same. Sapwood decay occurred in two phases. The first phase involved a typical white-rot process wherein acid-unhydrolysable residue (lignin) and holocellulose decayed simultaneously. White-rot species, such as Trichaptum abietinum and Phanerochaete sordida, were the dominant fungi and were likely the functional decomposers in this phase. The second phase involved selective decomposition of holocellulose. The dominant fungi in this phase included soft-rot species, such as Trichoderma spp., that can decompose holocellulose in wood that was previously delignified by white-rot species. In contrast to sapwood, heartwood experienced less loss of wood density and no clear change in lignocellulose composition, even in later stages of decay. Dominant fungi in heartwood included the latent inhabitants of living pine trees, such as Ascocoryne cylichnium, which is known to reduce colonization and wood decay by strong decomposers. These results suggest that, in addition to decay-resistant chemicals present in heartwood, the differences in the fungal communities present in heartwood and sapwood were responsible for the differences in their decay rates.





Similar content being viewed by others
References
Basham JT (1966) Heart rot of jack pine in Ontario. II. Laboratory studies on the pathogenicity and interrelationships of the principal heartwood-inhabiting fungi. Can J Bot 44:849–860
Basham JT (1973) Heart rot of black spruce in Ontario. II. The mycoflora in defective and normal wood of living trees. Can J Bot 51:1379–1392
Basham JT (1975) Heart rot of jack pine in Ontario. IV. Heartwood-inhabiting fungi, their entry and interactions within living trees. Can J For Res 5:706–721
Boddy L, Gibbon OM, Grundy MA (1985) Ecology of Daldinia concentrica: effects of abiotic variations on mycelial extension and interspecific interactions. Trans Br Mycol Soc 85:201–211
Bourchier RJ (1961) Laboratory studies on microfungi isolated from the stems of living lodgepole pine, Pinus contorta Dougl. Can J Bot 39:1373–1385
Bunnell FL, Houde I (2010) Down wood and biodiversity—implications to forest practices. Environ Rev 18:397–421
Christensen O (1984) The states of decay of woody litter determined by relative density. Oikos 42:211–219
Coates D, Rayner ADM (1985) Fungal population and community development in cut beech logs III. Spatial dynamics and strategies. New Phytol 101:183–198
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Eaton RA, Hale MDC (1993) Wood. Decay, pests and protection. Chapman & Hall, Tokyo
Eriksson K-EL, Blanchette RA, Ander P (1990) Microbial and enzymatic degradation of wood and wood components. Springer, Berlin
Fukasawa Y (2012) Effects of wood decomposer fungi on tree seedling establishment on coarse woody debris. For Ecol Manag 266:232–238
Fukasawa Y (2016) Seedling regeneration on decayed pine logs after the deforestation events caused by pine wilt disease. Annal For Res 59:191–198
Fukasawa Y, Osono T, Takeda H (2009) Dynamics of physicochemical properties and occurrence of fungal fruit bodies during decomposition of coarse woody debris of Fagus crenata. J For Res 14:20–29
Fukasawa Y, Osono T, Takeda H (2010) Beech log decomposition by wood-inhabiting fungi in a cool temperate forest floor: a quantitative analysis focused on the decay activity of a dominant basidiomycete Omphalotus guepiniformis. Ecol Res 25:959–966
Fukasawa Y, Osono T, Takeda H (2011) Wood decomposing abilities of diverse lignicolous fungi on nondecayed and decayed beech wood. Mycologia 103:474–482
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes a application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Griffin MA, Spakowicz DJ, Gianoulis TA, Strobel SA (2010) Volatile organic compound produced by organisms in the genus Ascocoryne and a re-evaluation of myco-diesel production by NRRL 50072. Microbiol 156:3814–3829
Harju AM, Venäläinen M, Anttonen S, Viitanen H, Kainulainen P, Saranpää P, Vapaavuori E (2003) Chemical factors affecting the brown-rot decay resistance of Scots pine heartwood. Trees 17:263–268
Harmon ME, Franklin JF, Swanson FJ, Sollins P, Gregory SV, Lattin JD, Anderson NH, Cline SP, Aumen NG, Sedell JR, Lienkaemper GW, Cromack K Jr, Cummins KW (1986) Ecology of coarse woody debris. Adv Ecol Res 15:133–302
Hillis WE (1987) Heartwood and tree exudates. Springer, Tokyo
Hintikka V, Korhonen K (1970) Effects of carbon dioxide on the growth of lignicolous and soil-inhabiting hymenomycetes. Commun Inst For Fenniae 62:1–22
Huse KJ (1981) The distribution of fungi in sound-looking stems of Picea abies in Norway. For Pathol 11:1–6
Jankowiak R (2005) Fungi associated with Ips typographus on Picea abies in southern Poland and their succession into the phloem and sapwood of beetle-infested trees and logs. For Pathol 35:37–55
Jankowiak R, Kurek M (2006) The early stages of fungal succession in Pinus sylvestris phloem and sapwood infested by Tomicus piniperda. Dendrobiol 56:27–36
Jankowiak R, Rossa R, Mista K (2007) Survey of fungal species vectored by Ips cembrae to European larch trees in Raciborskie forests (Poland). Czech Mycol 59:227–239
Japan Meteorological Agency (2014) Meteorological data. http://www.data.jma.gop/obd/stats/etrn/index.php. Accessed 3 Oct 2017
Jongman RHG, ter Braak CJF, van Tongeren OFR (1995) Data analysis in community and landscape ecology. Cambridge University Press, Cambridge
Käärik A (1967) Colonization of pine and spruce poles by soil fungi after six months. Mater Organ 2:97–108
Kebli H, Kernaghan G, Drouin P, Brais S (2014) Development and activity of early saproxylic fungal communities in harvested and unmanaged boreal mixedwood stands. Eur J For Res 133:905–918
King HGC, Heath GW (1967) The chemical analysis of small samples of leaf material and the relationship between the disappearance and composition of leaves. Pedobiologia 7:192–197
Koide K, Osono T, Takeda H (2005) Fungal succession and decomposition of Camellia japonica leaf litter. Ecol Res 20:599–609
Koljalg U, Nilsson RH, Abarenkov K, Tedersoo L, Taylor AFS, Bahram M et al (2013) Towards a unified paradigm for sequence-based identification of fungi. Mol Ecol 22:5271–5277
Levy JF (1982) The place of basidiomycetes in the decay of wood in contact with the ground. In: Frankland JC, Hedger JN, Swift MJ (eds) Decomposer basidiomycetes: their biology and ecology. Cambridge University Press, Cambridge, pp 161–178
Masuya H, Yamaoka Y (2012) The relationships between the beetle’s ecology and the pathogenicity of their associated fungi. J Japan For Soc 94:316–325 (in Japanese with English abstract)
Masuya H, Yamaura Y, Yamaoka Y, Kaneko S (2009) Ophiostomatoid fungi isolated from Japanese red pine and their relationships with bark beetles. Mycoscience 50:212–223
Nilsson T (1973) Studies on wood degradation and cellulolytic activity of microfungi. Studia Forestalia Suecica No. 104. Royal School of Forestry, Stockholm
Osono T (2007) Ecology of ligninolytic fungi associated with leaf litter decomposition. Ecol Res 22:955–974
Osono T, Fukasawa Y, Takeda H (2003) Roles of diverse fungi in larch needle litter decomposition. Mycologia 95:820–826
Preston CM, Trofymow JA, Sayer BG, Niu J (1997) 13CPMAS NMR investigation of the proximate analysis of fractions used to assess litter quality in decomposition studies. Can J Bot 75:1601–1613
Pyle C, Brown MM (1999) Heterogeneity of wood decay classes within hardwood logs. For Ecol Manag 114:253–259
Quack W, Scholl H, Budzikiewicz H (1980) Ascocorynin, a terphenylquinone from Ascocoryne sardoides. Phytochemistry 21:2921–2923
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rayner ADM, Boddy L (1988) Fungal decomposition of wood: its biology and ecology. Willey, Chichester
Roll-hansen F, Roll-hansen H (1979a) Ascocoryne species in living stems of Picea species: a literature review. For Pathol 9:275–280
Roll-hansen F, Roll-hansen H (1979b) Microflora of sound-looking wood in Picea abies stems. For Pathol 9:308–316
Ruttimann-Johnson C, Cullen D, Lamar RT (1994) Manganase peroxidases of the white rot fungus Phanerochaete sordida. Appl Environ Microbiol 60:599–605
Schmidt O (2006) Wood and tree fungi: biology damage, protection, and use. Springer, Tokyo
Son E, Kim JJ, Lim YW, Au-Yeung TT, Yang CYH, Breuil C (2011) Diversity and decay ability of basidiomycetes isolated from lodgepole pines killed by the mountain pine beetle. Can J Microbiol 57:33–41
Stokland J, Siitonen J, Gunnar Jonsson B (2012) Biodiversity in dead wood. Cambridge University Press, Cambridge
Strobel G, Tomsheck A, Geary B, Spakowicz D, Strobel S, Mattner S, Mann R (2010) Endophyte strain NRRL 50072 producing volatile organics is a species of Ascocoryne. Mycology 1:187–194
Svoboda M, Pouska V (2008) Structure of a Central-European mountain spruce old-growth forest with respect to historical development. For Ecol Manag 255:2177–2188
Tedersoo L, Bahram M, Polme S, Koljalg U, Yorou NS et al (2010) Global diversity and geography of soil fungi. Science 346:1256688
ter Braak CJF, Šmilauer P (2002) CANOCO reference manual and CanoDraw for windows user’s guide: software for canonical community ordination, version 4.5. Microcomputer Power, Ithaca
van der Wal A, Ottoson E, de Boer W (2015) Neglected role of fungal community composition in explaining variation in wood decay rates. Ecology 96:124–133
Wilhere GF (2003) Simulations of snag dynamics in an industrial Douglas-fir forest. For Ecol Manag 174:521–539
Yoneda T (1975) Studies on the rate of decay of wood litter on the forest floor. I. Some physical properties of decaying wood. Jpn J Ecol 25:40–46
Zabel R, Lombard FF, Wang CJK, Terracina F (1985) Fungi associated with decay in treated southern pine utility poles in the eastern United States. Wood Fiber Sci 17:75–91
Acknowledgements
I wish to thank Ayumi Matsuo and Takeshi Taniguchi for technical advises in DNA analysis. Thanks are extended to Toshihiro Yabuki for Trichoderma identification and Yosuke Degawa for Mortierella identification. I also thank Emma Gilmartin for her helpful comments on the draft manuscript. This work was supported by Japan Society for the Promotion of Science (JSPS) KAKENHI Grant number 23780156 for YF.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no conflicts of interest. All the experiments undertaken in this study comply with the current laws of Japan.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Fukasawa, Y. Fungal succession and decomposition of Pinus densiflora snags. Ecol Res 33, 435–444 (2018). https://doi.org/10.1007/s11284-017-1557-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-017-1557-x