Abstract
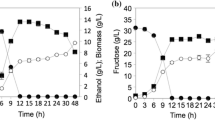
This work was mainly about the understanding of how urea and ammonium affect growth, glucose consumption and ethanol production of S. cerevisiae, in particular regarding the basic physiology of cell. The basic physiology of cell included intracellular pH, ATP, NADH and enzyme activity. Results showed that fermentation time was reduced by 19% when using urea compared with ammonium. The maximal ethanol production rate using urea was 1.14 g/L/h, increasing 30% comparing with the medium prepared with ammonium. Moreover, urea could decrease the synthesis of glycerol from glucose by 26% comparing with ammonium. The by-product of acetic acid yields decreased from 40 mmol/mol of glucose (with urea) to 24 mmol/mol of glucose (with ammonium). At the end of ethanol fermentation, cell number and pH were greater with urea than ammonium. Comparing with urea, ammonium decreased the intracellular pH by 14% (from 7.1 to 6.1). Urease converting urea into ammonia resulted in a more than 50% lower of ATP when comparing with ammonium. The values of NADH/DCW were 0.21 mg/g and 0.14 mg/g respectively with urea and ammonium, suggesting a 33% lower NADH. The enzyme activity of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was 0.0225 and 0.0275 U/mg protein respectively with urea and ammonium, which was consistent with the yields of glycerol.




Similar content being viewed by others
References
Azmat U, Gayathri C, Stanley B et al (2013) Yeast adaptation to weak acids prevents futile energy expenditure. Front Microbiol 4(142):1–10
Blomberg A, Adler L (1989) Roles of glycerol and glycerol-3-phosphate dehydrogenase (NAD+) in acquired osmotolerance of Saccharomyces cerevisiae. J Bacteriol 171(2):1087–1092
Brett CL, Donowitz M, Rao R (2006) Does the proteome encode organelle pH. FEBS Lett 580(3):717–719
Cooper TG, Sumrada R (1975) Urea transport in Saccharomyces cerevisiae. J Bacteriol 121(2):571–576
Edgley M, Brown AD (1983) Yeast water relations---physiological-changes induced by solute stress in Saccharomyces-cerevisiae and Saccharomyces-rouxii. Microbiology 129(11):3453–3463
Elberry HM, Majumdar ML, Cunningham TS, Sumrada RA, Cooper TG (1993) Regulation of the urea active transporter gene (DUR3) in Saccharomyces cerevisiae. J Bacteriol 175(15):4688–4698
Eva Albers CL, Gunnar Lide N, Niklasson C, Gustafsson L (1996) Influence of the nitrogen source on Saccharomyces cerevisiae. Appl Environ Microbiol 62:3187–3195
Godard P, Urrestarazu A, Vissers S, Kontos K, Bontempi G, Helden JV, André B (2007) Effect of 21 different nitrogen sources on global gene expression in the yeast Saccharomyces cerevisiae. Mol Cell Biol 27(8):3065–3086
Hensing MCM, Bangma KA, Raamsdonk LM, Hulster ED, Dijken JPV, Pronk JT (1995) Effects of cultivation conditions on the production of heterologous α-galactosidase by Kluyveromyces lactis. Appl Microbiol Biotechnol 43(1):58–64
Hou J, Lages NF, Oldiges M, Vemuri GN (2009) Metabolic impact of redox cofactor perturbations in Saccharomyces cerevisiae. Metab Eng 11(4–5):253–261
Kemsawasd V, Viana T, Ardö Y, Arneborg N (2015) Influence of nitrogen sources on growth and fermentation performance of different wine yeast species during alcoholic fermentation. Appl Microbiol Biotechnol 99(23):10191–10207
Kok SD, Kozak BU, Pronk JT, Maris AJAV (2012) Energy coupling in Saccharomyces cerevisiae : selected opportunities for metabolic engineering. FEMS Yeast Res 12(4):387–397
Milne N, Luttik MA, Cueto Rojas HF, Wahl A, van Maris AJ, Pronk JT, Daran JM (2015) Functional expression of a heterologous nickel-dependent, ATP-independent urease in Saccharomyces cerevisiae. Metab Eng 30:130–140
Mobley HL, Island MD, Hausinger RP (1995) Molecular biology of microbial ureases. Microbiol Rev 59(3):451–480
Orij R, Postmus J, Ter Beek A, Brul S, Smits GJ (2009) In vivo measurement of cytosolic and mitochondrial pH using a pH-sensitive GFP derivative in Saccharomyces cerevisiae reveals a relation between intracellular pH and growth. Microbiology 155(1):268–278
Schure EGT, Riel NAWV, Verrips CT (2000) The role of ammonia metabolism in nitrogen catabolite repression in Saccharomyces cerevisiae. FEMS Microbiol Rev 24(1):67–83
Sumrada R, Gorski M, Cooper T (1976) Urea transport-defective strains of Saccharomyces cerevisiae. J Bacteriol 125(3):1048–1056
Sunya S, Gorret N, Delvigne F (2012) Real-time monitoring of metabolic shift and transcriptional induction of yciG::luxCDABE E. coli reporter strain to a glucose pulse of different concentrations. J Biotechnol 157(3):379–390
Wang K, Mao Z, Zhang C, Zhang J, Zhang H, Tang L (2012) Influence of nitrogen sources on ethanol fermentation in an integrated ethanol-methane fermentation system. Bioresour Technol 120:206–211
Wong KH, Hynes MJ, Davis MA (2008) Recent advances in nitrogen regulation: a comparison between Saccharomyces cerevisiae and Filamentous fungi. Eukaryot Cell 7(6):917–925
Wu X, Bean WD, Wilson SR (2006) Ethanol production from pearl millet by using saccharomyces. Cereal Chem 83:127–131
Zeng X, Chen XS, Ren XD, Liu QR, Wang L, Sun QX, Tang L, Mao ZG (2014) Insights into the role of glucose and glycerol as a mixed carbon source in the improvement of epsilon-poly-L-lysine productivity. Appl Biochem Biotechnol 173(8):2211–2224
Zhang A, Chen X (2008) Improve ethanol yield through minimizing glycerol yield in ethanol fermentation of Saccharomyces cerevisiae. Chin J Chem Eng 16(4):620–625
Zhao X, Zou H, Chen J, Du G, Zhou J (2016) The modification of Gat1p in nitrogen catabolite repression to enhance non-preferred nitrogen utilization in Saccharomyces cerevisiae. Sci Rep 6:21603
Zhao X, Zou H, Fu J, Chen J, Zhou J, Du G (2013) Nitrogen regulation involved in the accumulation of urea in Saccharomyces cerevisiae. Yeast 30(11):437–447
Zhu Y, Yang ST (2004) Effect of pH on metabolic pathway shift in fermentation of xylose by Clostridium tyrobutyricum. J Biotechnol 110(2):143-157
Acknowledgements
This study was financially supported by the following projects: (1) Shandong postdoctoral innovation project. (2) Doctor’s fund of University of Jinan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflicts of interest to this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, X., Yang, Y., Huang, J. et al. Comparisons of urea or ammonium on growth and fermentative metabolism of Saccharomyces cerevisiae in ethanol fermentation. World J Microbiol Biotechnol 37, 98 (2021). https://doi.org/10.1007/s11274-021-03056-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-021-03056-9