Abstract
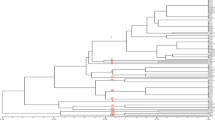
Alternaria leaf spot caused by Alternaria carthami is one of the most devastating diseases of safflower. Diversity among 95 isolates of A. carthami was determined using virulence assays, enzyme assays, dominant (ISSR) and co-dominant (SSR) markers. Collections and isolations were made from three major safflower producing states of India. The virulence assays categorised the population into four groups based on level of virulence. Estimation of activities of cell wall degrading enzymes (CWDE) yielded concurrent results to virulence assays with maximum CWDE activities in most virulent group. Eighteen ISSR primers were used and 23 polymorphic microsatellite markers were developed to assess the genetic diversity and determine the population structure of A. carthami. Analysis of ISSR profiles revealed high genetic diversity (Nei’s Genetic diversity index; h = 0.36). Microsatellite markers produced a total of 56 alleles with an average of 2.43 alleles per microsatellite marker and Nei’s genetic diversity index as h = 0.43. Unweighted Neighbor-joining and population structure analysis using both the marker systems differently arranged the isolates into three clusters. Distance analysis of the marker profiles provided no evidence for geographical clustering of isolates, indicating that isolates are randomly spread across India, signifying high potential of the fungus to adapt to diverse regions. Microsatellite markers clustered the isolates in consonance to the virulence groups in the dendrogram. This implies that the fungus has a high potential to adapt to resistant cultivars or fungicides. The information can aid in the breeding and deployment of A. carthami resistant varieties, and in early blight disease management in all safflower growing regions of the world.





Similar content being viewed by others
References
Alves JL, Saraiva RM, Mizubuti ESG, Carneiro SMTPG, Borsato LC, Woudenberg JHC, Lourenço V Jr (2016) First report of Alternaria carthami causing leaf spots on Carthamus tinctorius in Brazil. New Dis Rep 33:3–4
Anand T, Bhaskaran R, Karthikeyan TG, Rajesh M, Senthilraja G (2008) Production of cell wall degrading enzymes and toxins by Colletotrichum capsici and Alternaria alternata causing fruit rot of chillies. J Plant Prot Res 48:437–451
Bagherabadi S, Zafari D, Soleimani MJ (2015) Genetic diversity of Alternaria alternata isolates causing potato brown leaf spot, using ISSR markers in Iran. J Plant Pathol Microbiol 6:1–6
Balaban MO, Arreola AG, Marshall M, Peplow A, Wei CI, Cornell J (1991) Inactivation of pectinesterase in orange juice by supercritical carbon dioxide. J Food Sci 56:743–746
Benali S, Mohamed B, Eddine HJ, Neema C (2011) Advances of molecular markers application in plant pathology research. Eur J Sci Res 50:110–123
Bergman JW, Carlson G, Kushnak G, Riveland NR, Stallknecht G, Welty LE, Wichman D (1989) Registration of ‘Girard’ safflower. Crop Sci 29:828–829
Capote N, Pastrana AM, Aguado A, Sanchez-Torres P (2012) Molecular tools for detection of plant pathogenic fungi and fungicide resistance. In: Cumagun CJ (ed) Plant pathology. InTech, Rijeka, pp. 151–202
Chowdhury S (1944) An Alternaria disease of Safflower. J Ind Bot Soc 23:59–65
Dambal GI, Patil RS (2017) Screening of safflower germplasm against Alternaria leaf spot. Plant Arch 1:454–456
Dang HX, Pryor B, Peever T, Lawrence CB (2015) The Alternaria genomes database: a comprehensive resource for a fungal genus comprised of saprophytes, plant pathogens, and allergenic species. BMC Genom 16:239–247
Dela Torre Onofre MH, Garza Lopez JG (1989) Safflower production in north east Mexico. In: Ranga Rao V, Ramachandran M (eds) Proceedings of 2nd international Safflower conference, Hyderabad, India
Denison DA, Koehn RD (1977) Assay of cellulases. Mycologia LXIX:592
Deokar CD, Raghuwanshi KS (2002) Morphological variation of Alternaria carthami isolates on different growth media. Sesame Safflower Newsl 17:115–116
Deokar CD, Veer DM, Patil RC, Rao VR (1991) Survey of safflower diseases in Maharashtra state. Sesame Safflower Newsl 6:79–80
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Esfahani MN (2018) Analysis of virulence and genetic variability of Alternaria alternata associated with leaf spot disease in potato plants in Iran. Acta Mycol 53:1105–1114
Gannibal PB, Orina AS, Mironenko NV, Levitin MM (2014) Differentiation of the closely related species, Alternaria solani and A. tomatophila, by molecular and morphological features and aggressiveness. Eur J Plant Pathol 139:609–623
Gherbawy YA (2005) Genetic variation among isolates of Alternaria spp. from select Egyptian crops. Arch Phytopathol Plant Protect 38:77–89
Gilbert J, Knights SE, Potter TD (2008) International safflower production-an overview. In: Proceedings of 7th international Safflower conference, Wagga Wagga, Australia
Grunwald NJ, Goodwin SB, Milgroom MG, Fry WE (2003) Analysis of genotypic diversity data for populations of microorganisms. Phytopathology 93:738–746
Harrigan EKS (1987) Safflower registration of cv. Sironaria. Sesame Safflower Newsl 3:47–49
Indi DV, Lukade GM, Patil PS, Shambharkar DA (1988) Estimation of yield losses due to Alternaria leaf spot in safflower (c.o. Alternaria carthami Chowdhury) under dry land conditions. Pesticides 22:41–43
Irwin JAG (1976) Alternaria carthami, a seed-borne pathogen of safflower. Aust J Exp Agric 16:921–925
Jackson KJ, Irwin JAG, Berthelsen JE (1982) Alternaria carthami, a seedborne disease of safflower. Aust J Exp Agric Anim Husb 22:221–225
Kreis RA, Dillard HR, Smart CD (2016) Population diversity and sensitivity to Azoxystrobin of Alternaria brassicicola in New York State. Plant Dis 100:2422–2426
Kumar V, Haldar S, Pandey K, Singh R, Singh A, Singh P (2008) Cultural, morphological, pathogenic and molecular variability amongst tomato isolates of Alternaria solani in India. World J Microbiol Biotechnol 24:1003–1009
Lagercrantz U, Ellegren H, Andersson L (1993) The abundance of various polymorphic microsatellite motifs differs between plants and vertebrates. Nucleic Acids Res 21:1111–1115
Lalaoui F, Halama P, Dumortier V, Paul B (2000) Cell wall-degrading enzymes produced in vitro by isolates of Phaeosphaeria nodorum differing in aggressiveness. Plant Pathol 49:727–733
Lourenço JV, Rodrigues TT, Campos A, Bragança CA, Scheuermann KK, Reis A, Brommonschenkel SH, Maffia LA, Mizubuti ES (2011) Genetic structure of the population of Alternaria solani in Brazil. J Phytopathol 159:233–240
Madhu MR, Basavarajappa MP, Anand ID, Lokapur C (2017) Morpho-physiological variability in Alternaria carthami Chowdhury causing safflower leaf spot. Int J Curr Microbiol Appl Sci 6:3244–3250
Meng JW, Zhu W, He MH, Wu EJ, Yang LN, Shang LP, Zhan J (2015) High genotype diversity and lack of isolation by distance in the Alternaria solani populations from C hina. Plant Pathol 64:434–441
Mercado Vergnes D, Renard ME, Duveiller E, Maraite H (2006) Identification of Alternaria spp. on wheat by pathogenicity assays and sequencing. Plant Pathol 55:485–493
Milgroom MG, Fry WE (1997) Contributions of population genetics to plant disease epidemiology and management. Adv Bot Res 24:1–30
Ministry of Agriculture and Farmers’ Welfare, India. http://agriculture.gov.in
Morris PF, Connolly MS, St Clair DA (2000) Genetic diversity of Alternaria alternata isolated from tomato in California assessed using RAPDs. Mycol Res 104:286–292
Mundel HH (2008) Major achievements in safflower breeding and future challenges. In Proceedings of 7th international Safflower conference, Wagga Wagga, Australia
Mündel HH, Huang HC (2003) Control of major diseases of safflower by breeding for resistance and using cultural practices. In: Huang H-C, Acharya SA (eds) Advances in plant disease management, Research Signpost, Trivandrum, pp. 293–310
Mundt CC, Nieva LP, Vera Cruz CM (2002) Variation for aggressiveness within and between lineages of Xanthomonas oryzae pv. oryzae. Plant Pathol 51:163–168
Nei M (1973) Analysis of gene diversity in subdivided populations. Proc Natl Acad Sci USA 70:3321–3323
Ng WL, Tan SG (2015) Inter-simple sequence repeat (ISSR) markers: are we doing it right. ASM Sci J 9:30–39
Oeser B, Heidrich PM, Müller U, Tudzynski P, Tenberge KB (2002) Polygalacturonase is a pathogenicity factor in the Claviceps purpurea/rye interaction. Fungal Genet Biol 36:176–186
Open Government Data (OGD) Platform, India. National Informatics Centre (NIC), Ministry of Electronics and Information Technology, Government of India. https://data.gov.in/
Pariaud B, Ravigné V, Halkett F, Goyeau H, Carlier J, Lannou C (2009) Aggressiveness and its role in the adaptation of plant pathogens. Plant Pathol 58:409–424
Peever TL, Zeigler RS, Dorrance AE, Correa-Victoria F, Martin SS (2000) Pathogen population genetics and breeding for disease resistance. Online feature article for APSNet. American Phytopathological Society, St. Paul
Peever TL, Ibanez A, Akimitsu K, Timmer LW (2002) Worldwide phylogeography of the citrus brown spot pathogen, Alternaria alternata. Phytopathology 92:794–802
Perrier X, Jacquemoud-Collet JP (2006) DARwin software. http://darwin.cirad.fr/Darwin
Petrie GA (1974) Fungi associated with seeds of rape, turnip rape, flax and safflower in western Canada, 1968-73. Canad Plant Dis Survey 54:155–165
Petrunak DM, Christ BJ (1992) Isozyme variability in Alternaria solani and A. alternata. Phytopathology 82:1343–1343
Prasad MSL, Sujatha M, Chander Rao S (2009) Analysis of cultural and genetic diversity in Alternaria helianthi and determination of pathogenic variability using wild Helianthus species. J Phytopathol 157:609–617
Prasad MSL, Sujatha K, Naresh N (2012) Variation in morphological and cultural characters of isolates of Alternaria carthami. J Oilseeds Res 29:362–363
Pryor BM, Michailides TJ (2002) Morphological, pathogenic, and molecular characterization of Alternaria isolates associated with Alternaria late blight of pistachio. Phytopathology 92:406–416
Rampersad SN, Perez-Brito D, Torres-Calzada C, Tapia-Tussell R, Carrington CVF (2013) Genetic structure of Colletotrichum gloeosporioides sensu lato isolates infecting papaya inferred by multilocus ISSR markers. Phytopathology 103:182–189
Relekar NN, Khalikar PV, Nikam PS (2010) Survey and surveillance of Alternaria blight of safflower caused by Alternaria carthami in Marathwada region. J Plant Dis Sci 61:195–197
Rogers PM (2007) Diversity and biology among isolates of Alternaria dauci collected from commercial carrot fields. Ph.D. University of Wisconsin-Madison, Madison
Rotem J (1994) The genus Alternaria; biology, epidemiology and pathogenicity. APS Press, St. Paul
Sharma NR, Sasankan A, Singh A, Soni G (2012) Production of polygalacturonase and pectin methyl esterase from agrowaste by using various isolates of Aspergillus niger. Insight Microbiol 1:1–7
Somogyi M (1952) Determination of reducing sugars by Nelson-Somogyi method. J Biol Chem 200:245–245
Tautz D, Renz M (1984) Simple sequence repeats are ubiquitous repetitive components of eukaryotic genomes. Nucleic Acids Res 12:4127–4138
ten Have A, Tenberge KB, Benen JA, Tudzynski P, Visser J, van Kan JA (2002) The contribution of cell wall degrading enzymes to pathogenesis of fungal plant pathogens. In: Agricultural applications. Springer, Berlin Heidelberg, pp 341–358
van der Waals JE, Korsten L, Slippers B (2004) Genetic diversity among Alternaria solani isolates from potatoes in South Africa. Plant Dis 88:959–964
Wagh SS, Suryawanshi AP, Chavan PG (2017) Symptomatology and pathogenic variability of Alternaria carthami isolates from Maharashtra state infecting safflower crop. Agric Update 12:1983–1989
Wanjiru WM, Zhensheng K, Buchenauer H (2002) Importance of cell wall degrading enzymes produced by Fusarium graminearum during infection of wheat heads. Euro J Plant Pathol 108:803–810
Weber B, Halterman DA (2012) Analysis of genetic and pathogenic variation of Alternaria solani from a potato production region. Eur J Plant Pathol 134:847–858
Weir TL, Huff DR, Christ BJ, Romaine CP (1998) RAPD-PCR analysis of genetic variation among isolates of Alternaria solani and Alternaria alternata from potato and tomato. Mycologia 90:813–821
Weiss EA (1971) Castor, sesame and safflower. Barnes and Noble, New York
Woudenberg JH, Truter M, Groenewald JZ, Crous PW (2014) Large-spored Alternaria pathogens in section Porri disentangled. Stud Mycol 79:1–47
Yeh FC, Yang RC, Boyle TBJ, Ye ZH, Mao JX (1997) POPGENE, the user-friendly shareware for population genetic analysis. Molecular Biology and Biotechnology Centre, University of Alberta, Canada (program available from http://www.ualberta.ca/~fyeh/)
You FM, Huo N, Gu YQ, Luo MC, Ma Y, Hane D, Anderson OD (2008) BatchPrimer3: a high throughput web application for PCR and sequencing primer design. BMC Bioinf 9:253
Acknowledgements
The study was financially supported by Department of Science and Technology (DST-Purse II), Government of India. The authors gratefully acknowledge Dr. Charanjit Kaur, Department of Statistics, University of Delhi, Delhi and Dr. Ankita Bidalia for their help in statistical analyses. GA is grateful to University Grants Commission for providing fellowship as a Senior Research Fellow.
Funding
The study was financially supported by Department of Science and Technology (DST-Purse II), Government of India. GA performed the experiments and analysed the data. The work was designed and planned by RK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no potential conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11274_2018_2524_MOESM1_ESM.pdf
Supplementary Figure 1: Map showing collection sites for A. carthami isolates from 24 districts of three major safflower producing states of India. The numerals in each district represent the number of isolates collected from the infected safflower fields of the region. (PDF 138 KB)

11274_2018_2524_MOESM2_ESM.jpg
Supplementary Figure 2: Activity of Pectin methyl esterases in (A) Different isolates of A. carthami. In all plots, data is presented as means ±S.D. (n=5); (B) Box-plot depicting the range of PME activity in A. carthami isolates. (JPG 72 KB)

11274_2018_2524_MOESM3_ESM.jpg
Supplementary Figure 3: Activity of Polygalacturonases in (A) Different isolates of A. carthami. In all plots, data are presented as means ±S.D. (n=5); (B) Box-plot depicting the range of PG activity in A. carthami isolates. (JPG 67 KB)

11274_2018_2524_MOESM4_ESM.jpg
Supplementary Figure 4: Activity of Exo-1,4 glucanases (C1) in (A) Different isolates of A. carthami. In all plots, data are presented as means ±S.D. (n=5) ; (B) Box-plot depicting the range of exo-1,4 glucanases activity in A. carthami isolates. (JPG 66 KB)

11274_2018_2524_MOESM5_ESM.jpg
Supplementary Figure 5: Activity of C, 1,4-endoglucanases (Cx) in (A) Different isolates of A. carthami. In all plots, data are presented as means ±S.D. (n=5); (B) Box-plot depicting the range of C, 1,4-endoglucanases activity in A. carthami isolates. (JPG 75 KB)

11274_2018_2524_MOESM6_ESM.jpg
Supplementary Figure 6: Average lesion area formed by A. carthami isolates on leaves of safflower. In all plots, data are presented as means ±S.D. (n=5). (JPG 177 KB)
Rights and permissions
About this article
Cite this article
Anand, G., Kapoor, R. Population structure and virulence analysis of Alternaria carthami isolates of India using ISSR and SSR markers. World J Microbiol Biotechnol 34, 140 (2018). https://doi.org/10.1007/s11274-018-2524-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-018-2524-6