Abstract
Modified montmorillonite has been widely used for the adsorption of heavy metal Cd(II), but there are few studies on the preparation of adsorbents that are easy to recover and can be efficiently adsorbed. In this paper, magnetic S-MFe2O4/MMT (M=Fe, Zn) composites with huge specific surface multi-class functional groups were prepared on montmorillonite loaded with Zn and S doubly-modified Fe3O4 to explore its performance in Cd(II)-contamination, to reveal the removal mechanism of Cd contamination, and to evaluate the stabilization effect of S-MFe2O4/MMT (M=Fe, Zn). The results showed that the optimum adsorption pH of S-ZnFe2O4/MMT for single Cd(II) was 6.0, the maximum adsorption was 56.68 mg·g−1, and the adsorption process was by the quasi-secondary kinetic model and Langmuir isothermal adsorption model. In addition, the effects of the coexistence of Cr3+, Ni2+, and Pb2+ on the adsorption performance of Cd(II) were not significant, and the adsorption of Cd(II) remained above 55.70 mg·g−1 and showed better adsorption of Pb2+ (98.20 mg·g−1) and Cr3+ (92.78 mg·g−1). In conclusion, the composites can be used as an effective remediation reagent for Cd(II)-contaminated soil.
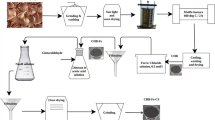
Graphical Abstract














Similar content being viewed by others
References
An, T., Chang, Y., Xie, J., et al. (2022). Deciphering physicochemical properties and enhanced microbial electron transfer capacity by magnetic biochar. Bioresource Technology, 363, 1–11. https://doi.org/10.1016/j.biortech.2022.127894
Bai, Q. Q. (2021). Geochemical characteristics and evaluation of a sewage irrigation site in Hebei province (pp. 1–78). Hebei GEO University. https://doi.org/10.27752/d.cnki.gsjzj.2021.000033
Bezbaruah, A. N., Shanbhogue, S. S., Simsek, S., et al. (2011). Encapsulation of iron nanoparticles in alginate biopolymer for trichloroethylene remediation. Journal of Nanoparticle Research, 13(12), 6673–6681. https://doi.org/10.1007/s11051-011-0574-x
Da, M. X., Ze, L. X., Zi, Q. M., et al. (2023). Mechanistic insights into the migration behavior of cadmium (Cd) in the soil–groundwater systems at a construction site: Experimental and numerical analysis. Journal of Environmental Chemical Engineering, 11(03), 1–12. https://doi.org/10.1016/j.jece.2023.109712
Ding, K. L., Zhou, X. Y., Lu, Y. L., et al. (2021). Removal performance and mechanisms of toxic hexavalent chromium (Cr (VI)) with ZnCl2 enhanced acidic vinegar residue biochar. Journal of Hazardous Materials, 420, 126551. https://doi.org/10.1016/j.jhazmat.2021.126551
Du, J. K., Bao, J. G., Lu, C. H., et al. (2016). Reductive sequestration of chromate by hierarchical FeS@Fe0 particles. Water Research, 102, 73–81. https://doi.org/10.1016/j.watres.2016.06.009
Duan, L., Wang, Q., et al. (2022). Zero valent iron or Fe3O4-loaded biochar for remediation of Pb contaminated sandy soil: Sequential extraction, magnetic separation, XAFS and ryegrass growth. Environmental Pollution, 308. https://doi.org/10.1016/j.envpol.2022.119702
Hosain, A. N. A., Nemr, A. E., Sikaily, A. E., et al. (2020). Surface modifications of nano chitosan-coated magnetic nanoparticles and their applications in Pb(II), Cu(II), and Cd(II) removal. Journal of Environmental Chemical Engineering, 8(05), 104316. https://doi.org/10.1016/j.jece.2020.104316
Hu, J. X., Wang, Y. G., Men, J. R., et al. (2021). Improving the photostriction of BiFeO3-based ceramics by bandgap and domain size engineering. Ceramics International, 48(04), 5145–5153. https://doi.org/10.1016/j.ceramint.2021.11.053
Huang, W., Zhuang, R. H., Liu, H., et al. (2022). Recent advances of the current situation and remediation methods of cadmium contamination in paddy soil. Journal of Natural Science of Hunan Normal University, 45(01), 49–56. https://doi.org/10.7612/j.issn.2096-5281.2022.01.006
Huang, Y., Wang, L., Wang, W., et al. (2019). Current status of agricultural soil pollution by heavy metals in China: A meta-analysis. Science of the Total Environment, 651(02), 3034–3042. https://doi.org/10.1016/j.scitotenv.2018.10.185
Jeong, H. Y., Han, Y.-S., Park, S. W., et al. (2010). Aerobic oxidation of mackinawite (FeS) and its environmental implication for arsenic mobilization. Geochimica et Cosmochimica Acta, 74(11), 3182–3198. https://doi.org/10.1016/j.gca.2010.03.012
Leong, Y. K., & Chang, J.-S. (2020). Bioremediation of heavy metals using microalgae: Recent advances and mechanisms. Bioresource Technology, 303, 122886. https://doi.org/10.1016/j.biortech.2020.122886
Li, X., Xie, S. Y., Zeng, G. M., et al. (2022). Research progress on the application of magnetic nanomaterials in water pollution control. Mini-Review Article, 20(03), 240–249. https://doi.org/10.2174/1570193x19666220328162619
Li, X. Y., Feng, L. B., Lu, G. X., et al. (1995). Preparation and characterization of nanocrystalline ZnFe2O4 particles and EPR study on their properties. Chemical Journal of Chinese Universities, 16(10), 1495–1499. https://doi.org/10.1139/v95-214
Lingamdinne, L. P., Godlaveeti, S. K., Angara, G. K. R., et al. (2022). Highly efficient surface sequestration of Pb2+ and Cr3+ from water using a Mn3O4 anchored reduced graphene oxide: Selective removal of Pb2+ from real water. Chemosphere, 299, 1–10. https://doi.org/10.1016/j.chemosphere.2022.134457
Loukili, H., Mabroukic, J., Anouzla, A., et al. (2021). Pre-treated Moroccan natural clays: Application to the wastewater treatment of textile industry. Desalination and Water Treatment, 240, 124–136. https://doi.org/10.5004/dwt.2021.27644
Lu, L., Wang, R. C., Chen, F. R., et al. (2005). Element mobility during pyrite⁃weathering: Implications for acid and heavy metal pollution at mining-impacted sites. Environmental geology, 49(01), 82–89. https://doi.org/10.1007/s00254-005-0061-8
Lv, L. Q., Wang, J. K., Ji, M. T., et al. (2022). Effect of structural characteristics and surface functional groups of biochar on thermal properties of different organic phase change materials: Dominant encapsulation mechanisms. Renewable Energy, 195, 1238–1252. https://doi.org/10.1016/j.renene.2022.06.117
Ma, S., Xiong, S. L., Xiong, L., et al. (2019). Adsorption efficiency of cadmium and arsenic by iron-modified sepiolite and it is influencing factors. Technology of Water Treatment, 45(10), 73–77. https://doi.org/10.16796/j.cnki.1000-3770.2019.10.015
Martínez-Quiroz, M., López-Maldonado, E. A., Ochoa-Terán, A., et al. (2017). Modification of chitosan with carbamoyl benzoic acids for testing its coagulant-flocculant and binding capacities in the removal of metallic ions typically contained in plating wastewater. Chemical Engineering Journal, 332(15), 749–756. https://doi.org/10.1016/j.cej.2017.09.042
Ainiwaer, M., Zhang, T., Zhang, N., et al. (2022). Synergistic removal of As(III) and Cd(II) by sepiolite-modified nanoscale zero-valent iron and a related mechanistic study. Journal of Environmental Management, 319, 1–9. https://doi.org/10.1016/j.jenvman.2022.115658
Ni, X. M., Li, Z. H., & Wang, Y. B. (2018). Adsorption characteristics of anionic surfactant sodium dodecylbenzene sulfonate on the surface of montmorillonite minerals. Frontiers in Chemistry, 6(02). https://doi.org/10.3389/fchem.2018.00390
Ni, X. M., Zhao, Z., Li, Z. H., et al. (2021). The adsorptive behavior of kaolinite to sodium dodecyl benzene sulphonate and the structural variation of kaolinite. Scientific Reports, 19, 1–11. https://doi.org/10.1038/s41598-021-81283-8
Peng, W., Li, X., Song, J., et al. (2018). Bioremediation of cadmium- and zinc-contaminated soil using rhodobacter sphaeroides. Chemosphere, 197(33), 33–41. https://doi.org/10.1016/j.chemosphere.2018.01.017
Quezada, G. R., Rozas, R. E., & Toledo, P. G. (2019). Ab initio calculations of partial charges at kaolinite edge sites and molecular dynamics simulations of cation adsorption in saline solutions at and above the pH of zero charge. The Journal of Physical Chemistry C, 123(37), 22971–22980. https://doi.org/10.1021/acs.jpcc.9b05339
Rizwan, M., Ali, S., Zia Ur Rehman, M., et al. (2018). Cadmium phytoremediation potential of Brassica crop species: A review. Science of the Total Environment, 631-632(01), 1175–1191. https://doi.org/10.1016/j.scitotenv.2018.03.104
Sharma, P., Prakash, J., Palai, T., et al. (2022). Surface functionalization of bamboo leave mediated synthesized SiO2 nanoparticles: Study of adsorption mechanism, isotherms and enhanced adsorption capacity for removal of Cr(VI) from aqueous solution. Environmental Research, 214(Pt 1), 113761. https://doi.org/10.1016/j.envres.2022.113761
Song, X. M., Tan, L. C., Ma, H. Y., et al. (2017). Facile preparation of S-doped magnetite hollow spheres for highly efficient sorption of uranium(VI). Dalton Transactions, 46(10), 3347–3352. https://doi.org/10.1039/c7dt00209b
Sun, Y., Lou, Z. M., Yu, J. B., et al. (2017). Immobilization of mercury(II) from aqueous solution using Al2O3-supported nanoscale FeS. Chemical Engineering Journal, 323, 483–491. https://doi.org/10.1016/j.cej.2017.04.095
Sun, Y. B., Sun, G. H., Xu, Y. M., et al. (2015). Evaluation of the effectiveness of sepiolite, bentonite, and phosphate amendments on the stabilization remediation of cadmium-contaminated soils. Journal of Environmental Management, 166(15), 204–210. https://doi.org/10.1016/j.jenvman.2015.10.017
Tian, X. Z., Jin, L. S., Ying, B., et al. (2015). Adsorption of Cd2+ from aqueous solution by steel slag-montmorillonite composite adsorbent. Acta Scientiae Circumstantiae, 35(01), 207–214. https://doi.org/10.13671/j.hjkxxb.2014.0753
Yan, N. N., Hu, B., Zheng, Z. Y., et al. (2023). Twice-milled magnetic biochar: A recyclable material for efficient removal of methylene blue from wastewater. Bioresource Technology, 372(03), 1–8. https://doi.org/10.1016/j.biortech.2023.128663
Yan, Z. M., Wang, Y. P., Ding, J. Q., et al. (2017). Methoxyl modification in furo[3, 2-c]pyridine-based iridium complexes towards highly efficient green- and orange-emitting electrophosphorescent devices. Journal of Materials Chemistry C, 5, 46. https://doi.org/10.1039/c7tc04269h
Yang, K., Peng, H. B., Wen, Y. H., et al. (2010). Re-examination of characteristic FTIR spectrum of a secondary layer in bilayer oleic acid-coated Fe3O4 nanoparticles. Applied Surface Science, 256(10), 3093–3097. https://doi.org/10.1016/j.apsusc.2009.11.079
Yang, X., Peng, Q. C., Liu, L. H., et al. (2020). Synergistic adsorption of Cd(II) and As(V) on birnessite under electrochemical control. Chemosphere, 247, 125822. https://doi.org/10.1016/j.chemosphere.2020.125822
Zeng, Z. L., Yu, C., Liao, R. P., et al. (2023). Preparation and characterization of sodium polyacrylate grafted montmorillonite nanocomposite for the adsorption of cadmium ions form aqueous solution. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 656, 1–12. https://doi.org/10.1016/j.colsurfa.2022.130389
Zheng, X. M., Dou, J. F., Jing, Y., et al. (2017). Removal of Cs+ from water and soil by ammonium-pillared montmorillonite/Fe3O4 composite. Journal of Environmental Sciences, 56(06), 12–24. https://doi.org/10.1016/j.jes.2016.08.019
Author Contributions
Yan Lei: data analysis, writing—original draft. Tingting Huo, Huanbo Wang: conceptualization, supervision, writing—review, and editing. Tunan Yin: read, corrected, and revised the manuscript. Xin Xiao: writing—review and editing. Yuancong Ma: writing—review and editing. Mengqing Liu: data analysis, writing—review and editing. All authors read and approved the final manuscript.
Data Availability Statements
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Funding
This work was supported by the Nature Science Foundation of Sichuan Province (2022NSFSC1223) and the National Natural Science Foundation of China (42007281).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• S-ZnFe2O4/MMT composites were successfully fabricated by hydrothermal synthesis of Fe3O4 modified by both Zn and S.
• S-ZnFe2O4/MMT composites showed a significant increase in the adsorption of Cd(II).
• The adsorption performance of the composites on Cd(II) was less affected by the coexistence of multiple cations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lei, Y., Huo, T., Yin, T. et al. Magnetic Sulfide–Modified MFe2O4/Montmorillonite (M=Fe, Zn) for Efficient Sorption of Cd(II). Water Air Soil Pollut 235, 82 (2024). https://doi.org/10.1007/s11270-023-06876-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-023-06876-7