Abstract
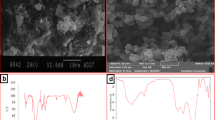
Untreated arsenic polluted groundwater is threatening people health, especially the people in rural areas. Soil may become one kind of promising natural material applied conveniently in rural areas for the treatment of arsenic polluted groundwater, due to its abundance, low cost, and high adsorption efficiency. The present study investigated arsenic(V) (As(V)) adsorption on two red soil samples weathered from granite (RSG) and sandstone (RSS). The two soil samples contain similar mineral types but show relatively high differences of content of iron, aluminum, and organic matter (OM), as well as point of zero charge (pHPZC) and specific surface area (SSA). Batch experiments were performed to examine the effect of initial As(V) concentration, solution pH, and temperature on the kinetics of the adsorption of As(V) by the two soil samples. The experimental results showed that the As(V) adsorption onto the two soil samples was influenced by the physicochemical properties of the soils, especially the content of iron and aluminum, the OM, as well as the pHPZC, and chemisorption was the main adsorption mechanism. The RSG sample with higher content of iron and aluminum and pHPZC showed relatively high adsorption efficiency. The OM played a negative role in the adsorption process, especially as the As/Fe molar ratio is higher. Higher adsorption capacities for the two soil samples were both obtained at lower initial As(V) concentration (1.50 and 4.0 mg/l), lower pH value (5.0), and higher temperature (313 K). Comparing to the RSS, the RSG is more suitable for the treatment of As(V)-polluted groundwater. Considering the experimental results and the natural conditions, the suggested operational conditions are pH around 7.0, temperature 293–303 K, As(V) concentration less than 4.0 mg/l, and hydraulic retention time no less than 180 min.





Similar content being viewed by others
References
Ahoulé, D. G., Lalanne, F., Mendret, J., Brosillon, S., & Maïga, A. H. (2015). Arsenic in African Waters: A Review. Water, Air, & Soil Pollution, 226, 302–314.
Aide, M., Beighley, D., & Dunn, D. (2016). Arsenic in the soil environment: a soil chemistry review. International Journal of Applied Agricultural Research, 11(1), 1–28.
Al Lawati, W. M., Rizoulis, A., Eiche, E., Boothman, C., Polya, D. A., Lloyd, J. R., Berg, M., Vasquez-Aguilar, P., & van Dongen, B. E. (2012). Characterization of organic matter and microbial communities in contrasting arsenic-rich Holocene and arsenic poor Pleistocene aquifers, Red River Delta, Vietnam. Applied Geochemistry, 27, 315–325.
Alshaebi, F. Y., Yaacob, W. Z. W., & Samsudin, A. R. (2010). Removal of Arsenic from Contaminated Water by Selected Geological Natural materials. Australian Journal of Basic & Applied Sciences, 4(9), 4413–4422.
Andjelkovic, I., Tran, D. H., Kaviri, S., Azari, S., Markovic, M., & Losic, D. (2015). Graphene aerogels decorated with α-FeOOH nanoparticles for efficient adsorption of arsenide from contaminated waters. Applied Materials & Interfaces, 7, 9758–9766.
Bentahar, Y., Hurel, C., Draoui, K., Khairoun, S., & Marmier, N. (2016). Adsorptive properties of Moroccan clays for the removal of arsenic(V) from aqueous solution. Applied Clay Science, 119, 385–392.
Chakraborti, D., Rahman, M. M., Ahamed, S., Dutta, R. N., Pati, S., & Mukherjee, S. C. (2016). Arsenic groundwater contamination and its health effects in Patna district (capital of Bihar) in the middle Ganga plain, India. Chemosphere, 152, 520–529.
Chen, H.W., Chen, H.H., Liu, L., Liu, F., Gong, R.Y., Wei, R.C., & Yi, Q.H. (2016). Impacts of Physicochemical Properties of Soil on Arsenic(V) Removal from Aqueous Phase: Adsorption Comparison Between Soils Weathering from Granite and Sandstone. Applied Clay Science, Submitted.
Chowdhury, S. R., & Yanful, E. K. (2010). Arsenic and chromium removal by mixed magnetite-maghemite nanoparticles and the effect of phosphate on removal. Journal of Environmental Management, 91, 2238–2247.
Dai, M., Xia, L., Song, S. X., Peng, C. S., & Lopez-Valdivieso, A. (2016). Adsorption of As(V) inside the pores of porous hematite in water. Journal of Hazardous Materials, 307, 312–317.
De Oliveira, L. K., Melo, C. A., Goveia, D., Lobo, F. A., Hernández, M. A. A., Fraceto, L. F., & Rosa, A. H. (2015). Adsorption/desorption of arsenic by tropical peat: influence of organic matter, iron and aluminium. Environmental Technology, 36(2), 149–159.
Dousova, B., Buzek, F., Rothwell, J., Krejcova, S., & Lhotka, M. (2012). Adsorption behavior of arsenic relating to different natural solids: soils, stream sediments and peats. Science of the Total Environment, 433, 456–461.
Du, J. J., Jing, C. Y., Duan, J. M., Zhang, Y. L., & Hu, S. (2014). Removal of arsenate with hydrous ferric oxide coprecipitation: Effect of humic acid. Journal of Environmental Sciences, 26, 240–247.
Escudero, C., Fiol, N., Villaescusa, I., & Bollinger, J. C. (2009). Arsenic removal by waste metal (hydr)oxide entrapped into calcium alginate beads. Journal of Hazardous Materials, 164, 533–541.
Feng, Q. Z., Zhang, Z. Y., Chen, Y., Liu, L. Y., Zhang, Z. J., & Chen, C. Z. (2013). Adsorption and desorption characteristics of arsenic on soils: kinetics, equilibrium, and effect of Fe(OH)3 colloid, H2SiO3 colloid and phosphate. Procedia Environmental Sciences, 18, 26–36.
Ho, Y. S., & McKay, G. (1999). Pseudo-second order model for sorption processes. Process Biochemistry, 34, 451–465.
Khezami, L., & Capart, R. (2005). Removal of chromium(VI) fromaqueous solution by activated carbons: kinetic and equilibrium studies. Journal of Hazardous Materials, B123, 223–231.
Kim, E. J., Yoo, J.-C., & Baek, K. (2014). Arsenic speciation and bioaccessibility in arsenic-contaminated soils: Sequential extraction and mineralogical investigation. Environmental Pollution, 186, 29–35.
Kim, E. J., Hwang, B. R., & Baek, K. (2015). Effects of natural organic matter on the coprecipitation of arsenic with iron. Environmental Geochemistry & Health, 37(6), 1029–1039.
Kumar, R., Kumar, R., Mittal, S., Arora, M., & Babu, J. N. (2016). Role of soil physicochemical characteristics on the present state of arsenic and its adsorption in alluvial soils of two agri-intensive region of Bathinda, Punjab, India. Journal of Soils & Sediments, 16(2), 605–620.
Kwon, M. S., Lee, M. G., Mok, Y. G., & Chung, J. W. (2013). Adsorption of Decomposed-Granite Soils Varying with Weathering on Heavy Metals. Journal of the Korean Geoenvironmental Society, 14(10), 59–64.
Lagergren, S. (1898). About the theory of so-called adsorption of soluble substances, Kungliga Svenska Vetenskapsakademiens Handlingar, 24 (4), 1–39.
Lawson, M., Polya, D. A., Boyce, A. J., Bryant, C., & Ballentine, C. J. (2016). Tracing organic matter composition and distribution and its role on arsenic release in shallow Cambodian groundwaters. Geochimica et Cosmochimica Acta, 178, 160–177.
Li, P. C., Xu, D. R., Chen, G. H., Xia, B., He, Z. L., & Fu, G. G. (2005). Constraints of petrography, geochemistry and Sr-Nd isotopes on the Jinjing granitoids from northeastern Hunan province, China: implications for petrogenesis and geodynamic setting. Acta Petrologica Sinica, 21(3), 921–934.
Liu, R. P., Zhu, L. J., He, Z., Lan, H. C., Liu, H. J., & Qu, J. H. (2015). Simultaneous removal of arsenic and fluoride by freshly-prepared aluminum hydroxide. Colloids and Surfaces A: Physicochemical & Engineering Aspects, 466, 147–153.
Liu, Y. K., Hu, P., Zheng, J. T., Wu, M. B., & Jiang, B. (2016). Utilization of spent aluminum for p-arsanilic acid degradation and arsenic immobilization mediated by Fe(II) under aerobic condition. Chemical Engineering Journal, 297, 45–54.
Ma, J., Guo, H. M., Lei, M., Zhou, X. Y., Li, F. L., Yu, T., Wei, R. F., Zhang, H. Z., Zhang, X., & Yang, W. (2015). Arsenic Adsorption and its Fractions on Aquifer Sediment: Effect of pH, Arsenic Species, and Iron/Manganese Minerals. Water, Air, & Soil Pollution, 226, 260–274.
Maiti, A., Basu, J. K., & De, S. (2010). Removal of Arsenic from Synthetic and Natural Groundwater Using Acid-Activated Laterite. Environmental Progress & Sustainable Energy, 29(4), 457–470.
Mak, M. S. H., & Lo, I. M. C. (2011). Influences of redox transformation, metal complexation and aggregation of fulvic acid and humic acid on Cr(VI) and As(V) removal by zero-valent iron. Chemosphere, 84, 234–240.
Mamindy-Pajany, Y., Hurel, C., Marmier, N., & Roméo, M. (2009). Arsenic adsorption onto hematite and goethite. Comptes Rendus Chimie, 12(8), 876–881.
Mertens, J., Rose, J., Wehrli, B., & Furrer, G. (2016). Arsenate uptake by Al nanoclusters and other Al-based sorbents during water treatment. Water Research, 88, 844–851.
Meunier, L., Koch, I., & Reimer, K. J. (2011). Effects of organic matter and ageing on the bioaccessibility of arsenic. Environmental Pollution, 159, 2530–2536.
Mikutta, R., Lorenz, D., Guggenberger, G., Haumaier, L., & Freund, A. (2014). Properties and reactivity of Fe-organic matter associations formed by coprecipitation versus adsorption: Clues from arsenate batch adsorption. Geochimica et Cosmochimica Acta, 144, 258–276.
Panagiotaras, D., & Nikolopoulos, D. (2015). Arsenic Occurrence and Fate in the Environment; A Geochemical Perspective. Journal of Earth Science & Climatic Change, 6(4), 1–9.
Panagiotaras, D., Papoulis, D., & Stathatos, E. (2015). Origin of Arsenic Toxicity–Geochemistry. Chapter 4. In Narayan Chakrabarty (Ed.), Arsenic Toxicity: Prevention and Treatment. Boca Raton: CRC press, Taylor & Francis publishers.
Paul, S., Chakraborty, S., Ali, M. N., & Ray, D. P. (2015). Arsenic distribution in environment and its bioremediation: A review. International Journal of Agriculture, Environment & Biotechnology, 8(1), 189–204.
Postma, D., Larsen, F., Thai, N. T., Trang, P. T. K., Jakobsen, R., Nhan, P. Q., Long, T. V., Viet, P. H., & Murray, A. S. (2012). Groundwater arsenic concentrations in Vietnam controlled by sediment age. Nature Geoscience, 5, 656–661.
Ranasinghe, R. M. S. C., Werellagama, D. R. I. B., & Weerasooriya, R. (2014). Arsenite removal from drinking water using naturally available laterite in Sri Lanka. Engineer, 17(2), 23–31.
Rodríguez-Lado, L., Sun, G. F., Michael, B., Zhang, Q., Xue, H. B., Zheng, Q. M., & Johnson, C. A. (2013). Groundwater arsenic contamination throughout China. Science, 341, 866–868.
Rowland, H. A. L., Gault, A. G., Lythgoe, P., & Polya, D. A. (2008). Geochemistry of aquifer sediments and arsenic-rich groundwaters from Kandal Province, Cambodia. Applied Geochemistry, 23(11), 3029–3046.
Sigrist, M., Albertengo, A., Brusa, L., Beldoménico, H., & Tudino, M. (2013). Distribution of inorganic arsenic species in groundwater from Central-West Part of Santa Fe Province, Argentina. Applied Geochemistry, 39, 43–48.
Singh, R., Singh, S., Parihar, P., Singh, V. P., & Prasad, S. M. (2015). Arsenic contamination, consequences and remediation techniques: A review. Ecotoxicology & Environmental Safety, 112, 247–270.
Smedley, P. L., & Kinniburgh, D. G. (2002). A review of the source, behavior and distribution of arsenic in natural waters. Applied Geochemistry, 17, 517–568.
Smith, E., & Naidu, R. (2009). Chemistry of inorganic arsenic in soils: kinetics of arsenic adsorption desorption. Environmental Geochemistry & Health, 31, 49–59.
Sutherland, C. (2004). Removal of heavy metals from waters using low cost adsorbents: process development. PhD Thesis. The University of the West Indies, Trinidad, p. 150–164.
Tan, B. X., & Wang, G. X. (1983). On the geologic age of the “Yuelu sandstone”. Geological Review, 29(6), 553–561.
Tareq, S. M., Maruo, M., & Ohta, K. (2013). Characteristics and role of groundwater dissolved organic matter on arsenic mobilization and poisoning in Bangladesh. Physics & Chemistry of the Earth, 58–60, 77–84.
Weber, W. J., & Morris, I. C. (1963). Kinetics of adsorption on carbon from solution. Journal of the Sanitary Engineering Division, American Society of Civil Engineers, 89(2), 31–60.
Yazdani, M., Tuutijärvi, T., Bhatnagar, A., & Vahala, R. (2016). Adsorptive removal of arsenic from aqueous phase by feldspars: Kinetics, mechanism, and thermodynamic aspects of adsorption. Journal of Molecular Liquids, 214, 149–156.
Acknowledgments
The authors would like to send sincere appreciation to China Hunan Provincial Science and Technology Department for the funds and the reviewers for their good proposals for revision of the article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, H., Liu, L., Gong, R. et al. Comparison of Kinetics of Arsenic(V) Adsorption on Two Types of Red Soil Weathered from Granite and Sandstone. Water Air Soil Pollut 227, 406 (2016). https://doi.org/10.1007/s11270-016-3107-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-016-3107-5