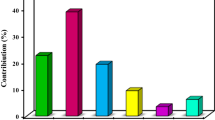
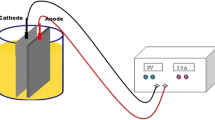
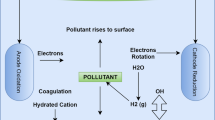
Abstract
In this study, electrocoagulation (EC) with hybrid Fe–Al electrodes was used to remove antimony from contaminated surface water. Response surface methodology was applied to investigate the interactive effects of the operating parameters on antimony removal and optimize these variables. Results showed that the relationship between operating parameters and the response was well described by a second-order polynomial equation. Under the optimal conditions of current density 2.58 mA/cm2, pH 5.24, initial concentration 521.3 μg/L, and time 89.17 min, more than 99 % antimony were removed. Besides, the antimony adsorption behavior in EC process was also investigated. Adsorption kinetics and isotherms studies suggested that the adsorption process followed well the pseudo-second-order kinetic model and the Langmuir adsorption model, respectively. Adsorption thermodynamics study revealed that the reaction was spontaneous, endothermic, and thermodynamically favorable. These results further proved that the main mechanism involved in antimony removal in EC process could be chemisorption.





Similar content being viewed by others
References
Ahmadi, M., Vahabzadeh, F., Bonakdarpour, B., Mofarrah, E., & Mehranian, M. (2005). Application of the central composite design and response surface methodology to the advanced treatment of olive oil processing wastewater using Fenton’s peroxidation. J Hazard Mater, 123(1-3), 187–195.
Allen, S. J. (1996). Types of adsorbent materials-use of adsorbents for removal of pollutants from wastewaters (p. 59). Boca Raton, FL, USA: CRC.
Aydin, Y. A., & Aksoy, N. D. (2009). Adsorption of chromium on chitosan: optimization, kinetics and thermodynamics. Chem Eng J, 151(1-3), 188–194.
Balasubramanian, N., Kojima, T., & Srinivasakannan, C. (2009). Arsenic removal through electrocoagulation: kinetic and statistical modeling. Chem Eng J, 155(1-2), 76–82.
Bhatti, M. S., Reddy, A. S., Kalia, R. K., & Thukral, A. K. (2011). Modeling and optimization of voltage and treatment time for electrocoagulation removal of hexavalent chromium. Desalination, 269(1-3), 157–162.
Boparai, H. K., Joseph, M., & O’Carroll, D. M. (2011). Kinetics and thermodynamics of cadmium ion removal by adsorption onto nano zerovalent iron particles. J Hazard Mater, 186(1), 458–465.
CEC (Council of the European Communities). (1976). Council Directive 76/substances discharged into aquatic environment of the community. Off J L, 129, 23–29.
CEC (Council of the European Communities). (1980). Council Directive relating to the quality of water intended for human consumption. 80/778/EEC.
CEPA. (2002). Water and wastewater monitoring analysis methods (4th ed.). China Environmental Science: Beijing.
Chen, X. M., Chen, G. H., & Yue, P. L. (2000). Separation of pollutants from restaurant wastewater by electrocoagulation. Sep Purif Technol, 19(1-2), 65–76.
Chien, S. H., & Clayton, W. R. (1980). Application of Elovich equation to the kinetics of phosphate release and sorption in soils. Soil Sci Soc Am J, 44(2), 265–268.
Dou, X. M., Mohan, D., Zhao, X. Q., & Pittman, C. U. (2015). Antimonate removal from water using hierarchical macro-/mesoporous amorphous alumina. Chem Eng J, 264, 617–624.
Feng, J., Yang, Z. H., Zeng, G. M., Huang, J., Xu, H. Y., Zhang, Y. Y., Wei, S. H., & Wang, L. K. (2013). The adsorption behavior and mechanism investigation of Pb(II) removal by flocculation using microbial flocculant GA1. Bioresource Technol, 148, 414–421.
Filella, M., Belzile, N., & Chen, Y. W. (2002). Antimony in the environment: a review focused on natural waters. I. Occurrence. Earth Sci Rev, 57(1-2), 125–176.
Ganesan, P., Lakshmi, J., Sozhan, G., & Vasudevan, S. (2013). Removal of manganese from water by electrocoagulation: adsorption, kinetics and thermodynamic studies. Can J Chem Eng, 91(3), 448–458.
Gomes, J. A. G., Daida, P., & Kesmez, M. (2007). Arsenic removal by electrocoagulation using combined Al-Fe electrode system and characterization of products. J Hazard Mater, 139(10), 220–231.
Guo, X. J., Wu, Z. J., & He, M. C. (2009). Removal of antimony(V) and antimony(III) from drinking water by coagulation-flocculation-sedimentation (CFS). Water Res, 43(17), 4327–4335.
Guo, X. J., Wu, Z. J., He, M. C., Meng, X. G., Jin, X., Qiu, N., & Zhang, J. (2014). Adsorption of antimony onto iron oxyhydroxides: adsorption behavior and surface structure. J Hazard Mater, 276, 339–345.
Gupta, S. S., & Bhattacharyya, K. G. (2006). Adsorption of Ni(II) on clays. J Colloid Interface Sci, 295(1), 21–32.
Ho, Y. S., & McKay, G. (1998). A comparison of chemisorption kinetic models applied to pollutant removal on various sorbents. Proc Saf Environ Prot, 76(4), 332–340.
Holt, P. K., Barton, G. W., & Mitchell, C. A. (2005). The future for electrocoagulation as a localised water treatment technology. Chemosphere, 59(3), 355–367.
Isa, M. H., Ezechi, E. H., Ahmed, Z., Magram, S. F., & Kutty, S. R. M. (2014). Boron removal by electrocoagulation and recovery. Water Res, 51, 113–123.
Kobya, M., Can, O. T., & Bayramoglu, M. (2003). Treatment of textile wastewaters by electrocoagulation using iron and aluminum electrodes. J Hazard Mater, 100(1-3), 163–178.
Kobya, M., Gebologlu, U., Ulu, F., & Oncel, S. (2011). Removal of arsenic from drinking water by the electrocoagulation using Fe and Al electrodes. Electrochim Acta, 56(14), 5060–5070.
Kobya, M., Demirbas, E., Gebologlu, U., Oncel, M. S., & Yildirim, Y. (2013). Optimization of arsenic removal from drinking water by electrocoagulation batch process using response surface methodology. Desalin Water Treat, 51(34-36), 1–10.
Lai, C. L., & Lin, S. H. (2003). Electrocoagulation of chemical mechanical polishing (CMP) wastewater from semiconductor fabrication. Chem Eng J, 95(1-3), 205–211.
Mall, I. D., Srivastava, V. C., Agarwal, N. K., & Mishra, I. M. (2005). Removal of Congo Red from aqueous solution by bagasse fly ash and activate d carbon: kinetic study and equilibrium isotherm analysis. Chemosphere, 61(4), 492–501.
Mall, I. D., Srivastava, V. C., Agarwal, N. K., & Mishra, I. M. (2007). Removal of Congo Red on coal-based mesoporous activated carbon. Dyes Pigm, 74(1), 34–40.
Myers, R. H., & Montgomery, D. C. (2002). Response surface methodology: process and product optimization using designed experiments (2nd ed.). New York: John Wiley and Sons.
Ölmez, T. (2009). The optimization of Cr(VI) reduction and removal by electrocoagulation using response surface methodology. J Hazard Mater, 162(2-3), 1371–1378.
Qu, J. H., & Liu, H. J. (2007). Electrochemical principles and techniques in water treatment (pp. 205–209). Beijing: Science Press.
SAC (Standardization Administration of the People’s Republic of China). (2006). Standards for drinking water quality. Beijing: SAC (Standardization Administration of the People’s Republic of China). GB-5749-2006.
Song, P. P., Yang, Z. H., Xu, H. Y., Huang, J., Yang, X., & Wang, L. K. (2014). Investigation of influencing factors and mechanism of antimony and arsenic removal by electrocoagulation using Fe–Al electrodes. Ind Eng Chem Res, 53(33), 12911–12919.
Sundaram, C. S., Viswanathan, N., & Meenakshi, S. (2008). Defluoridation chemistry of synthetic hydroxyapatite at nanoscale: equilibrium and kinetic studies. J Hazard Mater, 155(1-2), 206–215.
USEPA. (1979). Water related fate of the 129 priority pollutants. Washington, DC: USEPA. Doc., 745-R-00-007.
USEPA. (1984). Antimony: an environmental and health effects assessment. Washington, DC: US Environmental Protection Agency. Office of drinking water.
Vasudevan, S., & Oturan, M. A. (2014). Electrochemistry: as cause and cure in water pollution—an overview. Environ Chem Lett, 12(1), 97–108.
Ville, K., Toivo, K., Jaakko, R., & Ulla, L. (2013). Recent applications of electrocoagulation in treatment of water and wastewater-a review. Green and Sus Chem, 3, 89–121.
Vithanage, M., Rajapaksha, A. U., Dou, X. M., Bolan, N. S., Yang, J. E., & Ok, Y. S. (2013). Surface complexation modeling and spectroscopic evidence of antimony adsorption on iron-oxide-rich red earth soils. J Colloid Interf Sci, 406, 217–224.
Wan, W., Pepping, T. J., Banerji, T., Chaudhari, S., & Giammar, D. E. (2011). Effects of water chemistry on arsenic removal from drinking water by electrocoagulation. Water Res, 45(1), 384–392.
Weber, W. J., & Morris, J. C. (1963). Kinetics of adsorption on carbon from solution. Journal of Sanitary Engineering Division (Proceeding of the asce), 89, 31–59.
WHO. (2006). Health criteria and other supporting information in: Guidelines for Drinking-water Quality (Secondth ed.). Geneva: World Health Organization.
Winship, K. A. (1986). Toxicity of antimony and its compounds. Adverse Drug React Acute Poisoning Rev, 6(2), 67–90.
Yang, Z. H., Xu, H. Y., Zeng, G. M., Luo, Y. L., Yang, X., Huang, J., Wang, L. K., & Song, P. P. (2015). The behavior of dissolution/passivation and the transformation of passive films during electrocoagulation: influences of initial pH, Cr(VI) concentration, and alternating pulsed current. Electrochim Acta, 153(2), 149–158.
Yousuf, M., Mollah, A., Schennach, R., & Parga, J. R. (2001). Electrocoagulation (EC) science and applications. J Hazard Mater, 84(1), 29–41.
Zhu, J., Wu, F. C., Pan, X. L., Guo, J. Y., & Wen, D. S. (2011). Removal of antimony from antimony mine flotation wastewater by electrocoagulation with aluminum electrodes. J Environ Sci, 23(7), 1066–1071.
Acknowledgements
We are grateful for the financial support of the National Natural Science Foundation of China (51378189 and 51578223).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 238 kb)
Rights and permissions
About this article
Cite this article
Song, P., Yang, Z., Zeng, G. et al. Optimization, Kinetics, Isotherms, and Thermodynamics Studies of Antimony Removal in Electrocoagulation Process. Water Air Soil Pollut 226, 380 (2015). https://doi.org/10.1007/s11270-015-2615-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-015-2615-z