Abstract
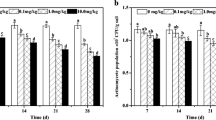
Because of its long half-life, endosulfan can persist for a long time in the environment, especially in soil. However, little is known about its effect on fungi, which is an important part of microorganisms in soil. In this study, agricultural soil treated with endosulfan (0.1 and 1.0 mg kg−1) in a laboratory experiment was analyzed over 42 days. The effect of endosulfan on bacterial and fungal quantity and community structure were determined by quantitative polymerase chain reaction (qPCR) and denaturing gradient gel electrophoresis (DGGE). The results revealed that endosulfan was removed more than 50 % after 42 days, and its removal fitted single first-order kinetics. The exposure to endosulfan caused a short-lived inhibition on fungal and bacterial quantity, but no effect was observed in both treatments after 42 days. Furthermore, this inhibition was greater in higher endosulfan-treated soil. A significant change in bacterial community structure was found in both treatments after endosulfan application, while the change of fungal community structure was observed only in 1 mg kg−1 endosulfan treatment.




Similar content being viewed by others
References
Adebayo, T., Ojo, O., & Olaniran, O. (2007). Effect of two insecticides karate and thiodan on population dynamics of four different soil microorganism. Research Journal of Biological Sciences, 2(5), 557–560.
Beyger, L., Orrego, R., Guchardi, J., & Holdway, D. (2012). The acute and chronic effects of endosulfan pulse-exposure on Jordanella floridae (Florida flagfish) over one complete life-cycle. Ecotoxicology and Environmental Safety, 76, 71–78.
Cotham, W. E., Jr., & Bidleman, T. F. (1989). Degradation of malathion, endosulfan, and fenvalerate in seawater and seawater/sediment microcosms. Journal of Agricultural and Food Chemistry, 37(3), 824–828.
Cremlyn, R. (1979). Pesticides: Preparation and mode of action. New York, NY: John Wiley & Sons. http://www.abebooks.com/Pesticides-Preparation-Mode-Action-R-CREMLYN/1385808514/bd.
Dar, S. A., Yousuf, A. R., Balkhi, M. U., Ganai, F. A., & Bhat, F. A. (2015). Assessment of endosulfan induced genotoxicity and mutagenicity manifested by oxidative stress pathways in freshwater cyprinid fish crucian carp (Carassius carassius L.). Chemosphere, 120, 273–283.
Downing, H. F., Delorenzo, M. E., Fulton, M. H., Scott, G. I., Madden, C. J., & Kucklick, J. R. (2004). Effects of the agricultural pesticides atrazine, chlorothalonil, and endosulfan on South Florida microbial assemblages. Ecotoxicology, 13(3), 245–260.
Ekundayo, E. (2006). Effect of pesticides used in the Niger delta basin of southern Nigerian soils microbial population environmental monitoring and assessment. Environmental Monitoring and Assessment, 89, 1–35.
Engelen, B., Meinken, K., Von Wintzingerode, F., Heuer, H., Malkomes, H. P., & Backhaus, H. (1998). Monitoring impact of a pesticide treatment on bacterial soil communities by metabolic and genetic fingerprinting in addition to conventional testing procedures. Applied and Environmental Microbiology, 64(8), 2814–2821.
Ferreira, E. P. d. B., Dusi, A. N., Costa, J. R., Xavier, G. R., & Rumjanek, N. G. (2009). Assessing insecticide and fungicide effects on the culturable soil bacterial community by analyses of variance of their DGGE fingerprinting data. European Journal of Soil Biology, 45(5), 466–472.
Fierer, N., Jackson, J. A., Vilgalys, R., & Jackson, R. B. (2005). Assessment of soil microbial community structure by use of taxon-specific quantitative PCR assays. Applied and Environmental Microbiology, 71(7), 4117–4120.
Gardes, M., & Bruns, T. D. (1993). ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Molecular Ecology, 2(2), 113–118.
Jha, M., & Mishra, S. (2005a). Biological responses of cyanobacteria to insecticides and their insecticide degrading potential. Bulletin of Environmental Contamination and Toxicology, 75(2), 374–381.
Jha, M., & Mishra, S. (2005b). Decrease in microbial biomass due to pesticide application/residues in soils under different cropping systems. Bulletin of Environmental Contamination and Toxicology, 75(2), 316–323.
Joseph, R., Reed, S., Jayachandran, K., Clark-Cuadrado, C., & Dunn, C. (2010). Endosulfan has no adverse effect on soil respiration. Agriculture, Ecosystems & Environment, 138(3), 181–188.
Kathpal, T. S., Singh, A., Dhankhar, J. S., & Singh, G. (1997). Fate of endosulfan in cotton soil under sub-tropical conditions of northern India. Pesticide Science, 50(1), 21–27.
Kaur, I., Mathur, R. P., Tandon, S. N., & Dureja, P. (1998). Persistence of endosulfan (technical) in water and soil. Environmental Technology, 19(1), 115–119.
Kumar, M., Lakshmi, V. C., & Khanna, S. (2008). Biodegradation and bioremediation of endosulfan contaminated soil. Bioresource Technology, 99(8), 3116–3122.
Li, X., Su, Z., Li, X., Zhang, C., & Zhang, H. (2010). Assessing the effects of acetochlor on soil fungal communities by DGGE and clone library analysis. Ecotoxicology, 19(6), 1111–1116.
Liu, Y.-Q., Moy, B., Kong, Y.-H., & Tay, J.-H. (2010). Formation, physical characteristics and microbial community structure of aerobic granules in a pilot-scale sequencing batch reactor for real wastewater treatment. Enzyme and Microbial Technology, 46(6), 520–525.
May, L. A., Smiley, B., & Schmidt, M. G. (2001). Comparative denaturing gradient gel electrophoresis analysis of fungal communities associated with whole plant corn silage. Canadian Journal of Microbiology, 47(9), 829–841.
Nübel, U., Engelen, B., Felske, A., Snaidr, J., Wieshuber, A., Amann, R. I., et al. (1996). Sequence heterogeneities of genes encoding 16S rRNAs in Paenibacillus polymyxa detected by temperature gradient gel electrophoresis. Journal of Bacteriology, 178(19), 5636–5643.
Pandey, S., & Singh, D. K. (2004). Total bacterial and fungal population after chlorpyrifos and quinalphos treatments in groundnut (Arachis hypogaea L.) soil. Chemosphere, 55(2), 197–205.
Pereira, J., Picanço, M., Silva, A., Santos, E., Tomé, H., & Olarte, J. (2008). Effects of glyphosate and endosulfan on soil microorganisms in soybean crop. Planta Daninha, 26(4), 825–830.
Pimentel, D., Hepperly, P., Hanson, J., Douds, D., & Seidel, R. (2005). Environmental, energetic, and economic comparisons of organic and conventional farming systems. BioScience, 55(7), 573–582.
Rao, D. M. R., & Murty, A. S. (1980). Persistence of endosulfan in soils. Journal of Agricultural and Food Chemistry, 28(6), 1099–1101.
Ritz, K., & Young, I. M. (2004). Interactions between soil structure and fungi. Mycologist, 18(2), 52–59.
Ros, M., Goberna, M., Moreno, J., Hernandez, T., Garcia, C., Insam, H., et al. (2006). Molecular and physiological bacterial diversity of a semi-arid soil contaminated with different levels of formulated atrazine. Applied Soil Ecology, 34(2), 93–102.
Ulčnik, A., Cigić, I. K., & Pohleven, F. (2013). Degradation of lindane and endosulfan by fungi, fungal and bacterial laccases. World Journal of Microbiology and Biotechnology, 29(12), 2239–2247.
Van Bruggen, A. H. C., & Semenov, A. M. (2000). In search of biological indicator for soil health and disease suppression. Applied Soil Ecology, 15(1), 13–24.
Verma, K., Agrawal, N., Farooq, M., Misra, R. B., & Hans, R. K. (2006). Endosulfan degradation by a Rhodococcus strain isolated from earthworm gut. Ecotoxicology and Environmental Safety, 64(3), 377–381.
Vilgalys, R., & Hester, M. (1990). Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. Journal of Bacteriology, 172(8), 4238–4246.
Walker, A. (1978). Simulation of the persistence of eight soil-applied herbicides. Weed Research, 18(5), 305–313.
Xie, H. J., Shi, Y. J., Zhang, J., Cui, Y., Teng, S. X., Wang, S. G., et al. (2010). Degradation of phthalate esters (PAEs) in soil and the effects of PAEs on soil microcosm activity. Journal of Chemical Technology and Biotechnology, 85(8), 1108–1116.
Xie, H., Gao, F., Tan, W., & Wang, S.-G. (2011). A short-term study on the interaction of bacteria, fungi and endosulfan in soil microcosm. Science of the Total Environment, 412, 375–379.
Zhao, C., Xie, H., Zhang, J., Xu, J., & Liang, S. (2013). Spatial distribution of organochlorine pesticides (OCPs) and effect of soil characters: A case study of a pesticide producing factory. Chemosphere, 90(9), 2381–2387.
Zoetendal, E. G., von Wright, A., Vilpponen-Salmela, T., Ben-Amor, K., Akkermans, A. D., & de Vos, W. M. (2002). Mucosa-associated bacteria in the human gastrointestinal tract are uniformly distributed along the colon and differ from the community recovered from feces. Applied and Environmental Microbiology, 68(7), 3401–3407.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jian Zhang and Jing Qin contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhang, J., Qin, J., Zhao, C. et al. Response of Bacteria and Fungi in Soil Microcosm under the Presence of Pesticide Endosulfan. Water Air Soil Pollut 226, 109 (2015). https://doi.org/10.1007/s11270-015-2309-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-015-2309-6