Abstract
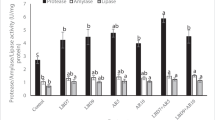
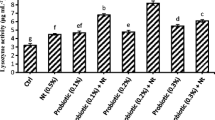
This study evaluated the effects of a multi-strain prebiotics and probiotics on the diet of tambaqui Colossoma macropomum. One hundred and fifty juvenile tambaqui (20.2 ± 3.6 g and 10.32 ± 5.78 cm) were randomly distributed in 15 experimental units with a volume of 80 L and fed for 45 days with a diet containing the symbiotic additive at five inclusion levels (0, 2, 4, 6 and 8 g kg feed− 1). At the end of the period, growth performance, survival, hemato-biochemical and intestinal parameters, as well as the fish’s resistance to stress were evaluated. The additive did not alter the growth performance, hemato-biochemical and intestinal parameters of the tambaqui. However, the hemato-biochemical parameters of aspartate aminotransferase (AST), plasma protein, hematocrit, glucose, triglycerides and cholesterol showed differences between treatments after the stress challenge. AST increased significantly during challenge and post-challenge. Plasma protein increased significantly during and after the challenge. The hematocrit was highest at 48 h after the challenge and lowest in the 2 g, 6 g and 8 g groups. Glucose was significantly reduced 24 h after the challenge, while triglycerides were lower 24 h and 48 h after the challenge. Cholesterol increased significantly in the challenge. There was an interaction between the factors sampling time and symbiotic concentration for hematocrit and glucose. In hypoxic stress situations, the 2 g concentration was more favorable for the fish. We recommend further studies with 2 g kg feed− 1 in trials lasting more than 45 days.



Similar content being viewed by others
Data Availability
Data will be made available upon reasonable request.
References
Affonso EG, Polez VLP, Corrêa CF, Mazon ADF, Araujo MRR, Moraes G, Rantin FT (2002) Blood parameters and metabolites in the teleost fish Colossoma macropomum exposed to sulfide or hypoxia. Comp Biochem Physiol Part - C: Toxicol Pharmacol 133(3):375–382. https://doi.org/10.1016/S1532-0456(02)00127-8
Afonso LO (2020) Identifying and managing maladaptive physiological responses to aquaculture stressors. Fish Physiol Acad Press 38:163–191. https://doi.org/10.1016/bs.fp.2020.10.002
Ahmed N, Turchini GM (2021) Recirculating aquaculture systems (RAS): environmental solution and climate change adaptation. J Clean Prod 297:126604. https://doi.org/10.1016/j.jclepro.2021.126604
Almeida-Val VMF, Val AL, Hochachka PW (1993) Hypoxia tolerance in Amazon fishes: status of an under-explored biological goldmine. In: Hochachka PW, Lutz PL, Sick TJ, Rosenthal M (eds) Surviving hypoxia: mechanisms of control and adaptation, 1st edn. CRC Press, pp 435–445
Amanajás RD, Val AL (2023) Thermal biology of tambaqui (Colossoma macropomum): General insights for aquaculture in a changing world. Rev Aquac 15(2):480–490. https://doi.org/10.1111/raq.12732
Amenyogbe E, Chen G, Wang Z, Huang J, Huang B, Li H (2020) The exploitation of probiotics, prebiotics and synbiotics in aquaculture: present study, limitations and future directions.: a review. Aquac Int 28:1017–1041. https://doi.org/10.1007/s10499-020-00509-0
Araújo JG, Guimarães IG, Mota CS, De Paula FG, Café MB, Pádua DMC (2017) Dietary available phosphorus requirement for tambaqui, Colossoma macropomum, juveniles based on growth, haematology and bone mineralization. Aquac Nutr 23(4):822–832. https://doi.org/10.1111/anu.12449
Araújo ERL, Barbas LAL, Ishikawa CM, Dias DDC, Sussel FR, Marques HLDA, Tachibana L (2018) Prebiotic, probiotic, and synbiotic in the diet of Nile tilapia post-larvae during the sex reversal phase. Aquac Int 26:85–97. https://doi.org/10.1007/s10499-017-0201-7
Ashley PJ (2007) Fish welfare: current issues in aquaculture. Appl Anim Behav Sci 104(3–4):199–235. https://doi.org/10.1016/j.applanim.2006.09.001
Baldisserotto B (2002) Fisiologia De Peixes Aplicada à Piscicultura. UFSM, Santa Maria
Cavero BAS, Rubim MAL, Pereira TM (2009) Criação comercial do tambaqui Colossoma macropomum (Cuvier, 1818). In: Tavares-Dias M (ed) Manejo E Sanidade De peixes em cultivo, 1st edn. Embrapa Amapá, pp 33–46
Chagas EC, Pilarski F, Sakabe R, Moraes FRD (2013) Productive performance and physiopathological responses of tambaqui fed with β-glucan enriched diet. Pesqui Agropecu Bras 48:899–905. https://doi.org/10.1590/S0100-204X2013000800013
Da Costa JAS, Sterzelecki FC, Natividade J, Souza RJF, Carvalho TCCD, Melo NFACD, Luz RK, Palheta GDA (2022) Residue from Açai Palm, Euterpe oleracea, as substrate for Cilantro, Coriandrum sativum, Seedling production in an Aquaponic System with Tambaqui, Colossoma macropomum. Agriculture 12(10):1555. https://doi.org/10.3390/agriculture12101555
Dawood MA (2021) Nutritional immunity of fish intestines: important insights for sustainable aquaculture. Rev Aquac 13(1):642–663. https://doi.org/10.1111/raq.12492
Dawood MA, Koshio S, Esteban MÁ (2018) Beneficial roles of feed additives as immunostimulants in aquaculture: a review. Rev Aquac 10(4):950–974. https://doi.org/10.1111/raq.12209
De Almeida LC, Lundstedt LM, Moraes G (2006) Digestive enzyme responses of tambaqui (Colossoma macropomum) fed on different levels of protein and lipid. Aquac Nutr 12(6):443–450. https://doi.org/10.1111/j.1365-2095.2006.00446.x
Del Valle JC, Bonadero MC, Gimenez AVF (2023) Saccharomyces cerevisiae as probiotic, prebiotic, synbiotic, postbiotics and parabiotics in aquaculture: an overview. Aquaculture 569:739342. https://doi.org/10.1016/j.aquaculture.2023.739342
Dias JA, Abe HA, Sousa NC, Couto MV, Cordeiro CA, Meneses JO, Cunha FS, Mouriño JLP, Martins ML, Barbas LAL, Carneiro PCF, Maria AN, Fujimoto RY (2018) Dietary supplementation with autochthonous Bacillus cereus improves growth performance and survival in Tambaqui Colossoma macropomum. Aquac Res 49(9):3063–3070. https://doi.org/10.1111/are.13767
Do Couto MVS, Sousa NDC, Abe HA, Dias JAR, Cordeiro CAM, Paixão PEG, Santos TBR, Cunha FS, Meneses JO, Nogueira Filho RM, Bomfim CNC, Honorato CA, Cardoso BT, Fujimoto RY (2022) Benefits of Virgin Coconut Oil in Diet to Colossoma macropomum (Cuvier, 1818). Aquac Nutr 2022:4387692. https://doi.org/10.1155/2022/4387692
Doğukan KAYA, Hersi MA, Ercüment GENÇ, Arslan HÖ (2022) Symbiotic effect of Bacillus clausii and Galacto-oligosaccharide on growth and survival rates in red cherry shrimp (Neocaridina Davidi). Mar Life Sci 4(2):146–151. https://doi.org/10.51756/marlife.1181522
Encarnação P (2016) Functional feed additives in aquaculture feeds. In: Nates SF (ed) Aquafeed formulation. Academic Press, Elsevier, pp 217–237
FAO (2022) The State of World Fisheries and Aquaculture 2022. Towards Blue Transformation. Rome, FAO. https://doi.org/10.4060/cc0461en
Fazio F (2019) Fish hematology analysis as an important tool of aquaculture: a review. Aquaculture 500:237–242. https://doi.org/10.1016/j.aquaculture.2018.10.030
Goldenfarb PB, Bowyer FP, Hall E, Brosious E (1971) Reproducibility in the hematology laboratory: the microhematocrit determination. Am J Clin Pathol 56(1):35–39. https://doi.org/10.1093/ajcp/56.1.35
Guimarães IG, Martins GP (2015) Nutritional requirement of two amazonian aquacultured fish species, Colossoma macropomum (Cuvier, 1816) and Piaractus brachypomus (Cuvier, 1818): a mini review. J Appl Ichthyol 31:57–66. https://doi.org/10.1111/jai.12976
Hilsdorf AWS, Hallerman E, Valladao GMR, Zaminhan-Hassemer M, Hashimoto DT, Dairiki JK, Takahashi LS, Albergaria FC, Gomes MES, Venturieri RLL, Moreira RG, Cyrino JEP (2022) The farming and husbandry of Colossoma macropomum: from amazonian waters to sustainable production. Rev Aquac 14(2):993–1027. https://doi.org/10.1111/raq.12638
Hoseinifar SH, Ringø E, Shenavar Masouleh A, Esteban MÁ (2016) Probiotic, prebiotic and synbiotic supplements in sturgeon aquaculture: a review. Rev Aquac 8(1):89–102. https://doi.org/10.1111/raq.12082
Hossain MK, Islam SM, Rafiquzzaman SM, Nuruzzaman M, Hossain MT, Shahjahan M (2022) Multi-species probiotics enhance growth of Nile tilapia (Oreochromis niloticus) through upgrading gut, liver and muscle health. Aquac Res 53(16):5710–5719. https://doi.org/10.1111/are.16052
Huang XJ, Choi YK, Im HS, Yarimaga O, Yoon E, Kim HS (2006) Aspartate aminotransferase (AST/GOT) and alanine aminotransferase (ALT/GPT) detection techniques. Sensors 6(7):756–782. https://doi.org/10.3390/s6070756
Hutjens MF (1991) Feed additives. Vet Clin North Am Food Anim Pract 7(2):525–540. https://doi.org/10.1016/S0749-0720(15)30789-1
IBGE – Brazilian Institute of Geography and Statistics (2023) SIDRA: survey of municipal livestock. 2023. https://sidra.ibge.gov.br/pesquisa/ppm/quadros/brasil/2019. Accessed 12 July 2023
Islam SM, Zahangir MM, Jannat R, Hasan MN, Suchana SA, Rohani MF, Shahjahan M (2020) Hypoxia reduced upper thermal limits causing cellular and nuclear abnormalities of erythrocytes in Nile tilapia, Oreochromis niloticus. J Therm Biol 90:102604. https://doi.org/10.1016/j.jtherbio.2020.102604
Islam SM, Rohani MF, Shahjahan M (2021) Probiotic yeast enhances growth performance of Nile tilapia (Oreochromis niloticus) through morphological modifications of intestine. Aquac Rep 21:100800. https://doi.org/10.1016/j.aqrep.2021.100800
Iwama GK, Afonso LO, Vijayan MM (1998) Stress in fish. Ann N Y Acad Sci 851:304–310. https://doi.org/10.1111/j.1749-6632.1998.tb09005.x
Jahan N, Islam SM, Rohani MF, Hossain MT, Shahjahan M (2021) Probiotic yeast enhances growth performance of rohu (Labeo rohita) through upgrading hematology, and intestinal microbiota and morphology. Aquaculture 545:737243. https://doi.org/10.1016/j.aquaculture.2021.737243
Jatobá A, Pereira MO, Vieira LM, Bitencourt M, Rodrigues E, Fachini FA, Moraes AV (2018) Action time and feed frequency of Lactobacillus plantarum for Nile tilapia. Arq Bras Med Vet Zootec 70:327–332. https://doi.org/10.1590/1678-4162-9870
Kotzent S, Gallani SU, Valladão GMR, Alves LDO, Pilarski F (2021) Probiotic potential of autochthonous bacteria from Tambaqui Colossoma macropomum. Aquac Res 52(5):2266–2275
Libanori MCM, Santos GG, Pereira SA, Lopes GR, Owatari MS, Soligo TA, Yamashita E, Pereira UP, Martins ML, Mouriño JLP (2021) Dietary supplementation with benzoic organic acid improves the growth performance and survival of Nile tilapia (Oreochromis niloticus) after challenge with Streptococcus agalactiae (Group B). Aquaculture 545:737204. https://doi.org/10.1016/j.aquaculture.2021.737204
Long Y, Yan J, Song G, Li X, Li X, Li Q, Cui Z (2015) Transcriptional events co-regulated by hypoxia and cold stresses in zebrafish larvae. BMC Genom 16:1–15. https://doi.org/10.1186/s12864-015-1560-y
Martos-Sitcha JA, Mancera JM, Prunet P, Magnoni LJ (2020) Welfare and stressors in fish: challenges facing aquaculture. Front Physiol 11:162. https://doi.org/10.3389/fphys.2020.00162
Merrifield DL, Dimitroglou A, Foey A, Davies SJ, Baker RT, Bøgwald J, Castex M, Ringø E (2010) The current status and future focus of probiotic and prebiotic applications for salmonids. Aquaculture 302(1–2):1–18. https://doi.org/10.1016/j.aquaculture.2010.02.007
Morais IS, O’sullivan FLA (2017) Biology, habitat and farming of tambaqui Colossoma macropomum (Cuvier, 1816). Sci Amazon 6:81–93. https://ainfo.cnptia.embrapa.br/digital/bitstream/item/153405/1/v6-n1-81-93-2017.pdf. Accessed 20 November 2023
Neves LDC, Favero GC, Beier SL, Ferreira NS, Palheta GDA, Melo NFACD, Luz RK (2020) Physiological and metabolic responses in juvenile Colossoma macropomum exposed to hypoxia. Fish Physiol Biochem 46(6):2157–2167. https://doi.org/10.1007/s10695-020-00868-8
Neves LDC, Silva WDS, Ferreira AL, Favero GC, Beier SL, Palheta GDA, Melo NFACD, Luz RK (2022) Physiological responses of juvenile Colossoma macropomum after different periods of air exposure. Aquaculture 548:737583. https://doi.org/10.1016/j.aquaculture.2021.737583
Ninawe AS, Selvin J (2009) Probiotics in shrimp aquaculture: avenues and challenges. Crit Rev Microbiol 35(1):43–66. https://doi.org/10.1080/10408410802667202
Owatari MS, Jesus GFA, Brum A, Pereira SA, Lehmann NB, Pereira UDP, Martins ML, Mouriño JLP (2018) Sylimarin as hepatic protector and immunomodulator in Nile tilapia during Streptococcus agalactiae Infection. Fish Shellfish Immunol 82:565–572. https://doi.org/10.1016/j.fsi.2018.08.061
Owatari MS, Silva LRD, Ferreira GB, Rodhermel JCB, Andrade JIAD, Dartora A, Jatobá A (2022) Body yield, growth performance, and haematological evaluation of Nile tilapia fed a diet supplemented with Saccharomyces cerevisiae. Anim Feed Sci Technol 293:115453. https://doi.org/10.1016/j.anifeedsci.2022.115453
Paixão AEM, Santos JC, Pinto MS, Pereira DSP, Ramos CECO, Cerqueira RB, Navarro RB, Silva RF (2017) Effect of commercial probiotics (Bacillus subtilis and Saccharomyces cerevisiae) on growth performance, body composition, hematology parameters, and Disease resistance against Streptococcus agalactiae in tambaqui (Colossoma macropomum). Aquac Int 25:2035–2045. https://doi.org/10.1007/s10499-017-0173-7
Pan CH, Chien YH, Wang YJ (2010) The antioxidant capacity response to hypoxia stress during transportation of characins (Hyphessobrycon callistus Boulenger) fed diets supplemented with carotenoids. Aquac Res 41(7):973–981. https://doi.org/10.1111/j.1365-2109.2009.02380.x
Peixe BR - Brazilian Association of Pisciculture (2023) Brazilian Pisciculture Yearbook. Edition 2023. São Paulo, Brazil, p. 65. https://www.peixebr.com.br/anuario/. Accessed 21 August 2023
Polakof S, Panserat S, Soengas JL, Moon TW (2012) Glucose metabolism in fish: a review. J Comp Physiol B: Biochem Syst Environ Physiol 182:1015–1045. https://doi.org/10.1007/s00360-012-0658-7
Porto LDA, Melo RMC, Beier SL, Luz RK, Favero GC (2021) Lophiosilurus alexandri, a sedentary bottom fish, adjusts its physiological parameters to survive in hypoxia condition. Fish Physiol Biochem 47:1793–1804. https://doi.org/10.1007/s10695-021-00996-9
Puvanasundram P, Chong CM, Sabri S, Yusoff MS, Karim M (2021) Multi-strain probiotics: functions, effectiveness and formulations for aquaculture applications. Aquac Rep 21:100905. https://doi.org/10.1016/j.aqrep.2021.100905
Ramesh D, Vinothkanna A, Rai AK, Vignesh VS (2015) Isolation of potential probiotic Bacillus spp. and assessment of their subcellular components to induce immune responses in Labeo rohita against Aeromonas hydrophila. Fish Shellfish Immunol 45(2):268–276. https://doi.org/10.1016/j.fsi.2015.04.018
Ribeiro SC, Castelo AS, Silva BMPD, Cunha ADS, Proietti Junior AA, Oba-Yoshioka ET (2016) Hematological responses of Tambaqui Colossoma macropomum (Serrassalmidae) fed with diets supplemented with essential oil from Mentha piperita (Lamiaceae) and challenged with Aeromonas hydrophila. Acta Amazon 46:99–106. https://doi.org/10.1590/1809-4392201501284
Richards JG (2009) Chap. 10 metabolic and molecular responses of fish to hypoxia. In: Richards JG, Farrell AP, Brauner CJ (eds) Fish Physiology 27. Elsevier, Amsterdam, pp 443–485. https://doi.org/10.1016/S1546-5098(08)00010-1JG
Rohani MF, Islam SM, Hossain MK, Ferdous Z, Siddik MA, Nuruzzaman M, Padeniya U, Brown C, Shahjahan M (2022) Probiotics, prebiotics and synbiotics improved the functionality of aquafeed: upgrading growth, reproduction, immunity and Disease resistance in fish. Fish Shellfish Immunol 120:569–589. https://doi.org/10.1016/j.fsi.2021.12.037
Rohani MF, Tarin T, Hasan J, Islam SM, Shahjahan M (2023) Vitamin E supplementation in diet ameliorates growth of Nile tilapia by upgrading muscle health. Saudi J Biol Sci 30(2):103558. https://doi.org/10.1016/j.sjbs.2023.103558
Rotta MA (2003) Aspectos gerais da fisiologia e estrutura do sistema digestivo dos peixes relacionados à piscicultura. Embrapa Technical note. https://www.infoteca.cnptia.embrapa.br/bitstream/doc/811108/1/DOC53.pdf. Accessed 27 July 2023
Roubach R, Gomes LC, Fonseca FAL, Val AL (2005) Eugenol as an efficacious anaesthetic for tambaqui, Colossoma macropomum (Cuvier). Aquac Res 36(11):1056–1061. https://doi.org/10.1111/j.1365-2109.2005.01319.x
Saint-Paul U (1984) Physiological adaptation to hypoxia of a neotropical characoid fish Colossoma macropomum, Serrasalmidae. Environ Biol Fishes 11:53–62. https://doi.org/10.1007/BF00001845
Sandre LCG, Buzollo H, Nascimento TMT, Neira LM, Jomori RK, Carneiro DJ (2017) Productive performance and digestibility in the initial growth phase of tambaqui (Colossoma macropomum) fed diets with different carbohydrate and lipid levels. Aquac Rep 6:28–34. https://doi.org/10.1016/j.aqrep.2017.02.003
Santos FA, Boaventura TP, Julio GSDC, Cortezzi PP, Figueiredo LG, Favero GC, Palheta GDA, Melo NFACD, Luz RK (2021) Growth performance and physiological parameters of Colossoma macropomum in a recirculating aquaculture system (RAS): importance of stocking density and classification. Aquaculture 534:736274. https://doi.org/10.1016/j.aquaculture.2020.736274
Siddik MA, Howieson J, Islam SM, Fotedar R (2022) Synbiotic feed supplementation improves antioxidant response and innate immunity of juvenile barramundi, lates calcarifer subjected to bacterial Infection. Aquaculture 552:737965. https://doi.org/10.1016/j.aquaculture.2022.737965
Val AL, Oliveira AMD (2021) Colossoma macropomum—A tropical fish model for biology and aquaculture. J Exp Zool A: Ecol Integr Physiol 335(9–10):761–770. https://doi.org/10.1002/jez.2536
Val AL, Silva MNP, Almeida-Val VMF (1998) Hypoxia adaptation in fish of the Amazon: a never-ending task. S Afr J Zool 33(2):107–114. https://doi.org/10.1080/02541858.1998.11448459
Wells RM, Pankhurst NW (1999) Evaluation of simple instruments for the measurement of blood glucose and lactate, and plasma protein as stress indicators in fish. J World Aquac Soc 30(2):276–284. https://doi.org/10.1111/j.1749-7345.1999.tb00876.x
Xie T, Gao Y, Qin H, Zhang J, Li M, Gao Y, Guan C, Jia Y (2023) Physiological response of spotted knifejaw (Oplegnathus punctatus) during transportation in offshore aquaculture net pen. Aquaculture 563:739029. https://doi.org/10.1016/j.aquaculture.2022.739029
Yilmaz S, Yilmaz E, Dawood MA, Ringø E, Ahmadifar E, Abdel-Latif HM (2022) Probiotics, prebiotics, and synbiotics used to control vibriosis in fish: a review. Aquaculture 547:737514. https://doi.org/10.1016/j.aquaculture.2021.737514
Zierler K (1999) Whole body glucose metabolism. Am J Physiol Endocrinol Metab 276(3):409–426. https://doi.org/10.1152/ajpendo.1999.276.3.E409
Acknowledgements
In memory of Professor Edilson Rodrigues Matos, we would like to thank for his valuable contribution to this research and on Amazon fish parasitology. The authors would like to thank the PROCAD Amazônia for financial support to Professor Nuno F. A. Correia de Melo, 40221741291, public notice n° 21/2018, project nº 88887.200588/2018-00.
Author information
Authors and Affiliations
Contributions
Rafael José Furtado Souza: Experimental execution, Manuscript writing. Edilson Rodrigues Matos: Experimental execution. Aldry Lorran da Silva Souza: Experimental execution. Paola Fabiana Fazzi-Gomes: Experimental execution, analysis. Nuno Filipe Alves Correia de Melo: Hematological analysis. Marco Shizuo Owatari: Data curation, Manuscript review and final writing. Glauber David Almeida Palheta: Methodology, Validation, Investigation. Rodrigo Takata: Methodology, Validation, Investigation, Data curation, Visualization. Fabio Carneiro Sterzelecki: Experimental execution, Data curation, Manuscript review, Supervision.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Animal Ethics
This research was approved by the Ethics Committee on Animal Use (CEUA) of the Federal Rural University of Amazon - UFRA, Belem, Brazil, protocol number 9354190820. The Juvenile tambaqui used in the study were purchased from a commercial fish farm located in Rondônia - Brazil and remained at the Sustainable Aquaculture Laboratory at UFRA for 10 days to acclimatize to the experimental conditions. All fish used in biological analyses were previously anesthetized with Eugenol (65 mg L– 1) (Roubach et al. 2005) and euthanized by spinal cord transection.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Souza, R.J.F., Matos, E.R., da Silva Souza, A.L. et al. Dietary effect of multi-strain prebiotics and probiotics on growth, hemato-biochemical parameters, intestinal histomorphometry, and resistance to hypoxia in juvenile tambaqui (Colossoma macropomum). Vet Res Commun 48, 1061–1072 (2024). https://doi.org/10.1007/s11259-023-10279-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10279-8