Abstract
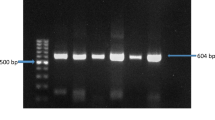
Infectious bursal disease (IBD) is considered as menace as it affects poultry industry globally causing immunosuppression, high mortality and heavy economic loss. Outbreaks of IBD were reported in many states of India including Kerala. VP1 gene acts as an important factor in the process of virus encapsidation and its involvement in viral virulence and viral replication indicates its importance in infectious bursal disease virus (IBDV). The present study was conducted to carry out the molecular characterization of VP1 gene of virulent IBDV in Kerala. A total of 42 samples were processed for the detection and analysis of VP1 gene of IBDV. Out of 42 samples, 21 samples were positive for VP1 gene of IBD. The phylogenetic analysis of the partial VP1 gene sequences reveals the clustering of IBDV isolates into very virulent IBDV (vvIBDV) and non-virulent IBDV (vIBDV). Eighteen isolates (11 isolates from vaccinated flock and 7 from non-vaccinated flocks) clustered with very virulent strains. Three isolates (2 isolates were from vaccinated flock and 1 from non-vaccinated flock) clustered with non-virulent IBDV strains, showing more evolutionarily similarity to south Indian strain VCN14/ABT/MVC/India. It is observed that vvIBDV isolates from this study have common ancestor with the south Indian strain PY12 but showed 9–10% divergence from this strains. The amino acid analysis of these 21 isolates revealed that 17 isolates possessed the characteristic vvIBDV TDN amino acid triplet, while the three isolates had non-vIBDV NEG amino acid triplet at 145/146/147 position. The remaining isolate 1/CVASP/IBDV/VP1 shows unique PDN triplet instead of TDN. Two vvIBDV isolates (15/CVASP/IBDV/VP1 and 18/CVASP/IBDV/VP1) showed 100% nucleotide and amino acid similarity with intermediate plus vaccine strain. Four vvIBDV isolates showed neutral amino acid substitution K251R which was earlier reported in Indian strains but first time in south Indian isolates. The most common unique amino acid substitution observed in our study was neutral E269D amino acid substitution in 12 isolates, neutral amino acid substitution T329S in five isolates, neutral T174N and non-polar to polar amino acid substitution A178T in isolate 10/CVASP/IBDV/VP1, non-polar to polar amino acid substitution P360R in isolate 17/CVASP/IBDV/VP1 and non-polar to polar amino acid substitution P188S in isolate 1/CVASP/IBDV/VP1. These novel mutations in our study reveal the role of genetic drift in the evolution of vvIBDV strains. The isolate 2/CVASP/IBDV/VP1 from non-vaccinated flock shows VP1 gene of non-vIBDV, but possessing VP2 of vvIBDV type indicates this is evolved by genetic shift of segments A and B. This is the first genetic characterization study of field VP1 gene of IBDV isolates in Kerala, India.

Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Akkara, T.S., Sankar, S., Chandran, R.A., Reshma, P.S., Reji, R.M., Nakka, A. and Mini, M., 2018. Molecular detection of infectious bursal disease virus from poultry in Thrissur district. Journal of Veterinary Animal Science, 49, 6–8.
Alfonso-Morales, A., Rios, L., Martínez-Pérez, O., Dolz, R., Valle, R., Perera, C.L., Bertran, K., Frías, M.T., Ganges, L., De Arce, H.D. and Majó, N., 2015. Evaluation of a phylogenetic marker based on genomic segment B of infectious bursal disease virus: facilitating a feasible incorporation of this segment to the molecular epidemiology studies for this viral agent. PloS one, 10, 0125853.
Bais, M.V., Kataria, R.S., Tiwari, A.K., Viswas, K.N., Reddy, A.V. and Prasad, N., 2004. Sequence analysis of an Indian field isolate of infectious bursal disease virus shows six unique amino acid changes in the VP1 gene. Vet. Res. Commun, 28, 641–646.
Berg, T.P.V.D., 2000. Acute infectious bursal disease in poultry: a review. Avian pathology, 29, 175–194.
Boot, H.J., ter Huurne, A.A.H., Hoekman, A.J., Peeters, B.P. and Gielkens, A.L., 2000. Rescue of very virulent and mosaic infectious bursal disease virus from cloned cDNA: VP2 is not the sole determinant of the very virulent phenotype. Journal of virology, 74, 6701–6711.
Cosgrove, A.S., 1962. An apparently new disease of chickens: avian nephrosis. Avian diseases, 6, 385–389.
Devigasri, C., Sankar, S., Rajalakshmi, N.S., Aswathy, N. and Mini, M., 2018. Concurrent infection of infectious bursal disease and pseudomoniasis in poultry-a case study. International Journal of Science, Environment and Technology, 7, 1777–1781
Eterradossi, N. and Saif, Y.M., 2020. Infectious bursal disease. Dis. Poult. 257–283.
Felice, V., Franzo, G., Catelli, E., Di Francesco, A., Bonci, M., Cecchinato, M., Mescolini, G., Giovanardi, D., Pesente, P. and Lupini, C., 2017. Genome sequence analysis of a distinctive Italian infectious bursal disease virus. Poultry science, 96, 43704377.
Gao, H.L., Wang, X.M., Gao, Y.L. and Fu, C.Y., 2007. Direct evidence of reassortment and mutant spectrum analysis of a very virulent infectious bursal disease virus. Avian diseases, 51, 93–899.
Gao, L., Li, K., Qi, X., Gao, H., Gao, Y., Qin, L., Wang, Y., Shen, N., Kong, X. and Wang, X., 2014. Triplet amino acids located at positions 145/146/147 of the RNA polymerase of very virulent infectious bursal disease virus contribute to viral virulence. J Gen Virol, 95, 888–897.
He, X., Xiong, Z., Yang, L., Guan, D., Yang, X. and Wei, P., 2014. Molecular epidemiology studies on partial sequences of both genome segments reveal that reassortant infectious bursal disease viruses were dominantly prevalent in southern China during 2000-2012. Archives of virology, 159, 3279–3292.
Hernández, M., Tomás, G., Marandino, A., Iraola, G., Maya, L., Mattion, N., Hernández, D., Villegas, P., Banda, A., Panzera, Y. and Pérez, R., 2015. Genetic characterization of South American infectious bursal disease virus reveals the existence of a distinct worldwide-spread genetic lineage. Avian pathology, 44, 212–221.
Hon, C.C., Lam, T.Y., Drummond, A., Rambaut, A., Lee, Y.F., Yip, C.W., Zeng, F., Lam, P.Y., Ng, P.T. and Leung, F.C., 2006. Phylogenetic analysis reveals a correlation between the expansion of very virulent infectious bursal disease virus and reassortment of its genome segment B. Journal of virology, 80, 8503–8509.
Hussain, A., Wu, T.T., Fan, L.J., Wang, Y.L., Muhammad, F.K., Jiang, N., Li, G.A.O., Kai, L.I., Gao, Y.L., Liu, C.J. and Cui, H.Y., 2020. The circulation of unique reassortment strains of infectious bursal disease virus in Pakistan. Journal of Integrative Agriculture, 19, 1867–1875.
Islam, M.R., Zierenberg, K. and Müller, H., 2001. The genome segment B encoding the RNA-dependent RNA polymerase protein VP1 of very virulent infectious bursal disease virus (IBDV) is phylogenetically distinct from that of all other IBDV strains. Archives of virology, 146, .2481–2492.
Islam, M.R., Rahman, S., Noor, M., Chowdhury, E.H. and Müller, H., 2012. Differentiation of infectious bursal disease virus (IBDV) genome segment B of very virulent and classical lineage by RT-PCR amplification and restriction enzyme analysis. Archives of virology, 157, 333–336.
Jackwood, D.J., Crossley, B.M., Stoute, S.T., Sommer-Wagner, S., Woolcock, P.R. and Charlton, B.R., 2012. Diversity of genome segment B from infectious bursal disease viruses in the United States. Avian diseases, 56, 165–172.
Kimura, M. 1980. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Molecular evolution, 16, 111–120.
Kumar, K., Singh, K.C.P. and Prasad, C.B., 2000. Immune responses to intermediate strain IBD vaccine at different levels of maternal antibody in broiler chickens. Trop Anim Hlth Prod, 32, 357
Kumar, S., Stecher, G., Li, M., Knyaz, C. and Tamura, K. 2018. MEGA X: molecular evolutionary genetics analysis across computing platforms. Molecular Boil Evolution 35, 1547–1549.
Mahgoub, H.A., 2012. An overview of infectious bursal disease. Archives of virology, 157, 2047–2057.
McFerran, J.B., McNulty, M.S., McKillop, E.R., Connor, T.J., McCracken, R.M., Collins, D.S. and Allan, G.M., 1980. Isolation and serological studies with infectious bursal disease viruses from fowl, turkeys and ducks: demonstration of a second serotype. Avian pathology, 9, 395–404.
Michel, L.O. and Jackwood, D.J., 2017. Classification of infectious bursal disease virus into genogroups. Archives of virology, 162, 3661–3670.
Mohanty, G.C., Pandey, A.P. and Rajya, B.S., 1971. Infectious bursal disease in chickens. Current Science, 40, 181–184.
Muller, H., Mundt, E., Eterradossi, N. and Islam, M.R. 2012. Current status of vaccines against infectious bursal disease. Avian Pathol, 41, 133–139.
Müller, H., Schnitzler, D., Bernstein, F., Becht, H., Cornelissen, D. and Lütticken, D.H., 1992. Infectious bursal disease of poultry: antigenic structure of the virus and control. Vet Microbial, 33, 175–183.
Nandhakumar, D., Rajasekhar, R., Logeshwaran, G., Ravishankar, C., Sebastian, S.R., Anoopraj, R., Sumod, K., Mani, B.K., Chaithra, G., Deorao, C.V. and John, K., 2019. Identification and genetic analysis of infectious bursal disease viruses from field outbreaks in Kerala, India. Tropical Animal Health and Production, 52, 989–997
Nwagbo, I.O., Shittu, I., Nwosuh, C.I., Ezeifeka, G.O., Odibo, F.J., Michel, L.O. and Jackwood, D.J., 2016. Molecular characterization of field infectious bursal disease virus isolates from Nigeria. Veterinary world, 9, 1420.
OIE Terrestrial Manual (2016) CHAPTER 2.3.12. Infectious bursal disease (Gumboro Disease). Available online:http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.03.12_IBD.pdf. Accessed 20 Nov 2019.
Pahar, B. and Rai, A., 1997. Immunogenicity of Infectious Bursal Disease Virus Strains Isolated in India. Journal of Applied Animal Research, 12, 137–144.
Pan, J., Vakharia, V.N. and Tao, Y.J., 2007. The structure of a birnavirus polymerase reveals a distinct active site topology. Proc Nati Acad Sci, 104, 7385–7390.
Patel, A.K., Pandey, V.C. and Pal, J.K., 2016. Evidence of genetic drift and reassortment in infectious bursal disease virus and emergence of outbreaks in poultry farms in India. Virus Diseases, 27, 161–169.
Rajkhowa, T.K., Vanlalruati, C. and Arya, R.S., 2018. Genetic characterization of infectious bursal disease viruses from field outbreaks of the North East Region of India. Avian diseases, 62, 218–225.
Rautenschlein, S., Kraemer, C., Vanmarcke, J. and Montiel, E. 2005. Protective efficacy of intermediate and intermediate plus infectious bursal disease virus (IBDV) vaccines against very virulent IBDV in commercial broilers. Avian Dis, 49, 231–237.
Valsala, K.V. and Gopalakrishnan Nair, M., Koshivarghese and Rajan A., 1988. Prevalence of infectious bursal disease in Kerala. Kerala Journal of Veterinary Science, 19, 60–64.
Wei, Y., Yu, X., Zheng, J., Chu, W., Xu, H., Yu, X. and Yu, L., 2008. Reassortant infectious bursal disease virus isolated in China. Virus Res, 131, 279–282.
Yang, H. and Ye, C., 2020. Reverse genetics approaches for live-attenuated vaccine development of infectious bursal disease virus. Current Opinion in Virology, 44, 139–144.
Yamaguchi, T., Ogawa, M., Inoshima, Y., Miyoshi, M., Fukushi, H. and Hirai, K. 1996. Identification of sequence changes responsible for the attenuation of highly virulent infectious bursal disease virus. Virol, 223, 219–223.
Acknowledgements
The authors acknowledge the Dean, College of Veterinary and Animal Sciences, Pookode, for providing all the required facilities to carry out the research work.
Author information
Authors and Affiliations
Contributions
1. C.V.D.: conducted the research work and wrote the manuscript.
2. R.R.: conceived and designed the research work.
3. C.R.: contributed in analysing and interpreting the molecular data.
4. N.D.: collected and processed the samples required in research work.
5. S.K.: contributed in analysing the data.
6. H.P.: contributed in collecting post-mortem samples for research work.
7. K.J.: approved the version to be published.
8. C.G.: contributed in drafting the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was performed per the guidelines for the care and use of animals by the Institutional Animal Ethics Committee, College of Veterinary and Animal Sciences, Pookode, and the Animal Welfare Board of India.
Consent to participate
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Deorao, C.V., Rajasekhar, R., Ravishankar, C. et al. Genetic variability in VP1 gene of infectious bursal disease virus from the field outbreaks of Kerala, India. Trop Anim Health Prod 53, 407 (2021). https://doi.org/10.1007/s11250-021-02852-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11250-021-02852-7