Abstract
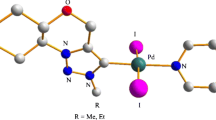
A series of calix[4]arene supported N-heterocyclic carbene palladium complexes were successfully prepared and characterised. Their X-ray crystal structures were obtained and are discussed. Notably, the dimeric compound b[5-(3-N-4,5-diphenylimidazol-2-yliden-1-yl)-25,26,27,28-tetrapropyloxy calix[4]arene] palladium(II) dibromide (cone) showed pseudo-polymorphism.







Similar content being viewed by others
References
Arduengo AJ, Harlow RL, Kline M (1991) A stable crystalline carbene. J Am Chem Soc 113(1):361–363. doi:10.1021/ja00001a054
Herrmann WA (2002) N-heterocyclic carbenes: a new concept in organometallic catalysis. Angew Chem Int Ed 41(8):1290–1309. doi:10.1002/1521-3773(20020415)41:8<1290:AID-ANIE1290>3.0.CO;2-Y
Crudden CM, Allen DP (2004) Stability and reactivity of N-heterocyclic carbene complexes. Coord Chem Rev 248(21–24):2247–2273. doi:10.1016/j.ccr.2004.05.013
Diez-Gonzalez S, Nolan SP (2007) N-heterocyclic carbene-copper(I) complexes in homogeneous catalysis. Synlett 14:2158–2167. doi:10.1055/s-2007-985577
Diez-Gonzalez S, Marion N, Nolan SP (2009) N-heterocyclic carbenes in late transition metal catalysis. Chem Rev (Washington, DC, USA) 109(8):3612–3676. doi:10.1021/cr900074m
Gade LH, Bellemin-Laponnaz S (2007) Mixed oxazoline-carbenes as stereodirecting ligands for asymmetric catalysis. Coord Chem Rev 251(5–6):718–725. doi:10.1016/j.ccr.2006.05.015
Egbert JD, Cazin CSJ, Nolan SP (2013) Copper N-heterocyclic carbene complexes in catalysis. Catal Sci Technol 3(4):912. doi:10.1039/c2cy20816d
Cazin CSJ (2013) N-heterocyclic carbenes. Dalton Trans 42(20):7254. doi:10.1039/c3dt90051g
Bezier D, Sortais J-B, Darcel C (2013) N-heterocyclic carbene ligands and iron: an effective association for catalysis. Adv Synth Catal 355(1):19–33. doi:10.1002/adsc.201200666
Przyojski JA, Arman HD, Tonzetich ZJ (2012) NHC complexes of cobalt(II) relevant to catalytic C–C coupling reactions. Organometallics 32(3):723–732. doi:10.1021/om3010756
Velazquez HD, Verpoort F (2012) N-heterocyclic carbene transition metal complexes for catalysis in aqueous media. Chem Soc Rev 41(21):7032–7060. doi:10.1039/c2cs35102a
Cheng L, Yang H, Zhang H, Zhao H, Lu Q (2012) N-heterocyclic carbene copper complexes: an efficient catalyst for organic synthesis. Huaxue Tongbao 75(7):606–613
Kantchev EAB, O’Brien CJ, Organ MG (2007) Palladium complexes of N-heterocyclic carbenes as catalysts for cross-coupling reactions—a synthetic chemist’s perspective. Angew Chem Int Ed 46(16):2768–2813. doi:10.1002/anie.200601663
Matthews SE, Beer PD (2005) Calixarene-based anion receptors. Supramol Chem 17(6):411–435. doi:10.1080/10610270500127089
Mandolini L, Ungaro R (2000) Calixarenes in Action. Imperial College Press, London. doi:10.1142/9781848160354_fmatter
Monnereau L, Sémeril D, Matt D (2012) Synthesis of calixarene-based bis(iminophosphoranes) and their use in suzuki-miyaura cross-coupling. Eur J Org Chem 14:2786–2791. doi:10.1002/ejoc.201200082
Brenner E, Matt D, Henrion M, Teci M, Toupet L (2011) Calix[4]arenes with one and two N-linked imidazolium units as precursors of N-heterocyclic carbene complexes. Coordination chemistry and use in Suzuki-Miyaura cross-coupling. Dalton Trans 40(38):9889–9898. doi:10.1039/C1DT10838G
Sémeril D, Jeunesse C, Matt D, Toupet L (2006) Regioselectivity with hemispherical chelators: increasing the catalytic efficiency of complexes of diphosphanes with large bite angles. Angew Chem Int Ed 45(35):5810–5814. doi:10.1002/anie.200601978
Semeril D, Lejeune M, Jeunesse C, Matt D (2005) Heck, Suzuki and Kumada-Corriu cross-coupling reactions mediated by complexes based on the upper rim of diphosphinated calix[4]arenes. J Mol Catal A: Chem 239(1–2):257–262. doi:10.1016/j.molcata.2005.06.024
Dospil G, Schatz J (2001) Synthesis and characterization of imidazole-substituted calix[4]arenes as simple enzyme-mimics with acyltransferase activity. Tetrahedron Lett 42(44):7837–7840. doi:10.1016/S0040-4039(01)01688-4
Frank M, Maas G, Schatz J (2004) Calix[4]arene-supported N-heterocyclic carbene ligands as catalysts for Suzuki cross-coupling reactions of chlorotoluene. Eur J Org Chem 3:607–613. doi:10.1002/ejoc.200300542
Brendgen T, Frank M, Schatz J (2006) The suzuki coupling of aryl chlorides in aqueous media catalyzed by in situ Generated calix[4]arene-based N-heterocyclic carbene ligands. Eur J Org Chem 10:2378–2383. doi:10.1002/ejoc.200500943
Dinares I, Garcia de Miguel C, Font-Bardia M, Solans X, Alcalde E (2007) Imidazolium-calix[4]arene molecular frameworks: bis(N-heterocyclic carbenes) as bidentate ligands. Organometallics 26(21):5125–5128. doi:10.1021/om7005722
Ren H, Xu Y, Jeanneau E, Bonnamour I, Tu T, Darbost U (2014) Synthesis, characterization and X-ray structures of N-heterocyclic carbene palladium complexes based on calix[4]arenes: highly efficient catalysts towards Suzuki-Miyaura cross-coupling reactions. Tetrahedron 70(17):2829–2837. doi:10.1016/j.tet.2014.02.051
Jaime C, De Mendoza J, Prados P, Nieto PM, Sanchez C (1991) Carbon-13 NMR chemical shifts. A single rule to determine the conformation of calix[4]arenes. J Org Chem 56(10):3372–3376. doi:10.1021/jo00010a036
Asiri AM, Heller HG, Hughes DS, Hursthouse MB, Kendrick J, Leusen FJ, Montis R (2014) A mechanophysical phase transition provides a dramatic example of colour polymorphism: the tribochromism of a substituted tri(methylene)tetrahydrofuran-2-one. Chem Cent J 8(1):1–11. doi:10.1186/s13065-014-0070-3
Karmakar A, Bania K, Baruah AM, Baruah JB (2007) Role of nitro-substituent in pseudo-polymorphism and in synthesis of metal carboxylato complexes of copper, zinc and manganese. Inorg Chem Commun 10(8):959–964. doi:10.1016/j.inoche.2007.04.026
Maruyama S, Ooshima H, Kato J (1999) Crystal structures and solvent-mediated transformation of Taltireline polymorphs. Chem Eng J 75(3):193–200. doi:10.1016/S1385-8947(99)00115-1
Nanubolu JB, Sridhar B, Babu VSP, Jagadeesh B, Ravikumar K (2012) Sixth polymorph of aripiprazole—an antipsychotic drug. CrystEngComm 14(14):4677–4685. doi:10.1039/C2CE25306B
Wittering KE, Agnew LR, Klapwijk AR, Robertson K, Cousen AJP, Cruickshank DL, Wilson CC (2015) Crystallisation and physicochemical property characterisation of conformationally-locked co-crystals of fenamic acid derivatives. CrystEngComm 17(19):3610–3618. doi:10.1039/C5CE00297D
Technologies A (2011) CrysAlisPro. Version 1.171.34.49 (release 20-01-2011 CrysAlis171.NET) (compiled 20 Jan 2011, 15:58:25)
Clark RC, Reid JS (1995) The analytical calculation of absorption in multifaceted crystals. Acta Crystallogr Sect A 51(6):887–897. doi:10.1107/S0108767395007367
Altomare A, Burla MC, Camalli M, Cascarano GL, Giacovazzo C, Guagliardi A, Moliterni AGG, Polidori G, Spagna R (1999) SIR97: a new tool for crystal structure determination and refinement. J Appl Crystallogr 32(1):115–119. doi:10.1107/S0021889898007717
Betteridge PW, Carruthers JR, Cooper RI, Prout K, Watkin DJ (2003) CRYSTALS version 12: software for guided crystal structure analysis. J Appl Crystallogr 36(6):1487. doi:10.1107/S0021889803021800
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ren, H., Ourri, B., Jeanneau, E. et al. Synthesis and crystal structures of palladium(II) complexes of bis-(N-heterocyclic carbenes) on a calix[4]arene platform. Transit Met Chem 41, 827–834 (2016). https://doi.org/10.1007/s11243-016-0084-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-016-0084-x