Abstract
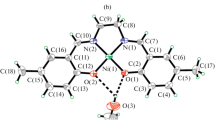
Three nickel(II) thiosemicarbazone complexes have been synthesized and characterized by spectroscopic and physicochemical techniques. The molecular structures of two of the complexes have been determined by single-crystal X-ray diffraction studies. In both of these complexes, the nickel center is coordinated by two tridentate ligands in a meridional fashion using C=S, C=N and neutral hydroxyl OH donors. The ligand is bound to nickel in its thione form in all three complexes. The synthesized compounds were tested for antimicrobial activity against Gram-positive and Gram-negative bacteria and fungi. Additionally, all the compounds were screened for antituberculosis activity. Several complex/organism combinations returned an MIC value of 0.8 μg/mL or better, which is almost 8 times more active than the standard (streptomycin, 6.25 μg/mL).





Similar content being viewed by others
References
Arguelles MCR, Silva ECL, Sanmartın J, Pelagatti P, Zani F (2005) J Inorg Biochem 99:2231–2239
Hu WX, Zhou W, Xia C, Wen X (2006) Bioorg Med Chem Lett 16:2213–2218
Afrasiabi Z, Sinn E, Padhye S, Dutta S, Newton C, Anson CE, Powell AK (2003) J Inorg Biochem 95:306–314
Bal T, Atasever B, Solakoglu Z, Kuruca SE, Ulkuseven B (2007) Eur J Med Chem 42:161–167
Basuli F, Peng SM, Bhattacharya S (1997) Inorg Chem 36:5645–5647
Pal I, Basuli F, Mak TCW, Bhattacharya S (2001) Angew Chem 40:2923–2925
Fostiak LM, Gracia I, Swearinger JK, Bermejo E, Castineivas A, West DX (2003) Polyhedron 22:83–92
Ze-Hua L, Chun-Ying D, Ji-Hui L, Young-Jiang L, Yu-Hua M, Xiao-Zeng Y (2000) New J Chem 24:1057–1062
Novakovi SB, Bogdanovic GA, Leovac VM (2005) Inorg Chem Commun 8:9–13
Iakovidou Z, Mioglou E, Mourelatos D, Kotsis A, Demertzis MA, Papagoergiou A, Miller JR, Kovala-Demertzi D (2001) Anticancer Drugs 12:65–70
Kovala-Demertzi D, Demertzis MA, Miller JR, Papadopoulou C, Dodorou C, Filousis G (2001) J Inorg Biochem 86:555–563
Andrews RK, Blakeley RL, Zerner B (1988) In: Sigel H, Sigel A (eds) Metal ions in biological systems. Marcel Dekker, New York
Halcrow MA, Christou G (1994) Chem Rev 94:2421–2481
Kasuga NC, Sekino K, Koumo C, Shimada N, Ishikawa M, Nomiya K (2001) J Inorg Biochem 84:55–65
Shawish HB, Paydar M, Looi CY, Wong YL, Movahed E, Halim SNA, Wong WF, Mustafa MR, Maah MJ (2014) Transit Met Chem 39:81–94
Bottari B, Maccari R, Monforte F, Ottana R, Vigorita MG, Bruno G, Nicolo F, Rotondo A, Rotondo E (2001) Bioorg Med Chem 9:2203–2211
Para A, Klisiewicz-Panszczyk T, Jurek I (2001) Acta Pol Pharm 58:405–408
Sen AK, Gupta SK (1962) J Indian Chem Soc 39:628–634
Cooper RI, Thompson AL, Watkin DJ (2010) J Appl Cryst 43:1100–1107
Betteridge PW, Carruthers JR, Cooper RI, Prout K, Watkin DJ (2003) J Appl Cryst 36:1487
Watkin DJ, Prout CK, Pearce LJ (1996) CAMERON. Chemical Crystallography Laboratory, Oxford
Burnett MN, Johnson CK (1996) ORTEP-III: Oak Ridge thermal ellipsoid plot program for crystal structure illustrations. Oak Ridge National Laboratory Report ORNL-6895
Lourenco MCS, de Souza MVN, Pinheiro AC, Ferreira ML, Goncalves RSB, Nogueira TCM, Peraltab MA (2007) Arkivoc 15:181–191
Schwalbe R, Steele-Moore L, Goodwin AC (2007) Antimicrobial susceptibility testing protocols. CRC Press, New York
Naik AD, Annigeri SM, Gangadharmath UB, Revankar VK, Mahale VB (2002) J Mol Struct 616:119–127
Prabhakaran R, Kalaivani P, Poornima P, Dallemer F, Paramaguru G, Vijaya Padma V, Renganathan R, Huang R, Natarajan K (2012) Dalton Trans 41:9323–9336
West DX, Nasar AA (1999) Transit Met Chem 24:617–621
Lever ABP (1984) Inorganic electronic spectroscopy, 2nd edn. Elsevier, Amsterdam
Campbell MJM (1975) Coord Chem Rev 15:279–319
Afrasiabi Z, Sinn E, Lin W, Ma Y, Campana C (2005) Padhye S J Inorg Biochem 99:1526–1531
Leovac VM, Markovi S, Divjakovi V, Szecsenyi KM, Joksovi MD, Lebanc I (2008) Acta Chim Slov 55:850–860
Acknowledgments
The authors thank USIC, Karnatak University, Dharwad, for providing spectral facilities. One of the authors (Sandeep P. Netalkar) is thankful to Department of Science and Technology for providing financial assistance under INSPIRE fellowship program.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Netalkar, P.P., Netalkar, S.P. & Revankar, V.K. Nickel(II) complexes of thiosemicarbazones: synthesis, characterization, X-ray crystallographic studies and in vitro antitubercular and antimicrobial studies. Transition Met Chem 39, 519–526 (2014). https://doi.org/10.1007/s11243-014-9827-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-014-9827-8