Abstract
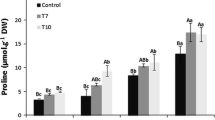
Type III polyketide synthases play significant roles in the biosynthesis of multiple plant secondary metabolites and plant adjustment to environmental stresses. AsPECPS, a type III polyketide synthase, is the key enzyme involved in the biosynthesis of 2-(2-phenylethyl) chromone derivatives which are the main components determining the quality of agarwood and special defensive substances of Aquilaria sinensis. However, the functions of AsPECPS in plant resistance have not been shown. Our findings indicated that AsPECPS was localized in the cytoplasm and cell wall. The expression of AsPECPS was significantly induced by salt stress and abscisic acid (ABA) treatment. Heterologous overexpression of AsPECPS in Nicotiana benthamiana enhanced the salt-stress tolerance at post-gemination stage showed by the transgenic plants with increased fresh weight, height, root length, and number of lateral roots. Furthermore, the overexpressing lines (OE) in soil retained more lateral roots, more proline content and lower malondialdehyde content under salinity stress. Additionally, AsPECPS-OE plants were less sensitive to ABA as indicated through the OE lines showing higher gemination rate, greater survival rate, and better growth characteristics. Further analyses demonstrated that the transgenic plants accumulated lower levels of ROS through down-regulating the expression of Rboh family genes and up-regulating the expression and activities of antioxidant enzymes under salt stress and ABA treatment. Overexpression of AsPECPS modulated the abundance of genes associated with ABA signaling pathway under ABA treatment. Taken together, our results indicated that overexpression of AsPECPS in N. benthamiana enhanced the plant tolerance to salt and ABA stresses.
Key Message
Overexpression of AsPECPS in N. benthamiana, a type III polyketide synthase involved in the biosynthesis of 2-(2-phenylethyl) chromone, enhanced the plant tolerance to salt and ABA stresses.
AbstractSection Graphical abstract







Similar content being viewed by others
Data availability
All data analyzed or generated in our study are included in this article and its supplementary information flies.
Abbreviations
- ABA :
-
Abscisic acid
- ABI3 :
-
ABSCISIC ACID INSENSITIVE3
- ABI5 :
-
ABSCISIC ACID INSENSITIVE5
- APX :
-
Peroxidase
- BCA :
-
Bicinchoninic acid
- CAT :
-
Catalase
- Chl :
-
Chlorophyl
- DAB :
-
3,3′-Diaminobenzidine
- GFP :
-
Green fluorescent protein
- MDA :
-
Malondialdehyde
- MeJA :
-
Methyl jasmonate
- MS :
-
Murashige and Skoog
- NBT :
-
Nitro blue tetrazolium
- NCED :
-
9-Cis-epoxycarotenoid dioxygenase
- OE :
-
Overexpression lines
- PCR :
-
Polymerase chain reaction
- PKS :
-
Polyketide synthase
- qRT-PCR :
-
Quantitative real-time polymerase chain reaction
- ROS :
-
Reactive oxygen species
- SA :
-
Salicylic acid
- SOD :
-
Superoxide dismutase
- UV :
-
Ultraviolet
- WT :
-
Wild type
References
Abe I (2020) Biosynthesis of medicinally important plant metabolites by unusual type III polyketide synthases. J Nat Med 74:639–646. https://doi.org/10.1007/s11418-020-01414-9
Abe I, Morita H (2010) Structure and function of the chalcone synthase superfamily of plant type III polyketide synthases. Nat Prod Rep 27:809–838. https://doi.org/10.1039/b909988n
Austin MB, Noel JP (2003) The chalcone synthase superfamily of type III polyketide synthases. Nat Prod Rep 20:79–110. https://doi.org/10.1039/b100917f
Bisht R, Bhattacharyya A, Shrivastava A, Saxena P (2021) An overview of the medicinally important plant Type III PKS derived polyketides. Front Plant Sci 12:746908. https://doi.org/10.3389/fpls.2021.746908
Bolat I, Korkmaz K, Dogan M, Turan M, Kaya C, Seyed Hajizadeh H, Kaya O (2024) Enhancing drought, heat shock, and combined stress tolerance in Myrobalan 29C rootstocks with foliar application of potassium nitrate. BMC Plant Biol 24:140. https://doi.org/10.1186/s12870-024-04811-4
Brasileiro ACM, Gimenes MA, Pereira BM, Mota APZ, Aguiar MN, Martins ACQ, Passos MAS, Guimaraes PM (2023) The stilbene synthase family in Arachis: a genome-wide study and functional characterization in response to stress. Genes (Basel) 14. https://doi.org/10.3390/genes14122181
Capelle V, Remoué C, Moreau L, Reyss A, Mahé A, Massonneau A, Falque M, Charcosset A, Thévenot C, Rogowsky P, Coursol S, Prioul JL (2010) QTLs and candidate genes for desiccation and abscisic acid content in maize kernels. BMC Plant Biol 10:2. https://doi.org/10.1186/1471-2229-10-2
Chen C, Twito S, Miller G (2014) New cross talk between ROS, ABA and auxin controlling seed maturation and germination unraveled in APX6 deficient Arabidopsis seeds. Plant Signal Behav 9:e976489. https://doi.org/10.4161/15592324.2014.976489
Chernys JT, Zeevaart JA (2000) Characterization of the 9-cis-epoxycarotenoid dioxygenase gene family and the regulation of abscisic acid biosynthesis in avocado. Plant Physiol 124:343–353. https://doi.org/10.1104/pp.124.1.343
Chizzali C, Swiddan AK, Abdelaziz S, Gaid M, Richter K, Fischer TC, Liu B, Beerhues L (2016) Expression of biphenyl synthase genes and formation of phytoalexin compounds in three fire blight-infected pyrus communis cultivars. PLoS One 11:e0158713. https://doi.org/10.1371/journal.pone.0158713
Cui XY, Du YT, Fu JD, Yu TF, Wang CT, Chen M, Chen J, Ma YZ, Xu ZS (2018) Wheat CBL-interacting protein kinase 23 positively regulates drought stress and ABA responses. BMC Plant Biol 18:93. https://doi.org/10.1186/s12870-018-1306-5
Du XM, Yin WX, Zhao YX, Zhang H (2001) The production and scavenging of reactive oxygen species in plants. Sheng Wu Gong Cheng Xue Bao 17:121–125
Fan M, Yang W, He M, Li Y, Peng Z, Wang G (2022) Occurrence, synthesis and biological activity of 2-(2-phenyethyl)chromones. Eur J Med Chem 237:114397. https://doi.org/10.1016/j.ejmech.2022.114397
Gao ZH, Wei JH, Yang Y, Zhang Z, Zhao WT (2012) Selection and validation of reference genes for studying stress-related agarwood formation of Aquilaria sinensis. Plant Cell Rep 31:1759–1768. https://doi.org/10.1007/s00299-012-1289-x
Geng X, Chen Y, Zhang S, Gao Z, Liu S, Yang Q, Wu J, Chen X (2022) Genome-wide analysis of type-iii polyketide synthases in wheat and possible roles in wheat sheath-blight resistance. Int J Mol Sci 23. https://doi.org/10.3390/ijms23137187
Giulietti A, Overbergh L, Valckx D, Decallonne B, Bouillon R, Mathieu C (2001) An overview of real-time quantitative PCR: applications to quantify cytokine gene expression. Methods 25:386–401. https://doi.org/10.1006/meth.2001.1261
Goodin MM, Dietzgen RG, Schichnes D, Ruzin S, Jackson AO (2002) pGD vectors: versatile tools for the expression of green and red fluorescent protein fusions in agroinfiltrated plant leaves. Plant J 31:375–383. https://doi.org/10.1046/j.1365-313x.2002.01360.x
Hou Q, Li S, Shang C, Wen Z, Cai X, Hong Y, Qiao G (2022) Genome-wide characterization of chalcone synthase genes in sweet cherry and functional characterization of CpCHS1 under drought stress. Front Plant Sci 13:989959. https://doi.org/10.3389/fpls.2022.989959
Huang M, Ma S, Qiao M, Fu Y, Li Y (2023) Quality similarity between induced agarwood by fungus and wild agarwood. J Agric Food Chem 71:15620–15631. https://doi.org/10.1021/acs.jafc.3c04322
Jan R, Aaqil Khan M, Asaf S, Lubna, Park JR, Lee IJ, Kim KM (2021) Flavonone 3-hydroxylase relieves bacterial leaf blight stress in rice via overaccumulation of antioxidant flavonoids and induction of defense genes and hormones. Int J Mol Sci 22. https://doi.org/10.3390/ijms22116152
Kalbina I, Li S, Kalbin G, Björn LO, Strid Å (2008) Two separate UV-B radiation wavelength regions control expression of different molecular markers in Arabidopsis thaliana. Funct Plant Biol 35:222–227. https://doi.org/10.1071/FP07197
Khawula S, Gokul A, Niekerk LA, Basson G, Keyster M, Badiwe M, Klein A, Nkomo M (2023) Insights into the effects of hydroxycinnamic acid and its secondary metabolites as antioxidants for oxidative stress and plant growth under environmental stresses. Curr Issues Mol Biol 46:81–95. https://doi.org/10.3390/cimb46010007
La VH, Tran DH, Han VC, Nguyen TD, Duong VC, Nguyen VH, Tran AT, Nguyen THG, Ngo XB (2023) Drought stress-responsive abscisic acid and salicylic acid crosstalk with the phenylpropanoid pathway in soybean seeds. Physiol Plant 175:e14050. https://doi.org/10.1111/ppl.14050
Lijuan C, Huiming G, Yi L, Hongmei C (2015) Chalcone synthase EaCHS1 from Eupatorium adenophorum functions in salt stress tolerance in tobacco. Plant Cell Rep 34:885–894. https://doi.org/10.1007/s00299-015-1751-7
Millar AA, Jacobsen JV, Ross JJ, Helliwell CA, Poole AT, Scofield G, Reid JB, Gubler F (2006) Seed dormancy and ABA metabolism in Arabidopsis and barley: the role of ABA 8’-hydroxylase. Plant J 45:942–954. https://doi.org/10.1111/j.1365-313X.2006.02659.x
Morita H, Lee YE, Shi SP (2023) Identification of a diarylpentanoid-producing polyketide synthase in the biosynthesis of 2-(2-phenylethyl)chromones in agarwood. J Nat Med 77:667–676. https://doi.org/10.1007/s11418-023-01743-5
Morita H, Wong CP, Abe I (2019) How structural subtleties lead to molecular diversity for the type III polyketide synthases. J Biol Chem 294:15121–15136. https://doi.org/10.1074/jbc.REV119.006129
Napsucialy-Mendivil S, Dubrovsky JG (2018) Genetic and phenotypic analysis of lateral root development in Arabidopsis thaliana. Methods Mol Biol 1761:47–75. https://doi.org/10.1007/978-1-4939-7747-5_4
Nong W, Law STS, Wong AYP, Baril T, Swale T, Chu LM, Hayward A, Lau DTW, Hui JHL (2020) Chromosomal-level reference genome of the incense tree Aquilaria sinensis. Mol Ecol Resour 20:971–979. https://doi.org/10.1111/1755-0998.13154
Pandith SA, Dhar N, Rana S, Bhat WW, Kushwaha M, Gupta AP, Shah MA, Vishwakarma R, Lattoo SK (2016) Functional promiscuity of two divergent paralogs of Type III plant polyketide synthases. Plant Physiol 171:2599–2619. https://doi.org/10.1104/pp.16.00003
Pathi KM, Tula S, Tuteja N (2013) High frequency regeneration via direct somatic embryogenesis and efficient Agrobacterium-mediated genetic transformation of tobacco. Plant Signal Behav 8:e24354. https://doi.org/10.4161/psb.24354
Pirasteh-Anosheh H, Samadi M, Kazemeini SA, Ozturk M, Ludwiczak A, Piernik A (2023) ROS homeostasis and antioxidants in the halophytic plants and seeds. Plants (Basel) 12:3023. https://doi.org/10.3390/plants12173023
Pothiraj R, Ravikumar MJ, Suthanthiram B, Subbaraya U, Krishnamurthy P (2021) Genome-scale analyses of polyketide synthases in banana: phylogenetics and expression profiling forecast their candidacy in specialized metabolism. Gene 778:145472. https://doi.org/10.1016/j.gene.2021.145472
Rong YQ, Li TZ, Liu X, Shi SP, Wang XH, Tu PF (2021) AsTal1 from Aquilaria sinensis regulates ABA signaling-mediated seed germination and root growth in Nicotiana benthamiana. Plant Cell Tiss Org 147:97–106. https://doi.org/10.1007/s11240-021-02110-6
Sano N, Marion-Poll A (2021) ABA metabolism and homeostasis in seed dormancy and germination. Int J Mol Sci 22:5069. https://doi.org/10.3390/ijms22105069
Sawano H, Matsuzaki T, Usui T, Tabara M, Fukudome A, Kanaya A, Tanoue D, Hiraguri A, Horiguchi G, Ohtani M, Demura T, Kozaki T, Ishii K, Moriyama H, Fukuhara T (2017) Double-stranded RNA-binding protein DRB3 negatively regulates anthocyanin biosynthesis by modulating PAP1 expression in Arabidopsis thaliana. J Plant Res 130:45–55. https://doi.org/10.1007/s10265-016-0886-0
Spormann S, Nadais P, Sousa F, Pinto M, Martins M, Sousa B, Fidalgo F, Soares C (2023) Accumulation of proline in plants under contaminated soils-are we on the same page? Antioxidants (Basel) 12. https://doi.org/10.3390/antiox12030666
Sundaravelpandian K, Chandrika N, Tsai YH, Schmidt W (2013) PFT1-controlled ROS balance is critical for multiple stages of root hair development in Arabidopsis. Plant Signal Behav 8:e24066. https://doi.org/10.4161/psb.24066
Vannozzi A, Dry IB, Fasoli M, Zenoni S, Lucchin M (2012) Genome-wide analysis of the grapevine stilbene synthase multigenic family: genomic organization and expression profiles upon biotic and abiotic stresses. BMC Plant Biol 12:130. https://doi.org/10.1186/1471-2229-12-130
Wang G, Liu X, Gan SS (2023) The ABA-AtNAP-SAG113 PP2C module regulates leaf senescence by dephoshorylating SAG114 SnRK3.25 in Arabidopsis. Mol Hortic 3:22. https://doi.org/10.1186/s43897-023-00072-1
Wang X, Zhang Z, Dong X, Feng Y, Liu X, Gao B, Wang J, Zhang L, Wang J, Shi S, Tu P (2017) Identification and functional characterization of three type III polyketide synthases from Aquilaria sinensis calli. Biochem Biophys Res Commun 486:1040–1047. https://doi.org/10.1016/j.bbrc.2017.03.159
Wang XH, Gao BW, Nakashima Y, Mori T, Zhang ZX, Kodama T, Lee YE, Zhang ZK, Wong CP, Liu QQ, Qi BW, Wang J, Li J, Liu X, Abe I, Morita H, Tu PF, Shi SP (2022) Identification of a diarylpentanoid-producing polyketide synthase revealing an unusual biosynthetic pathway of 2-(2-phenylethyl)chromones in agarwood. Nat Commun 13:348. https://doi.org/10.1038/s41467-022-27971-z
Wang Y, Yang L, Chen X, Ye T, Zhong B, Liu R, Wu Y, Chan Z (2016) Major latex protein-like protein 43 (MLP43) functions as a positive regulator during abscisic acid responses and confers drought tolerance in Arabidopsis thaliana. J Exp Bot 67:421–434. https://doi.org/10.1093/jxb/erv477
Wu X, Zhang S, Liu X, Shang J, Zhang A, Zhu Z, Zha D (2020) Chalcone synthase (CHS) family members analysis from eggplant (Solanum melongena L.) in the flavonoid biosynthetic pathway and expression patterns in response to heat stress. PLoS One 15:e0226537. https://doi.org/10.1371/journal.pone.0226537
Xie M, Wu D, Duan G, Wang L, He R, Li X, Tang D, Zhao X, Liu X (2014) AtWNK9 is regulated by ABA and dehydration and is involved in drought tolerance in Arabidopsis. Plant Physiol Biochem 77:73–83. https://doi.org/10.1016/j.plaphy.2014.01.022
Yu D, Xu F, Zeng J, Zhan J (2012) Type III polyketide synthases in natural product biosynthesis. IUBMB Life 64:285–295. https://doi.org/10.1002/iub.1005
Zandi P, Schnug E (2022) Reactive oxygen species, antioxidant responses and implications from a microbial modulation perspective. Biology (Basel) 11:155. https://doi.org/10.3390/biology11020155
Zhang L, Wang J, Li B, Gao BW, Wang XH, Shi SP, Tu PF (2013) Synthesis of unnatural small molecules by plant specific polyketide synthases. Chinese J Org Chem 12:2469–2484. https://doi.org/10.6023/cjoc201307012
Zhang Z, Wang X, Yang W, Wang J, Su C, Liu X, Li J, Zhao Y, Shi S, Tu P (2016) Five 2-(2-phenylethyl)chromones from sodium chloride-elicited Aquilaria sinensis cell suspension cultures. Molecules 21:555. https://doi.org/10.3390/molecules21050555
Zou H, Han L, Yuan M, Zhang M, Zhou L, Wang Y (2022) Sequence analysis and functional verification of the effects of three key structural genes, PdTHC2’GT, PdCHS and PdCHI, on the Isosalipurposide synthesis pathway in Paeonia delavayi var. lutea. Int J Mol Sci 23:5696. https://doi.org/10.3390/ijms23105696
Funding
This research was supported by National Natural Science Foundation of China (32170374) and Natural Science Foundation of Inner Mongolia (2023MS08011).
Author information
Authors and Affiliations
Contributions
Xinyu Mi: Conceptualization, Methodology, Writing-original draft. Yingying Feng: Conceptualization, Methodology, Writing-review. Fanyuan Guan: Data curation. Yuyan Zheng: Data analysis. Hailing Qiu: Investigation, Resources. Bowen Gao: Validation, Resources. Baowei Wang: Data analysis, Resources. Xiao Liu: Supervision. Juan Wang: Supervision. Pengfei Tu: Supervision. Shepo Shi: Writing-revision & editing, Supervision. Xiaohui Wang: revision & editing, Supervision Funding acquisition, Project administration.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Yi Li
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11240_2024_2739_MOESM1_ESM.pptx
Supplementary file1 (PPTX 59948 KB). Fig. S1 Total RNA from A. sinensis and N. benthamiana. a: Total RNA from A. sinensis. b: Total RNA from N. benthamiana. 1-15: The samples of A.sinensis. 15-31: The samples of N. benthamiana. Fig. S2 The melting curves and standard curved lines for quantification of the primers. a: The melting curves of primers. b: The standard curved lines for qualifications of the primers. Fig. S3 Genetic transformation of A. sinensis AsPECPS in N. benthamiana. a: PCR method to identify AsPECPS transgenic lines. b: The protein level of AsPECPS in transgenic lines were determined by Western blotting with an anti-PECPS antibody prepared in rabbits. Fig. S4 The phenotype and statistical analysis of seed germination of WT and OE lines under different NaCl treatment. a: The seed phenotype of WT and OE lines under NaCl treatment. b: The statistical analysis of seed germination of WT and OE line under different NaCl treatment at different time point. Error bars represent in the standard deviation of three biological replicates. The figures order of Supplementary file 1 should be corrected as follows: The Fig. S4 should be corrected as Fig. S2, while the Fig. S2 and S3 should be corrected as Fig. S3 and S4, respectively.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mi, X., Feng, Y., Guan, F. et al. Overexpression of plant polyketide synthase AsPECPS from Aquilaria sinensis enhances the tolerance of the transgenic Nicotiana benthamiana to salt stress and ABA treatment. Plant Cell Tiss Organ Cult 157, 8 (2024). https://doi.org/10.1007/s11240-024-02739-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11240-024-02739-z