Abstract
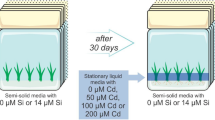
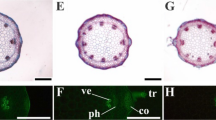
Despite having the ability to bioaccumulate trace elements such as cadmium (Cd), many species also present morphophysiological disorders that can hamper their use as phytoremediation plants. Since it can lead to alterations in biomass accumulation, the employment of elements that mitigate stress, such as silicon (Si), can diminish the deleterious effects caused by trace elements. The objective of this study was to analyze the anatomical and physiological modulations induced by the synergy between Cd and Si in Alternanthera tenella Colla plants, as well as to elucidate whether Si can mitigate the harmful effects caused by Cd under in vitro conditions. Nodal segments were cultured in MS medium containing a concentration gradient of Cd (0, 50, 100, or 200 μM) combined with two levels of Si (0 or 40 μM) for a total of eight treatments. After 34 days, the plants' anatomy, physiology, and tolerance index were analyzed. The plants presented anatomical adjustments such as lower stomatal index and number of vessel elements, suggesting lower translocation of Cd to the aerial part. When cultured with 200 μM Cd, the plants presented the lowest Chl a/b ratio (5.55). In the presence of Si, the decline of this ratio was smaller (6.66). Plants exposed to Cd concentrations of 50 μM without Si presented a significant decrease in the performance of the photosynthetic apparatus and tolerance index. The presence of Si in the medium reduced the damages caused by cadmium to the plants' physiology, resulting in greater growth and higher tolerance to this element.
Key Message
Alterations in the leaf anatomy played a fundamental role in plants' adjustment to stress Cd-induced. Si can mitigate the deleterious effects of Cd in A. tenella plants.







Similar content being viewed by others
Abbreviations
- ABS/RC:
-
Absorption flux per reaction center (RC)
- Chl a :
-
Chlorophyll a
- Chl b :
-
Chlorophyll b
- Chl total :
-
Total chlorophyll
- DI0/RC:
-
Dissipated energy flux per RC
- F0 :
-
Initial fluorescence
- Fm :
-
Maximal fluorescence intensity
- Ft :
-
Fluorescence at time t after start of actinic illumination
- FV/F0 :
-
Ratio of the de-excitation rate constants for photochemical and nonphotochemical events
- PI(ABS) :
-
Performance index based on absorption
- PI(Total) :
-
Total performance index, which measures the performance up until the final electron acceptors of PSI
- RC/ABS:
-
Total number of active reaction center
- VL :
-
Relative variable fluorescence at 0.15 ms (step L)
- VI :
-
Relative variable fluorescence at 30 ms (step I)
- VJ :
-
Relative variable fluorescence at 2 ms (step J)
- VK :
-
Relative variable fluorescence at 0.3 ms (step K)
- WK :
-
Represents the damage to oxygen-evolving complex
- WL :
-
Indicates disturbance in the thylakoid membranes, reducing the energetic connectivity between the PSII units
- φP0 :
-
Maximum quantum yield of primary photochemistry (at t = 0)
- φE0 :
-
Quantum yield of electron transport (at t = 0)
- φD0 :
-
Quantum yield of energy dissipation (at t = 0)
- δR0 :
-
Efficiency/probability with which an electron from the intersystem electron carriers moves to reduce end electron acceptors at the PSI acceptor side (RE)
References
Acosta-Motos JR, Diaz-Vivancos P, Alvarez S, Fernández-García N, Sanchez-Blanco MJ, Hernández JA (2015) NaCl-induced physiological and biochemical adaptive mechanisms in the ornamental Myrtus communis L. plants. J Plant Physiol 183:41–51. https://doi.org/10.1016/j.jplph.2015.05.005
Adrees M, Ali S, Rizwan M, Zia-Ur-Rehman M, Ibrahim M, Abbas F, Farid M, Qayyum MF, Irshad MK (2015) Mechanisms of silicon-mediated alleviation of heavy metal toxicity in plants: a review. Ecotoxicol Environ Saf 119:186–197. https://doi.org/10.1016/j.ecoenv.2015.05.011
Arnon DI (1949) Copper enzymes in isolated chloroplasts Polyphenoloxidase in Beta vulgaris. Plant Physiol 24(1):1–15. https://doi.org/10.1104/pp.24.1.1
Bari MA, Prity SA, Das U, Akther MS, Sajib SA, Reza MA, Kabir AH (2020) Silicon induces phytochelatin and ROS scavengers facilitating cadmium detoxification in rice. Plant Biol 22(3):472–479. https://doi.org/10.1111/plb.13090
Begovic L, Galic V, Abicic I, Loncaric Z, Lalic A, Mlinaric S (2020) Implications of intra-seasonal climate variations on chlorophyll a fluorescence and biomass in winter barley breeding program. Photosynthetica 58(4):995–1008. https://doi.org/10.32615/ps.2020.053
Bhat JA, Shivaraj S, Singh P, Navadagi DB, Tripathi DK, Dash PK, Solanke AU, Sonah H, Deshmukh R (2019) Role of silicon in mitigation of heavy metal stresses in crop plants. Plants 8(3):71. https://doi.org/10.3390/plants8030071
Brestic M, Zivcak M, Kalaji HM, Carpentier R, Allakhverdiev SI (2012) Photosystem II thermostability in situ: environmentally induced acclimation and genotype-specific reactions in Triticum aestivum L. Plant Physiol Biochem 57:93–105. https://doi.org/10.1016/j.plaphy.2012.05.012
Cai Y, Zhang S, Cai K, Huang F, Pan B, Wang W (2020) Cd accumulation, biomass and yield of rice are varied with silicon application at different growth phases under high concentration cadmium-contaminated soil. Chemosphere 242:125–128. https://doi.org/10.1016/j.chemosphere.2019.125128
Chung YS, Kim SH, Park CW, Na CI, Kim Y (2020) Treatment with silicon fertilizer induces changes in root morphological traits in soybean (Glycine max L.) during early growth. J Crop Sci Biotech 23:445–451. https://doi.org/10.1007/s12892-020-00052-7
Corso D, Delzon S, Lamarque LJ, Cochard H, Torres-Ruiz JM, King A, Brodribb T (2020) Neither xylem collapse, cavitation, or changing leaf conductance drive stomatal closure in wheat. Plant Cell Environ 43(4):854–865. https://doi.org/10.1111/pce.13722
Ding Y, Wang Y, Zheng X, Cheng W, Shi R, Feng R (2017) Effects of foliar dressing of selenite and silicate alone or combined with different soil ameliorants on the accumulation of As and Cd and antioxidant system in Brassica campestris. Ecotoxicol Environ Saf 142:207–215. https://doi.org/10.1016/j.ecoenv.2017.04.001
Dobrikova AG, Apostolova EL, Hanć A, Yotsova E, Borisova P, Sperdouli I, Adamakis IDS, Moustakas M (2021) Cadmium toxicity in Salvia sclarea L.: an integrative response of element uptake, oxidative stress markers, leaf structure and photosynthesis. Ecotoxicol Environ Saf 209:118–151. https://doi.org/10.1016/j.ecoenv.2020.111851
Emamverdian A, Ding Y, Xie Y, Sangari S (2018) Silicon mechanisms to ameliorate heavy metal stress in plants. Biomed Res Int 20:258–279. https://doi.org/10.1155/2018/8492898
Etesami H, Jeong BR (2018) Silicon (Si): Review and future prospects on the action mechanisms in alleviating biotic and abiotic stresses in plants. Ecotoxicol Environ Saf 147:881–896. https://doi.org/10.1016/j.ecoenv.2017.09.063
Ferreira E, Cavalcanti P, Nogueira D (2018) ExpDes. pt: Experimental Designs package. R package version 1.2. 0. Retrieved Sept 11:20–48
Franić M, Galić V, Mazur M, Šimić D (2018) Effects of excess cadmium in soil on JIP-test parameters, hydrogen peroxide content and antioxidant activity in two maize inbreds and their hybrid. Photosynthetica 56:660–669. https://doi.org/10.1007/s11099-017-0710-7
Grajek H, Rydzyński D, Piotrowicz-Cieślak A, Herman A, Maciejczyk M, Wieczorek Z (2020) Cadmium ion-chlorophyll interaction–Examination of spectral properties and structure of the cadmium-chlorophyll complex and their relevance to photosynthesis inhibition. Chemosphere 261:127434. https://doi.org/10.1016/j.chemosphere.2020.127434
Gu HH, Qiu H, Tian T, Zhan SS, Chaney RL, Wang SZ, Tang YT, Morel JL, Qiu RL (2011) Mitigation effects of silicon rich amendments on heavy metal accumulation in rice (Oryza sativa L.) planted on multi-metal contaminated acidic soil. Chemosphere 83:1234–1240. https://doi.org/10.1016/j.chemosphere.2011.03.014
Guo Y, Zhang Y, Lu Y, Shi J, Chen S, Strasser R, Qiang S, Hu Z (2020) Effect of AtLFNR1 deficiency on chlorophyll a fluorescence rise kinetics OJIP of Arabidopsis. Photosynthetica 58(2):391–398. https://doi.org/10.32615/ps.2019.167
Haider FU, Liqun C, Coulter JA, Cheema SA, Wu J, Zhang R, Wenjun M, Farooq M (2021) Cadmium toxicity in plants: Impacts and remediation strategies. Ecotoxicol Environ Saf 211:111–127. https://doi.org/10.1016/j.ecoenv.2020.111887
Houri T, Khairallah Y, Al Zahab A, Osta B, Romanos D, Haddad G (2020) Heavy metals accumulation effects on the photosynthetic performance of geophytes in Mediterranean reserve. J King Saud Univ Sci 32(1):874–880. https://doi.org/10.1016/j.jksus.2019.04.005
Hu B, Shao S, Ni H, Fu Z, Hu L, Zhou Y, Min X, She S, Chen S, Huang M, Zhou L, Li Y, Shi Z (2020) Current status, spatial features, health risks, and potential driving factors of soil heavy metal pollution in China at province level. Environ Pollut 266:114961. https://doi.org/10.1016/j.envpol.2020.114961
Huang H, Li M, Rizwan M, Dai Z, Yuan Y, Hossain MM, Cao M, Xiong S, Tu S (2021) Synergistic effect of silicon and selenium on the alleviation of cadmium toxicity in rice plants. J Hazard Mater 401:123–149. https://doi.org/10.1016/j.jhazmat.2020.123393
Janečková H, Husičková A, Lazár D, Ferretti U, Pospíšil P, Špundová M (2019) Exogenous application of cytokinin during dark senescence eliminates the acceleration of photosystem II impairment caused by chlorophyll b deficiency in barley. Plant Physiol Biochem 136:43–51. https://doi.org/10.1016/j.plaphy.2019.01.005
Johansen DA (1940) Plant microtechnique. McGraw-Hill Book Company Inc, London, pp 530–550
Kalaji HM, Jajoo A, Oukarroum A, Brestic M, Zivcak M, Samborska IA, Cetner MD, Łukasik I, Goltsev V, Ladle RJ (2016) Chlorophyll a fluorescence as a tool to monitor physiological status of plants under abiotic stress conditions. Acta Physiol Plant 4:1–11. https://doi.org/10.1007/s11738-016-2113-y
Klotzbücher A, Klotzbücher T, Jahn R, Van Chien H, Hinrichs M, Sann C, Vetterlein D (2018) Effects of Si fertilization on Si in soil solution, Si uptake by rice, and resistance of rice to biotic stresses in Southern Vietnam. Paddy Water Environ 16(2):243–252. https://doi.org/10.1007/s10333-017-0610-2
Liu L, Li W, Song W, Guo M (2018) Remediation techniques for heavy metal-contaminated soils: Principles and applicability. Sci Total Environ 633:206–219. https://doi.org/10.1016/j.scitotenv.2018.03.161
Lux A, Sottnikova A, Opatrna J, Greger M (2004) Differences in structure of adventitious roots in Salix clones with contrasting characteristics of cadmium accumulation and sensitivity. Physiol Plant 120(4):537–545. https://doi.org/10.1111/j.0031-9317.2004.0275.x
Malhotra C, Kapoor RT (2019) Silicon: a sustainable tool in abiotic stress tolerance in plants. In: Hasanuzzaman M, Hakeem KR, Nahar K, Alharby HF (eds) Plant Abiotic Stress Tolerance. Springer, Cham, Switzerland, pp 333–356. https://doi.org/10.1007/978-3-030-06118-0_14
Martins JPR, Rodrigues LCA, Silva TS, Santos ER, Falqueto AR, Gontijo ABPL (2019) Sources and concentrations of silicon modulate the physiological and anatomical responses of Aechmea blanchetiana (Bromeliaceae) during in vitro culture. Plant Cell Tissue Organ Cult 137:397–410. https://doi.org/10.1007/s11240-019-01579-6
Martins JPR, Souza AFC, Rodrigues LCA, Braga PCS, Gontijo ABPL, Falqueto AR (2020a) Zinc and selenium as modulating factors of the anatomy and physiology of Billbergia zebrina (Bromeliaceae) during in vitro culture. Photosynthetica 58:1068–1077. https://doi.org/10.32615/ps.2020.058
Martins JPR, Vasconcelos LL, Braga PCS, Rossini FP, Conde LT, Rodrigues LCA, Falqueto AR, Gontijo ABPL (2020b) Morphophysiological responses, bioaccumulation and tolerance of Alternanthera tenella Colla (Amaranthaceae) to excess copper under in vitro conditions. Plant Cell Tissue Organ Cult 143(2):303–318. https://doi.org/10.1007/s11240-020-01917-z
Martins JPR, Conde LT, Falqueto AR, Gontijo ABPL (2021) Selenium biofortified Aechmea blanchetiana (Bromeliaceae) can resist lead-induced toxicity during in vitro culture. Acta Physiol Plant 43:149. https://doi.org/10.1007/s11738-021-03323-0
Menegale MC, Castro GSA, Mancuso M (2015) Silı́cio: interação com o sistema solo-planta. J Agron Sci 4:435–454
Mott KA, Peak D (2018) Effects of the mesophyll on stomatal responses in amphistomatous leaves. Plant Cell Environ 41(12):2835–2843. https://doi.org/10.1111/pce.13411
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Paradiso R, Arena C, De Micco V, Giordano M, Aronne G, De Pascale S (2017) Changes in leaf anatomical traits enhanced photosynthetic activity of soybean grown in hydroponics with plant growth-promoting microorganisms. Front Plant Sci 8:674. https://doi.org/10.3389/fpls.2017.00674
Pereira AS, Cortez PA, Almeida AAF, Prasad MNV, França MGC, Cunha M, Jesus RM, Mangabeira PAO (2017) Morphology, ultrastructure, and element uptake in Calophyllum brasiliense Cambess (Calophyllaceae J. Agardh) seedlings under cadmium exposure. Environ Sci Pollut Res 24(18):176–188.https://doi.org/10.1007/s12298-017-0494-z
Pereira TS, Pereira TS, Souza CLFC, Lima EJA, Batista BL, Lobato AKS (2018) Silicon deposition in roots minimizes the cadmium accumulation and oxidative stress in leaves of cowpea plants. Physiol Mol Biol Plant 24(1):99–114. https://doi.org/10.1007/s12298-017-0494-z
Pires-Lira MF, Castro EM, Lira JMS, Oliveira C, Pereira FJ, Pereira MP (2020) Potential of Panicum aquanticum Poir. (Poaceae) for the phytoremediation of aquatic environments contaminated by lead. Ecotoxicol Environ Saf 193:110–136. https://doi.org/10.1016/j.ecoenv.2020.110336
Ranjbarfordoei A, Samson R, Van Damme P (2006) Chlorophyll fluorescence performance of sweet almond [Prunus dulcis (Miller) D. Webb] in response to salinity stress induced by NaCl. Photosynthetica 44(4):513–522. https://doi.org/10.1007/s11099-006-0064-z
Rodrigues LCA, Martins JPR, Júnior OA, Guilherme LRG, Pasqual M, Castro EM (2017) Tolerance and potential for bioaccumulation of Alternanthera tenella Colla to cadmium under in vitro conditions. Plant Cell Tissue Organ Cult 130:507–519. https://doi.org/10.1007/s11240-017-1241-4
Rouphael Y, De Micco V, Arena C (2017) Effect of Ecklonia maxima seaweed extract on yield, mineral composition, gas exchange, and leaf anatomy of zucchini squash grown under saline conditions. J Appl Phycol 29:459–470. https://doi.org/10.1007/s10811-016-0937-x
Roychowdhury A, Datta R, Sarkar D (2018) Heavy metal pollution and remediation. In: Török B, Dransfield T (eds) Green Chemistry. University of Massachusetts Boston, Boston, MA, USA, pp 359–373. https://doi.org/10.1016/B978-0-12-809270-5.00015-7
Santos ER, Martins JPR, Rodrigues LCA, Gontijo ABPL, Falqueto AR (2020) Morphophysiological responses of Billbergia zebrina Lindl. (Bromeliaceae) in function of types and concentrations of carbohydrates during conventional in vitro culture. Ornam Hort 26:18–34. https://doi.org/10.1590/2447-536X.v26i1.2092
Scholz AK, Klepsch M, Karimi Z, Jansen S (2013) How to quantify conduits in wood? Front Plant Sci 4:56. https://doi.org/10.3389/fpls.2013.00056
Silva-Cunha LF, Oliveira VP, Nascimento AWS, Silva BRS, Batista BL, Alsahli AA, Lobato AKS (2021) Leaf application of 24-epibrassinolide mitigates cadmium toxicity in young Eucalyptus urophylla plants by modulating leaf anatomy and gas exchange. Physiol Plant 173:67–87. https://doi.org/10.1111/ppl.13182
Stirbet A, Govindjee SA (2011) On the relation between the Kautsky effect (chlorophyll a fluorescence induction) and Photosystem II: basics and applications of the OJIP fluorescence transient. J Photoch Photobio 104:236–257. https://doi.org/10.1016/j.jphotobiol.2010.12.010
Strasser RJ, Tsimilli-Michael M, Srivastava A (2004) Analysis of the Chlorophyll a Fluorescence Transient. In: Papageorgiou GC, Govindjee (eds) Chlorophyll a Fluorescence. Advances in Photosynthesis and Respiration. Springer, Dordrecht 19:321–362. https://doi.org/10.1007/978-1-4020-3218-9_12
Suman J, Uhlik O, Viktorova J, Macek T (2018) Phytoextraction of heavy metals: a promising tool for clean-up of polluted environment? Front Plant Sci 9:1476. https://doi.org/10.3389/fpls.2018.01476
Ur-Rahman S, Xuebin Q, Zhao Z, Du Z, Imtiaz M, Mehmood F, Hongfei L, Hussain B, Ashraf MN (2021) Alleviatory effects of Silicon on the morphology, physiology, and antioxidative mechanisms of wheat (Triticum aestivum L.) roots under cadmium stress in acidic nutrient solutions. Sci Rep 11:1958. https://doi.org/10.1038/s41598-020-80808-x
Vaculík M, Lukačová Z, Bokor B, Martinka M, Tripathi DK, Lux A (2020) Alleviation mechanisms of metal (loid) stress in plants by silicon: a review. J Exp Bot 71(21):6744–6757. https://doi.org/10.1093/jxb/eraa288
Wellburn R (1994) The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J Plant Physiol 144(3):307–313. https://doi.org/10.1016/S0176-1617(11)81192-2
Wilkins DA (1957) A technique for the measurement of lead tolerance in plants. Nature 180:37–38. https://doi.org/10.1038/180037b0
Xiang M, Chen S, Wanga L, Donga Z, Huanga J, Zhanga Y, Strasser RJ (2013) Effect of vulculic acid produced by Nimbya alternantherae on the photosynthetic apparatus of Alternanthera hiloxeroides. Plant Physiol Biochem 65:81–88. https://doi.org/10.1016/j.plaphy.2013.01.013
Yanhui C, Hongrui W, Beining Z, Shixing G, Zihan W, Yue W, Huihui Z, Guangyu S (2020) Elevated air temperature damage to photosynthetic apparatus alleviated by enhanced cyclic electron flow around photosystem I in tobacco leaves. Ecotoxicol Environ Saf 204:111136. https://doi.org/10.1016/j.ecoenv.2020.111136
Zhang HH, Xu N, Wu X, Wang J, Ma S, Li X, Sun G (2018) Effects of four types of sodium salt stress on plant growth and photosynthetic apparatus in sorghum leaves. J Plant Interact 13(1):506–513. https://doi.org/10.1080/17429145.2018.1526978
Zhang L, Mcevoy D, Le Y, Ambrose C (2021) Live imaging of microtubule organization, cell expansion, and intercellular space formation in Arabidopsis leaf spongy mesophyll cells. Plant Cell 33:623–641. https://doi.org/10.1093/plcell/koaa036
Acknowledgements
The authors acknowledge the scholarship granted by CAPES (Coordination for the Improvement of Higher Education Personnel), and FAPES (Espírito Santo Research Foundation). The authors also acknowledge Luiz Carlos de Almeida Rodrigues for technical assistance in making the figures.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
FPR, SWM, EC, PCSB, and LTC performed experiments. FPR and JPRM wrote the manuscript and performed the statistical analysis. ARF and ABPLG provided the structure and contributed to the design and interpretation of the results. All the authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interest.
Additional information
Communicated by Mohammad Reza Abdollahi.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rossini, F.P., Martins, J.P.R., Moreira, S.W. et al. In vitro morphophysiological responses of Alternanthera tenella colla (Amaranthaceae) to stress induced by cadmium and the attenuating action of silicon. Plant Cell Tiss Organ Cult 150, 223–236 (2022). https://doi.org/10.1007/s11240-022-02263-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-022-02263-y