Abstract
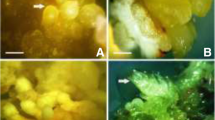
Byrsonima, especially the species Byrsonima intermedia, is an endangered Brazilian plant that has been widely used as food and for its therapeutic characteristics. However, this species faces challenges with sexual propagation, and somatic embryogenesis has emerged as a viable alternative option for propagation. Therefore, this study aimed to establish a protocol for inducing somatic embryogenesis in B. intermedia. For the induction of callus from in vitro seedling leaves, different subcultures (three subcultures, 60 days each) and concentrations of different cytokinins (BAP, TDZ, Kin and ZEA) combined with varying NAA solutions were tested. Different concentrations of NAA were also analyzed in the induction of pro-embryogenic calli. For the induction of embryogenic calli and somatic embryos, the pro-embryogenic calli were subcultured on MS medium without adding growth regulators. The somatic embryos that originated were inoculated on a maturation medium containing different concentrations of gibberellic acid (GA3). The formation of secondary embryos was also analyzed using different concentrations (0, 2.88, and 8.66 µM) of GA3 and different types of lids (Conventional lid, Biossama® commercial lid and conventional lid with membranes). The results show that for the induction of somatic embryos, the use of kinetin with NAA presented the formation of somatic embryos in the second (4.76 µM CIN + 0.54 µM NAA) and third (5.17 µM CIN + 10.54 µM NAA) subcultures. The use of 28.87 µM GA3 favored the formation of seedlings. The Biossama lid and 2.88 µM GA3 showed higher formation of secondary embryos.






Similar content being viewed by others
Abbreviations
- BAP:
-
Benzylaminopurine
- TDZ:
-
Thidiazuron
- Kin:
-
Kinetin
- ZEA:
-
Zeatin
- NAA:
-
Naphthalene acetic acid
- GA3 :
-
Gibberellic acid
- WPM:
-
Wood plant medium
- MS medium:
-
Murashige and Skoog medium
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
References
Al-Khayri JM, Naik PM (2017) Date palm micropropagation: advances and applications. Ciênc Agrotecnol 41(4):347–358. https://doi.org/10.1590/1413-70542017414000217
Almeida EP de, Oliveira RP, de Dantas JLL (2000) Protocolo para a embriogênese somática do mamoeiro. Pesq Agropec Bras 35(10):2017–2024. https://doi.org/10.1590/S0100-204X2000001000013
Asthana P, Rai MK, Jaiswal U (2017) Somatic embryogenesis from sepal explants in Sapindus trifoliatus, a plant valuable in herbal soap industry. Ind Crops Prod 100:228–235. https://doi.org/10.1016/j.indcrop.2017.02.034
Barberini S, Danti R, Lambardi M (2016) Somatic plant regeneration from selected common cypress (Cupressus sempervirens L.) clones resistant to the bark canker disease. Plant Cell Tissue Organ Cult 124:393–403. https://doi.org/10.1007/S11240-015-0902-4
Bezerra JS, Willadino L, Camara TR (2001) Crescimento de calos embriogênicos de milho submetidos ao estresse salino. Sci Agric 58(2):259–263. https://doi.org/10.1590/S0103-90162001000200007
Chée RP, Cantliffe DJ (1988) Somatic embryonic patterns and plant regeneration in Ipomoea batatas Poir. In Vitro Cell Dev Biol Plant 24(6):955–958. https://doi.org/10.1007/BF02623910
Ferreira DF (2014) Sisvar: a guide for its bootstrap procedures in multiple comparisons. Ciênc Agrotecnol 38(2):109–112. https://doi.org/10.1590/S1413-70542014000200001
Flores R, Nicoloso FT, Vasconcellos NJ, Maldaner J, Garlet TMB (2007) Embriogênese somática e organogênese indireta em Pfaffia tuberosa (Spreng.) Hicken. Rev Bras Biociênc 5(2):993–995
George EF (1996) Plant propagation by tissue culture: part 1, the technology. Exegetics, Edington
Gomes GAC, Paiva R, Paiva PDO, Santiago EJA (2003) Plant regeneration from callus culture of Maclura tinctoria, an endangered woody species. In Vitro Cell Dev Biol Plant 39(3):293–295. https://doi.org/10.1590/S1413-70542003000200010
Gonçalves LA, Geraldine RM, Picoli EAT, Vendrame WA, Carvalho CR, Otoni WC (2008) In vitro propagation of Herreria salsaparrilha Martius (Herreriaceae) as affected by different sealing materials and gaseous exchanges. Plant Cell Tissue Organ Cult 92(3):243–250. https://doi.org/10.1007/s11240-007-9327-z
Gray DJ, Benton CM (1990) Micropropagation and plant establishment of muscadine grape. Proc Fla State Hortic Soc 103:300–302
Hakman I, Arnold S (1988) Somatic embryogenesis and plant regeneration from suspension cultures of Picea glauca (white spruce). Plant Physiol 72:579–587. https://doi.org/10.1111/j.1399-3054.1988.tb09168.x
Inácio MC, Paz TA, Bertoni BW, Pereira AMS (2015) Germination of Cochlospermum regium seeds: influence of seed size, vials, vial sealing in vitro, and substrate in vivo. Eur J Med Plants 6(1):26–33. https://doi.org/10.9734/EJMP/2015/15077
Lai CC, Yu TA, Yeh SD, Yang JS (1998) Enhancement of in vitro growth of papaya multishoots by aeration. Plant Cell Tissue Organ Cult 53(3):221–225. https://doi.org/10.1023/A:1006004216935
Lamb CRC, Milach SCK (2001) Regeneração de plantas a partir de segmentos de base de folhas em aveia. Ciênc Rural 31(5):751–755. https://doi.org/10.1590/S0103-84782001000500002
Lenis-Manzano SJ, Araujo ACG, Valle CB, Santana EF, Carneiro VTC (2010) Histologia da embriogênese somática induzida em embriões de sementes maduras de Urochloa brizantha apomítica. Pesq Agropec Bras 45(5):435–441. https://doi.org/10.1590/S0100-204X2010000500001
Lloyd G, Mccown B (1980) Commercially-feasible micropropagation of mountain laurel, Kalmia latifolia, by use of shoot-tip culture. Proc Int Plant Propag Soc 30:421–427
Lorenzi H (2002) Árvores brasileiras: manual de identificação e cultivo de plantas arbóreas nativas do Brasil, vol 1. Nova Odessa, São Paulo
Matsumoto K, Cabral GB, Teixeira JB, Rech EL (1991) Embriogênese somática a partir de folhas imaturas de mandioca in vitro. Rev Bras Fisiol Veg 3(2):107–110
Mazri MA, Meziani R (2015) Micropropagation of date palm: a review. Cell Dev Biol 4(3):160. https://doi.org/10.4172/2168-9296.1000160 2015.
Mohamed MAH, Alsadon AA (2010) Influence of ventilation and sucrose on growth and leaf anatomy of micropropagated potato plantlets. Sci Hortic 123:295–300. https://doi.org/10.1016/j.scienta.2009.09.014
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Naaty B, Mbithe CM, Nyende AB, Njenga P, Muli JK (2017) In vitro regeneration via somatic embryogenesis of Schizozygia Coffeoides Baill (Mpelepele). Am J Plant Biol 2(2):66–72. https://doi.org/10.11648/j.ajpb.20170202.14
Nogueira RC, Paiva R, Castro AH, Vieira CV, Abbade LC, Alvarenga AA (2004) Germinação in vitro de murici-pequeno (Byrsonima intermedia A. Juss.). Ciênc Agrotecnol 28(5):1053–1059. https://doi.org/10.1590/S1413-70542004000500012
Nogueira RC, Paiva R, Oliveira LM, Soares GA, Soares FP, Castro AH, Paiva PDO (2007) Indução de calos em explantes foliares de murici-pequeno. Ciênc Agrotecnol 31(2):366–370. https://doi.org/10.1590/S1413-70542007000200015
Ouyang Y, Chen Y, Lü J, Teixeira da Silva JA, Zhang X, Ma G (2016) Somatic embryogenesis and enhanced shoot organogenesis in Metabriggsia ovalifolia W. T. Wang. Sci Rep 6:24662. https://doi.org/10.1038/srep24662
Pereira AR, Carvalho SP, Pasqual M, Santos FC (2007) Embriogênese somática direta em explantes foliares de Coffea arabica L. cv. Acaiá Cerrado: efeito de cinetina e ácido giberélico. Ciênc Agrotecnol 31(2):332–336. https://doi.org/10.1590/S1413-70542007000200010
Quatrano RS (1987) The role of hormones during seed development. In: Davies PJ (ed) Plant hormones and their role in plant growth and development. Kluwer Academic, Dordrecht, pp 494–514
Raju CS, Aslam A, Shajahan A (2015) High-efficiency direct somatic embryogenesis and plant regeneration from leaf base explants of turmeric (Curcuma longa L.). Plant Cell Tissue Organ Cult 122:79–87. https://doi.org/10.1007/s11240-015-0751-1
Rodrigues VEG, Carvalho DAd (2001) Plantas medicinais no domínio dos cerrados. UFLA, Lavras
Rout GR, Samantaray S, Das P (2000) In vitro manipulation and propagation of medicinal plants. Biotechnol Adv 18(2):91–120. https://doi.org/10.1016/S0734-9750(99)00026-9
Sa FP, Ledo AS, Amorim JAE, Silva AVC, Pasqual M (2016) In vitro propagation and acclimatization of genipapo accessions. Ciênc Agrotecnol 40(2):155–163. https://doi.org/10.1590/1413-70542016402036015
Sannomiya M, Cardoso CR, Figueiredo ME, Rodrigues CM, dos Santos LC, dos Santos FV, Serpeloni JM, Cólus IM, Vilegas W, Varanda EA (2007) Mutagenic evaluation and chemical investigation of Byrsonima intermedia A. Juss. leaf extracts. J Ethnopharmacol 112(2):319–326. https://doi.org/10.1016/j.jep.2007.03.014
Santana JRF Paiva R, Souza AV, Oliveira LM (2011) Effect of different culture tube caps and concentrations of activated charcoal and sucrose on in vitro growth and budding induction of Annona glabra L. Ciênc Agrotecnol 35(5):916–923. https://doi.org/10.1590/S1413-70542011000500008
Santos MRA, Paiva R, Gomes GAC, Paiva PDO, Paiva LV (2003) Estudos sobre superação de dormência em sementes de Smilax japecanga. Grisebach. Ciênc Agrotecnol 27(2):319–324. https://doi.org/10.1590/S1413-70542003000200010
Schenk RO, Hildebrandt AC (1972) Medium and techniques for induction and growth of monocotyledonous plant cell cultures. Can J Bot 50:199–204. https://doi.org/10.1139/b72-026
Sharp WR, Sondahl MR, Caldas LS, Maraffa SB (1980) The physiology on in vitro asexual embryogenesis. Hortic Rev 2:268–310. https://doi.org/10.1002/9781118060759.ch6
Silva LC, Paiva R, Swennen R, Andrè E, Panis B (2013) Shoot-tip cryopreservation by droplet vitrification of Byrsonima intermedia A. Juss.: a woody tropical and medicinal plant species from Brazilian cerrado. CryoLetters 34(4):338–348
Silva LC, Paiva R, Swennen R, Andrè E, Panis B (2014) Cryopreservation of Byrsonima intermedia embryos followed by room temperature thawing. Acta Sci Agron 36(3):309–315. https://doi.org/10.4025/actasciagron.v36i3.19688
Silva LC, Paiva R, Vargas DP, Silva DPC, Sandro Barbosa S, Herrera RC (2015) Decontaminant solution on in vitro growth of Byrsonima intermedia seedlings. Ciênc Rural 45(4):674–679. https://doi.org/10.1590/0103-8478cr20120078
Soares FP, Paiva R, Alvarenga AA, Nogueira RC, Emrich EB, Martinotto C (2007) Organogênese direta em explantes caulinares de mangabeira (Hancornia speciosa Gomes). Ciênc Agrotecnol 31(4):1048–1053. https://doi.org/10.1590/S1413-70542007000400016
Soares FP, Paiva R, Alvarenga AA, Nery FC, Vargas DP, Silva DRG (2011) Taxa de multiplicação e efeito residual de diferentes fontes de citocinina no cultivo in vitro de Hancornia speciosa Gomes. Ciênc Agrotecnol 35(1):152–157. https://doi.org/10.1590/S1413-70542011000100019
Souza RR, Paiva PDO, da Silva RR, Reis MV, Nery FC, Paiva R (2016) Optimization of jenipapo in vitro seed germination process. Ciênc Agrotecnol 40(6):658–664. https://doi.org/10.1590/1413-70542016406014816
Torres AC, Caldas LS, Buso JA (1999) Cultura de tecidos e transformação genética de plantas. EMBRAPA Hortaliças, Brasília
You XL, Tan X, Dai JL, Li YH, Choi YE (2012) Large-scale somatic embryogenesis and regeneration of Panax notoginseng. Plant Cell Tissue Organ Cult 108:333–338. https://doi.org/10.1007/s11240-011-0030-8
Acknowledgements
To the National Council for Scientific and Technological Development (CNPq), the Coordination for the Improvement of Higher Education Personnel (CAPES) and the Research Support Foundation of the State of Minas Gerais (FAPEMIG) for the financial support of this study.
Author information
Authors and Affiliations
Contributions
DPCdS: main author of the manuscript, performing all analysis, tabulating data and discussing the results with the other authors. PR: main researcher, responsible for the laboratory where the experiment was executed. RCH, LCS, GNF and MVdR: assisted in the implementation of tests, tabulation and interpretation of data and construction of related tables and charts. Authors also assisted with writing and discussion of all results found.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Sergio J. Ochatt.
Rights and permissions
About this article
Cite this article
da Silva, D.P.C., Paiva, R., Herrera, R.C. et al. Somatic embryogenesis of Byrsonima intermedia A. Juss.: induction and maturation via indirect approach. Plant Cell Tiss Organ Cult 133, 115–122 (2018). https://doi.org/10.1007/s11240-017-1366-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-017-1366-5