Abstract
Many sciences are described by two conceptually different but complementary theories, one visual and insightful the other better suited to computation. Chemistry has a powerful computational theory in quantum mechanics, but its visual and insightful theory of valence has never been properly developed. The rigorous theory of valence described here takes the atom as its fundamental particle. As the theory has no knowledge of the atom’s internal structure, the valence is not associated with the electron, hence it is free to adopt fractional values. Each atom is characterized by two experimental properties: its valence determined from the compositions of observed compounds, and its size determined from its observed coordination numbers. These two properties are able to describe a surprising amount of chemistry. The amount of valence an atom typically uses to form a bond determines both the conditions under which bonds can be formed and their resulting properties. A valence definition of electronegativity removes the need to distinguish between ionic and covalent bonds by defining the more nuanced concept of bond polarization, clearing up much of the confusion found in popular bonding models. The theory is compatible with the physical picture provided by quantum mechanics but not with the physically unrealistic ionic model. It can be used for analyzing, modeling, and teaching of chemical structure and reactivity, but having no knowledge of the internal structure of the atom it cannot be used to calculate energies or any of the properties that depend on the energy; for that quantum mechanics must be used.


Adapted from Fig. 2.2 of Brown [15]

Adapted from Fig. 2.3 of Brown [15]



Reproduced from Fig. 3.1 in Brown [15]. Copyright 2016 Oxford University Press Reproduced with the permission of the licensor through PLSclear






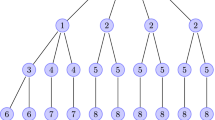
Adapted from Fig. 6.5 in Brown [15]





Copyright 2016 Oxford University Press Reproduced with the permission of the licensor through PLSclear

Copyright 2016 Oxford University Press Reproduced with the permission of the licensor through PLSclear
Similar content being viewed by others
Data availability
Not applicable.
References
Lewis GN (1923) Valence and the structure of molecules and crystals. Chemical Catalogue Co. Inc
McNaught AD, Wilkinson A (1997) Blackwell Scientific Publications, Oxford. Online version (20319-). ISBN 0–9678550–9–8. https://doi.org/10.1351/goldbook
Bader RWF (1990) Atoms in molecules, a quantum theory. Clarendon Press, Oxford
Tsirelson V, Stash A (2020) Orbital-free quantum crystallography: view on forces in crystals. Acta Cryst B76:769–778
Shannon RD, Prewitt CD (1969) Effective ionic radii in oxides and fluorides. Acta Cryst B25:925–946
Brown ID (2017) How big are atoms in crystals? Struct Chem 28:1377–1387. https://doi.org/10.1007/s11224-017-0942-y
Gibbs GV, Ross NL, Cox DF, Rosso KM (2014) Insights into the crystal chemistry of earth materials rendered by electron density distributions: Pauling’s rules revisited. Amer Min 99:1071–1084
Vadav P, Tandon H, Malik B, Suhag V, Chakraborty T (2022) A quest for the universal atomic radii. Struct Chem 33:389–394. https://doi.org/10.1007/s11224-021-01850-7
Gagné O, Hawthorne FC (2016) Bond-length distributions for ions bonded to oxygen: alkali and alkaline-earth metals. Acta Cryst B72:602–625
Gagné OC, Hawthorne FC (2018) Bond-length distributions for ions bonded to oxygen: metalloids and post-transition metals. Acta Cryst B74:63–78
Gagné OC, Hawthorne FC (2018) Bond-length distributions for ions bonded to oxygen: Results for the non-metals and discussion of lone-pair stereoactivity and the polymerization of PO4. Acta Cryst B74:79–96
Gagné OC, Hawthorne FC (2020) Bond-length distributions for ions bonded to oxygen: Results for the transition metals and quantification of the factors underlying bond-length variation in inorganic solids. IUCrJ 7:581–629. https://doi.org/10.1107/S2052252520005928
Brown ID (1988) What factors determine cation coordination numbers. Acta Cryst B44:545–553
Gagné O, Hawthorne FC (2017) Empirical Lewis-acid strengths for 135 cations bonded to oxygen. Acta Cryst B73:956–961
Brown ID (2016) The chemical bond in inorganic chemistry: the bond valence model, 2nd edn. OUP, Oxford
Rao GH, Brown ID (1998) Determination of bonding and valence distrtibution in inorganic solids by the maximum entropy method. Acta Cryst B54:221–230
Raebiger H, Lany S, Zunger A (2008) Charge self-regulation upon changing the oxidation state of transition metals in insulators. Nature 453:763–766
Pauling L (1960) The nature of the chemical bond, 3rd edn. Cornell University Press, Ithaca
Brown ID, Skowron A (1990) Electronegativity and Lewis acid strength. J Amer Chem Soc 112:3401–3403
Allred AL, Rochow EG (1958) A scale of electronegativity based on electrostatic force. J Inorg Nucl Chem 5:264–288
Preiser C, Lösel J, Brown ID, Kunz M, Skowron A (1999) Long range Coulomb forces and localised bonds. Acta Cryst B55:698–711
Day MC, Hawthorne FC (2020) A structural hierarchy for silicate minerals: chain, ribbon, and tube silicates. Miner Mag 84:165–244. https://doi.org/10.1180/mgn.2020.13
Lussier AJ, Hawthorne FC (2021) Structure topology and graphical representation of decorated and undecorated chains of edge-sharing octahedra. Can Miner 59:9–30
Day MC, Hawthorne FC (2022) Bond topology of chain, ribbon and tube silicates. Part I. Graph- theory generation of infinite one-dimensional arrangements of (TO4)n– tetrahedra. Acta Crystallogr A78:212–233
Gagné O, Mercier PHJ, Hawthorne FC (2018) A priori bond-valence and bond-length calculations in rock-forming minerals. Acta Cryst B74:470–482. https://doi.org/10.1107/S2052520618010442
Simon A, Dronskovsksi R, Krebs B, Hettich B (1987) Angew Chem 99:160–161
Pauling L (1947) Atomic radii and interatomic distances in metals. J Amer Chem Soc 69:542–554
Brown ID, Shannon RD (1973) Empirical bond-strength—bond-length curves for oxides. Acta Cryst A29:266–282
Brown ID, Altermatt D (1985) Bond-valence parameters obtained from a systematic analysis of the Inorganic Crystal Structure Database. Acta Cryst B41:244–247
Adams S (2001) Relationship between bond valence and bond softness of alkali halides and chalcogenides. Acta Cryst B57:278–287
Gagné O, Hawthorne FC (2015) Comprehensive derivation of bond-valence parameters for ion pairs involving oxygen. Acta Cryst B71:562–578
Brown ID (2016) On-line listing of bond valence parameters https://www.iucr.org/resources/data/datasets/bond-valence-parameters
Urusov VS (2003) Theoretical analysis and empirical manifestation of the distortion theorem. Zeit Krist 218:709–719
Lufaso MW, Woodward PM (2001) Prediction of the crystal structures of perovskites using the software program SPuDS. Acta Cryst B57:725–738
Harvey MA, Baggio S, Baggio R (2006) A new simplifying approach to molecular geometry description: the vectorial bond-valence model. Acta Cryst B62:1038–1042
van Driel M, Verweel HS (1938) Ueber dei Structur der Tripelnitrite. Zeit Krist 96:308–314
Alcock NW (1972) Secondary bonding to nonmetallic elements. Adv Inorg Rad Chem 15:1–57
Finger LW, Hazen RM (1978) Crystal structure and compression of ruby to 46 Kbar. J App Phys 49:5823–5826
Bickmore BR, Wander MFC, Edwards J, Maurer J, Shepherd K, Meyer E, Johansen WJ, Frank RA, Andros C, Davis M (2013) Electronic structure effects in the vectorial-bond-valence model. Amer Min 98:340–349
Brown ID (2009) On the valences of bonds in the oxycomplexes of Sn2+. Acta Cryst B65:684–693
Berustigui P, Hu VS, Eriksson SG (2001) A low temperature phase transition in CsPbF3. J Phys Cond Matter 13:5077–5088
Gillespie RJ, Hargittai I (1991) The VSEPR model of molecular geometry. Prentice Hall, New York
Gillespie RJ, Popelier PLA (2001) Chemical bonding and molecular geometry: from Lewis to electron densities. OUP, New York
Curnow OJ (1998) A simple qualitative molecular-orbital/valence-bond description of the bonding in main group hypervalent: molecules. J Chem Ed 75:910–915
Brown ID (1976) On the Geometry of O-H...O Hydrogen Bonds. Acta Cryst A32:24–31
Brown ID (1987) The structural chemistry and solvent properties of dimethyl sulfoxide. J Sol Chem 16:205–223
Duhlev R, Brown ID, Chr B (1991) Divalent metal halide double salts in equilibrium with their aqueous solutions I. Factors determining their compositions. J Solid State Chem 95:39–50
Hawthorne FC (1992) The role of OH and H2O in oxides and oxysalt minerals. Zeit Krist 201:183–206
Sanderson RT (1983) Polar covalence. Academic Press Inc., New York
Hirschfeld FL, Rzotkiewicz S (1974) Electrostatic binding in the first-row AH and A2 diatomic molecules. Mol Phys 27:1319–1343
Brown ID (1980) A structural model for lewis acids and bases. An analysis of the structural chemistry of the acetate and trifluoroacetate ions. J Chem Soc Dalton Trans 1980:1118–1123
Dunitz JD, Orgel LE (1960) Stereochemistry of inorganic solids. Adv Inorg Chem Radiochem 2:1–60
Kunz M, Brown ID (1995) Out-of-center distortions around octahedrally coordinated d0-transition metals. J Solid State Chem 115:395–406
Brown ID (2009) Recent developments in the methods and applications of the bond valence3 model. Chem Rev 109:6858–6919
Swenson J, Adams S (2001) The application of the bond valence method to reverse Monte Carlo produced structural models of superionic glasses. Phys Rev B 64:024204
Skowron A, Brown ID (1990) Refinement of the crystal structure of Robinsonite Pb4Sb6S13. Acta Cryst C46:527–531
van Smaalen S, Lüdecke J (2000) The valence states of vanadium in the low-temperature superstructure of NaV2O5. Europhys Lett 49:250–254
Wright SE, Foley FA, Hughes JM (2000) Optimization of site occupancies in minerals using quadratic programming. Amer Miner 85:524–531
Waltersson K (1978) A method based upon `bond strength’ calculations for finding probable lithium sites in crystal structures. Acta Cryst A34:901–905
Donnay G, Allmann R (1970) How to recognise O2−, OH− and H2O in crystal structures determined by x-rays. Amer Miner 55:1003–1015
Brown ID (1988) Chemical modelling of frenkel defects in fluorite. Solid State Ionics 31:203–208
Adams S (2000) Modeling conduction pathways in bond valence superpotential maps. Solid State Ionics 136–137:2424–3251
Adams S, Rao RP (2014) Understanding ion conduction and energy storage materials with bond-valence-based methods. In: Brown ID, Poeppelmeier KR (eds) Structures and Bonding 158 129–160, Springer, Dordrecht, Heidelberg, New York, London
Avdee M, Sale M, Adams S, Rao RP (2012) Screening of the alkali-metal ion containing materials from the Inorganic Crystal Structure Database (ICSD) for high ionic conductivity pathways using the bond valence method. Solid State Ionics 225:43–46
Sale M, Avdeev M (2012) 3DBVSMAPPER: a program for automatically generating bond-valence sum landscapes. J App Cryst 45:1054–1056
Withers RL, Thompson JG, Rae AD (1991) The crystal chemistry underlying ferroelectricity ini Bi4Ti3O12 and related Aurivillius phases. J Solid State Chem 93:404–417
Brown ID (1992) Modelling the Structures of La2NiO4. Zeit Krist 199:255–272
Salinas-Sanchez A, Garcia-Muñoz JL, Rodriguéz-Carvajal J, Saez-Puche R, Martinez JL (1992) Structural characterization of R2BaCuO5 (R = Y, Lu, Yb, Tm, Er, Ho, Dy, Gd, Eu and Sm) oxides by x-ray and neutron diffraction. J Solid State Chem 100:201–211
Green R, Vogt T (2012) Structures and self-activating photoluminescent properties of Sr3−xAxGaO4F (A=Ba, Ca) materials. J Solid State Chem 194:375–384
Sullivan E, Avdeev M, Vogt T (2012) Structure distortions in Sr3−xAxMO4F (A=Ca, Ba; M=Al, Ga, In) in anti-perovskites and corresponding changes in photoluminescence. J Solid State Chem 194:297–306
Lufaso MW, Woodward PM (2014) Using bond valences to model the structures of ternary and quaternary oxides. In Brown ID, Poeppelmeier KR (Eds) Struct Bond 158:59–90. Springer, Dordrecht, Heidelberg, New York, London
Zhao J, Ross NL, Angel RJ (2004) New view of he high-pressure behaviour of GdFeO3-type perovskites. Acta Cryst B60263–271
Zhao J, Ross NL, Angel RJ (2006) Estimation of polyhedral compressibilities and structural evolution of GdFeO3-type perovskites at high pressures. Acta Cryst B62:431–439
Whitfield RE, Welberry TR, Pasćiaka M, Goossens DJ (2014) Use of bond-valence sums in modelling the diffuse scattering from PZN (PbZn1/3Nb2/3O3). Acta Cryst A70:626–635
Levi E, Aurbach D, Isnard O (2013) Bond-valence model for cluster compounds. I Common lattice strains. Acta Cryst B69:419–425. https://doi.org/10.1107/s2052519213021271
Levi E, Aurbach D, Isnard O (2013) Bond-valence model for cluster compounds. II Matrix effect. Acta Cryst B69:427–438. https://doi.org/10.1107/s2052519213021283
Brown ID (1991) The influence of internal strain on the charge distribution and superconducting transition temperature in Ba2YCu3Ox. J Solid State Chem 90:155–167
Rao GH, Brown ID, Bärner E (1999) Structural effects in R0.5Sr0.5MnO3 perovskites (R = rare earth). J Phys Condens Matter 11:8103–8109
Dent-Glasser L (1979) Non-existent Silicates Zeit Krist 149:291–325
Brown ID (1981) The bond valence method: an empirical approach to chemical structure and bonding. pp 1–30 in O'Keeffe M, Navrotsky A Eds (1981) Structures and Bonding in Crystals. II. Proceedings of the Conference on Structure and Bonding in Crystals, Castle Hot Springs, Arizona, 1980, sponsored by Arizona State University. Academic Press: New York
Hawthorne FC (1985) Towards a structural classification of minerals: the VIMIVT2Φn minerals. Amer Miner 70:455–473
Hawthorne FC (2012) A bond-topological approach to theoretical mineralogy: crystal structure, chemical composition and chemical reactions. Phys Chem Miner 39:841–874
Hawthorne FC (2015) Toward theoretical mineralogy: a topological approach. Amer Min 100:696–713
Hawthorne FC, Schindler M (2008) Understanding the weakly bonded constituents in oxysalt minerals. Zeit Krist 223:41–68
Hawthorne FC, Della Ventura G, Oberti R, Robert J-L, Iezzi G (2005) Short range order in minerals: Amphiboles. Can Miner 43:1895–1920
Hawthorne FC, Schindler M (2014) Crystallization and dissolution in aqueous solution: a bond valence approach. in Bond Valences. in Brown ID, Poeppelmeier KR (Eds) Struct Bond 158:161–190. Springer, Dordrecht, Heidelberg, New York, London
Bargar JR, Brown GE Jr, Parks GA (1997) Surface complexation of Pb(II) at oxide water interfaces I. XAFS and bond valence determination of mononuclear and polynuclear Pb(II) sorption products on aluminum oxides. Geochim Cosmochim Acta 61:2617–2637
Bargar JR, Brown GE Jr, Parks GA (1997) Surface complexation of Pb(II) at oxide water interfaces II. XAFS and bond valence determination of mononuclear Pb(II) sorption products and surface functional groups on iron oxides. Geochim Cosmochim Acta 61:2639–2652
Bargar JR, Towle SN, Brown GE Jr, Parks GA (1997) XAFS and bond valence determination of the structures and compositions of surface functional groups and Pb(II) and Co(II) sorption products on single-crystal α-Al2O3. J Colloid Interface Sci 185:473–492
Xia K, Bleam W, Helmke PA (1997) Studies of the nature of Cu2+ and Pb2+ binding sites in soil humic substances using X-ray absorption spectroscopy. Geochim Cosmochim Acta 61:2211–2221
Xia K, Bleam W, Helmke PA (1997) Studies of the nature of binding sites of first row transition elements bound to aquatic and soil humic substances using X-ray absorption spectroscopy. Geochim Cosmochim Acta 61:2223–2235
Manceau A, Gates WP (1997) Surface structural model for ferrihydrite. Clay and Clay Miner 45:448–460
Brown GE Jr, Farges F, Calas G (1995) X-ray scattering and X-ray spectroscopy studies of mineral melts. Rev Miner 32:317–409
Brown ID, Dabkowski A, McCleary A (1997) The thermal expansion of chemical bonds. Acta Cryst B53:750–761
Wu Z, Farges F (1999) Anharmonicity around Th in crystalline oxide-type compounds: an in-situ high temperature XAFS spectroscopy study to 1500 °C. Physica B 266:282–289
Bickmore BR (2014) Structure and acidity in aqueous solutions and oxide–water interfaces. In: Valences B, Brown ID, Poeppelmeier KR (eds) Structures and Bonding 158. Springer, Dordrecht, Heidelberg, New York, London, pp 191–204
Mitchell KAR, Schlatter SA, Sodhi RNS (1986) Further analysis of surface bond lengths measured for chemisorption on metal surfaces. Can J Chem 64:1435–1439
Enterkin JA, Poeppelmeier KR (2014) Bonding at oxide surfaces. In: Valences B, Brown ID, Poeppelmeier KR (eds) (2014) Structure and Bonding 158. Springer, Dordrecht, Heidelberg, New York, London, pp 205–232
Yan Y, Chisholm MF, Duscher G, Pennycook SJ (1998) Atomic structure of a Ca-doped [001] tilt grain boundary in MgO. J Electron Microscopy 47:115–120
Browning ND, Buban JP, Moltaji HO, Duscher G (1999) Investigating the structure-property relatios at grain boundaries in MgO using bond valence pair potentials and multiple scattering analysis. J Amer Ceram Soc 82:366–372
Browning ND, Pennycook SJ (1996) Direct experimental determination of the atomic structure at internal interfaces. J Phys D 29:1779–1788
Browning ND, Moltaji HO, Buban JP (1998) Investigation of three-dimensional grain-boundary structure in oxides through multiple-scattering analysis of spatially resolved electron-energy-loss spectra. Phys Rev B 58:8289–8300
McGibbon MM, Browning ND, Chisholm MF, McGibbon AJ, Pennycook SJ, Ravikumar V et al (1994) Direct determination of the grain boundary atomic structure in SrTiO. Science 266:102–104
McGibbon MM, Browning ND, McGibbon AJ, Pennycook SJ (1996) The atomic structure of asymmetric [001] tilt boundaries in SrTiO3. Phil Mag A73:625–641
Andersson A (1982) An oxidized surface state model of vanadium oxides and its application to catalysis. J Solid State Chem 42:263–275
Depero LE (1993) Coordination geometry and catalytic activity of vanadium on TiO2. J Solid State Chem 103:528–532
Thorp HH (1992) Bond valence sum analysis of metal-ligand bond lengths in metalloenzymes and model complexes. Inorg Chem 31:1585–1588
Hati S, Datta D (1995) Nature of the active sites in haemocyanin and iron-rich hydgrogenases: the bond valence sum approach. J Chem Soc Dalton Trans 1995:1177–1182
Scarrow RC, Brennan BA, Cummings JG, Jia H, Duong DJ, Kindt JT et al (1996) X-ray spectroscopy of nitrile hydratase at pH7 and pH9. Biochem 55:10078–10088
Bell J, Ash DE, Snyder WM, Kulathila R, Blackburn NJ, Merkler DJ (1997) Structural and functional investigations on the role of zinc in bifunctional rat peptidylglycine α-amidating enzyme. Biochemistry 36:16239–16244
Clark-Baldwin K, Tierney DL, Govindaswamy N, Gruff ES, Kim C, Berg J et al (1998) The limitations of x-ray absorption spectroscopy for determining the structure of zinc sites in proteins. When is a tetrathiolate not a teterathiolate? J Amer Chem Soc 120:8401–8409
Dooley M, Scott RA, Knowles PK, Colangelo CM, McGuirl MA, Brown DE (1998) Structures of the Cu(I) and Cu(II) forms of amine oxidases from x-ray absorption spectroscopy. J Amer Chem Soc 120:2599–2605
Xiang S, Short SA, Wolfenden R, Carter CW (1996) Cytidene deaminase complexed to 3-deazacytidine: A “valence buffer” in zinc enzyme catalysis. Biochem 35:1335–1341
Deng H Jr, Burgner JW II, Callender R (1993) Comparison of vibrational frequencies of critical bonds in ground-state complexes and in a vanadate-based transition-state analog complex of muscle phosphoglucomutase. mechanistic implications. Biochemistry 32:12984–12992
Deng H, Burgner JW II, Callender RH (1998) Structure of the ribonuclease-uridine-vanadate transformation state analogue complex by Raman difference spectroscopy. Mechanistic implications J Amer Chem Soc 130:4717–4722
Ray WJ Jr, Burgner JW II, Deng H, Callender R (1993) Internal chemical bonding in solutions of simple phosphates and vanadates. Biochemistry 32:12977–12983
Müller P, Köpke S, Sheldrick GM (2003) Is the bond-valence method able to identify metal atoms in protein structures? Acta Cryst D59:32–37
Nayal M, Di Cera E (1994) Predicting Ca2+-binding sites in proteins. Proc Natl Acad Sci USA 91:817–821
Nayal M, Di Cera E (1996) Valence screening of water in protein crystals reveals potential Na+ binding sites. J Mol Biol 256:228–234
Garner CD, Collinson D, Pidcock E (1996) The nature of coordination sites of transition metals in proteins. Phil Trans R Soc Lond A 354:325–357
Shield GP, Raithby PR, Allen FA, Motherwell WDS (2000) The assignment and validation of metal oxidation states in the Cambridge Structural Database. Acta Cryst B56:455–465
Palenik GJ (1997) Bond valence sums in coordination chemistry using oxidation state independent Ro values. A simple calculation of the oxidation states of titanium in compounds containing Ti–N, Ti–O, and Ti–Cl bonds. Inorg Chem 36:3394–3397
Harris SE, Orpen AG, Bruno IJ, Taylor R (2005) Factors affecting d block metal-ligand bond lengths: Towards an automated library of molecular geometry for metal complexes. J Chem Inf Model 451727–1748
Hunter KH, Rodriguez J-MG, Becker NM (2022) A review of research on the teaching and learning of chemical bonding. J Chem Ed 99:2451–2464. https://doi.org/10.1021/acs.jchemed.2c00034
Bader RWF (2009) Bond paths are not chemical bonds. J Phys Chem 113:10391–10396
Acknowledgements
I wish to acknowledge the stimulating discussions I have had with Frank Hawthorne and Olivier Gagné. I wish also to acknowledge the many inspiring discussions I have had over the years with my McMaster University students and colleagues.
Author information
Authors and Affiliations
Contributions
I. D. Brown is responsible for the whole paper.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Competing interests
The author declares no competing interests.
Additional information
Dedicated to the memory of Jack Dunitz, Ron Gillespie and Richard Bader whose inspiration contributed to the ideas presented here.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- a priori bond valence (‘Simple bonds’ section)
-
Bond valences calculated from the bond network using the network Eqs. 4 and 5. Using Eq. 7 it can be converted into an a priori bond length.
- acid
-
See Lewis acid.
- atom (‘Atoms’ section)
-
The basic particle of valence theory. The atomic number of an atom indicates which element it is. In valence theory, atoms are characterized by their valence and their average coordination number. They also possess the properties of bonding strength and electronegativity. Atoms are always spherical and electroneutral. Their physical structure is is most conveniently described by the core and valence shell model.
- atomic valence V (‘First things: valence, coordination number, and equilibrium’ section)
-
The same as valence. This name is used when it is necessary to avoid confusion with the bond valence.
- average coordination number < N > (‘First things: valence, coordination number, and equilibrium’ section)
-
The average of the experimentally measured coordination numbers of an atom with a given valence. Unless otherwise stated the ligands are assumed to be oxygen.
- base
-
See Lewis base.
- bond (‘Atoms’ section)
-
The attachment that links two atoms when their valence shells overlap. Bonds possess the properties of bond valence and polarization as well as bond length.
- bond flux φ (‘Simple bonds’ section)
-
A physical description of the bond valence in terms of the electrostatic flux linking the cores of two bonded atoms to their shared valence It can be calculated using the ionic model and is expected to be the same as the bond valence.
- bond length R (‘Simple bonds’ section)
-
The distance between the centers of the two atoms.
- bond network (‘Simple bonds’ section)
-
A network of bonds linking the atoms that form a compound. In valence theory the bond network is the primary description of the structure of the compound. The properties of the bonds, such as their bond valence. bond polarization and bond length are largely determined by the topology of the bond network and the identity of the constituent atoms.
- bond polarization ψ (‘Simple bonds’ section)
-
The degree of polarization of a bond, equal to the difference in the electronegativities of the two atoms that form the bond.
- bond polarity (‘Simple bonds’ section)
-
The bond polarization describes the bond as directed from the atom with the larger to the atom with the smaller electronegativity.
- bond valence s (‘First things: valence, coordination number, and equilibrium’ section)
-
The amount of valence an atom uses to form a given bond, hence it is a measure of the strength of the bond. In most cases both atoms contribute equal amounts of valence to form a bond. Depending on how its value is determined the valence is known as the a priori bond valence. the bond flux or the experimental bond valence.
- bond valence vector s (‘Simple bonds’ section)
-
A vector directed along the line joining two bonded atoms having the magnitude of the bond valence.
- bonding strength S (‘Atoms’ section)
-
The bond valence of a typical bond formed by an atom. It is equal to the valence of the atom divided by its corresponding average coordination number (Eq. 1). The bonding strength of the atom with the smaller electronegativity is also known as its Lewis acid strength, that of the atom with the larger electronegativity is also known as its Lewis base strength.
- complex (‘Interesting Bonds’ section)
-
A collection of strongly bonded atoms including those traditionally called molecules or complex ions. The outer surface of the complex is normally composed of Lewis bases. Hydrogen atoms, if attached, act as its Lewis acid function. The bonding strengths of complexes are the external bonding strengths of the atoms on the surface of the complex.
- compound
-
An aggregation of atoms held together by bonds.
- coordination number N
-
The number of bonds formed by an atom.
- coordination sphere
-
The atoms that are bonded to the given atom.
- core (‘Atoms’ section)
-
In the core and valence shell model of an atom the core is the portion of an atom that is never used for bonding. It includes the atomic nucleus and the portion of the negative charge that is not part of the valence shell.
- core and valence shell model (‘Atoms’ section)
-
The core and valence shell model provides a notional picture of an atom that is useful in understanding the atom’s properties. It is not a true physical picture of an atom, nor is it an essential part of valence theory.
- deformation density (‘Atoms’ section)
-
The difference between the promolecule charge density and the experimentally observed negative charge density in a given compound.
- effective Lewis acid strength S ae(‘Interesting bonds’ section)
-
The Lewis acid strength of the solvent water at a given pH.
- effective Lewis base strength S be (‘Interesting bonds’ section)
-
The Lewis base strength of the solvent water at a given pH.
- electronegativity χ (‘Atoms’ section)
-
In valence theory this is defined for Main Group elements as the bonding strength of the atom in its highest valence state. For hydrogen see ‘Hydrogen bonds and aqueous chemistry’ section and for transition metals see ‘Ammonia and the ammonium complex’ section. It is an intrinsic property of an atom.
- experimental atomic valence (‘Simple bonds’ section)
-
The sum of the experimental bond valences of the bonds formed by the given atom.
- experimental bond valence p (‘Simple bonds’ section)
-
The bond valence determined from the observed bond lengths using Eq. 7.
- external valence (‘Interesting Bonds’ section)
-
The valence available to a complex for forming bonds to other atoms in the compound that are not part of the complex. It is equal to the sum of the valences of the atoms forming a complex, having regard to their signs. It is also equal to the sum of the valences of the external bonds formed by the individual atoms in the complex.
- flux, electrostatic flux (‘Simple bonds’ section)
-
See bond flux.
- Lewis acid (‘Simple bonds’ section)
-
In a bond, the atom with the lower electronegativity.
- Lewis acid strength S a (‘Simple bonds’ section)
-
The bonding strength of a Lewis acid.
- Lewis base (‘Simple bonds’ section)
-
In a bond, the atom with the higher electronegativity.
- Lewis base strength S b (‘Simple bonds’ section)
-
The bonding strength of a Lewis base.
- lone pair (‘Interesting Bonds’ section)
-
Two electron units of charge in the valence shell of an atom that are not used for bonding. Lone pairs become stereoactive if the atom is bonded to an atom with a larger bonding strength (Rule 10).
- primary bond (‘Interesting Bonds’ section)
-
A bond that is significantly stronger then the average in a coordination sphere, particularly in the presence of stereoactive lone pairs.
- promolecule (‘Atoms’ section)
-
The charge density (electron density) created by placing the charge densities of the isolated neutral atoms at the positions of the atoms in the given compound.
- secondary bond (‘Interesting Bonds’ section)
-
A bond that is significantly weaker than the average in a coordination sphere, particularly in the presence of stereoactive lone pairs.
- valence V (‘First things: valence, coordination number, and equilibrium’ section)
-
Defined by IUPAC (Rule 1), valence is a number that indicates the number of oxygen or hydrogen atoms with which it can combine to form a binary compound, thus valence can only be determined when an atom is part of a compound. According to this definition valence is a positive number, but in valence theory valence is identified with that portion of the negative charge of an atom that is used to form bonds. It is measured in valence units, each of which is equal to the electron charge. Valences of atoms are usually integers, though there are exception. The valence adopted by an atom depends on its atomic number and the electronegativities of its bonded neighbors as described by Rule 10. In the core and valence shell model the valence is always located in the valence shell. It may be referred to as the atomic valence to differentiate it from the bond valence.
- valence shell (‘Atoms’ and ‘Interesting Bonds’ section)
-
The notional outer shell of an atom in the core and valence shell model. It contains all the charge representing the valence of the atom but may also contain non-bonding negative charge called lone pairs.
- valence unit (vu) (‘First things: valence, coordination number, and equilibrium’ section)
-
The unit of valence associated with one electron unit of charge, see valence.
- valence vector
-
See bond valence vector.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Brown, I.D. A rigorous theory of valence. Struct Chem 34, 361–389 (2023). https://doi.org/10.1007/s11224-023-02128-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-023-02128-w