Abstract
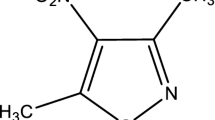
Calorimetry provides an accurate and reliable method to determine the enthalpies of formation of organic molecules in the gas phase. From the experimental formation enthalpies and the absolute entropies, obtained by theoretical calculations, it is possible to perform Gibbs energy calculations. This thermodynamic function is widely used to describe various thermodynamic processes, such as chemical equilibrium, and allows the calculation of equilibrium constants. The specific standard combustion energies of 3,5-dimethylisoxazole-4-carboxylic acid, 5-methylisoxazole-3-carboxylic acid, 5-amino-3-methylisoxazole, and 3-amino-5-methylisoxazole were determined by using a combustion calorimeter. The sublimation enthalpies of the compounds were determined by means of the Knudsen effusion technique. From these values, the molar standard enthalpy of formation in gaseous phase of each compound was calculated. Theoretical calculations at the G3 and G4 levels were performed, and a study of the molecular and electronic structure of these compounds was carried out. The calculated enthalpies of formation are in very good agreement with the experimental values. From both the experimental and theoretical results, five gas phase chemical equilibria were studied: one of isomerization, two of CO2 loss, and two of NH3 loss. The equilibrium constants for each process were calculated, which allow prediction of the direction of the chemical process from a thermodynamic viewpoint.










Similar content being viewed by others
References
Morohashi H, Sato H (2005)| Nuevos derivados de sulfonamida, España ES 2 224 395 T3
Bartlett RR (1995) Medicamento para el tratamiento de reacciones de rechazo en trasplantes de órganos, España ES 2 065 738 t3
Cross B, Los M, Doehner Jr RF, Ladner DW, Johnson JL, Jung ME, Kamhi VM, Tseng SS, Finn JM, Wepplo PJ (1994) Procedimiento para la preparación de composiciones herbicidas de (2-imidazolin-2-il)-heteropiridina condensada, España ES 2 046 167
Lerman O, Hermolin J (1989) Un procedimiento mejorado para la preparación de 3-amino-5-metilisoxazol, España ES 2 005 019
Flores H, Camarillo EA, Amador P (2012) Enthalpies of combustion and formation of Benzenesulfonamide and some of its derivatives. J Chem Thermodyn 47:408–411
Flores H, Mentado J, Camarillo EA, Ximello A, Amador MP (2013) The standard molar enthalpies of combustion and formation of 2-Methylbenzothiazole, 2-Methylbenzoxazole, and 2-methyl-2-thioazoline. Struct Chem 24:2083–2088
Camarillo EA, Mentado J, Flores H, Hernández-Pérez JM (2014) Standard enthalpies of formation of 2-Aminobenzothiazoles in the crystalline phase by rotating-bomb combustion calorimetry. J Chem Thermodyn 72:296–273
Wendlandt WW (1986) Thermal analysis, 3rd edn. Wiley, New York
Höhne GWH, Hemminger WF, Flammercheim HJ (2003) Differential scanning calorimetry, 2nd edn. Springer-Verlag, New York
Chiu L, Liu H, Li M (1999) Heat capacity of Alkanolamines by differential scanning calorimetry. J Chem Eng Data 44:631–636
Becker L, Aufderhaar O, Gmehling J (2000) Measurment of heat capacities for nine organic substances by tian – Calvet calorimetry. J Chem Eng Data 45:661–665
Meija T, Coplen TB, Berglund M, Brand WA, De Bièvre P, Gröning M, Holden NE, Irrgeher J, Loss RD, Walczyk T, Prohaska T (2016) Atomic weights of the elements 2013 (IUPAC technical report). Pure Appl Chem 88(3):265–291
Pan ML, Lou YH, Li JF (2011) 5-methyl-1,2-ozaxole-3-carboxylic acid. Acta Cryst E67:o2545
Good WD, Smith NK (1969) Enthalpies, of combustion of toluene, benzene, cyclohexane, cyclohexene, methylcyclopentane, 1-methylcyclopentene, and n-hexane. J Chem Eng Data 14:102–106
Roux MV, Dávalos JZ, Jiménez P, Flores H, Sainz JL, Abboud JL (1999) Structural effects on the thermochemical properties of sulfur compounds: I. Enthalpy of combustion, vapour pressures, enthalpy of sublimation, and standard molar enthalpy of formation in the gaseous phase of 1,3-ditiane. J Chem Thermodyn 31:635–646
Washburn EN (1933) Standard states for bomb calorimetry. J Res Natl Bur Stand (US) 10:525–558
National Institute of Standard and Technology, Certificate of Analysis, Standard Reference Material 39j Benzoic Acid (Calorimetric Standard), (2007), 1–4.
Flores H, Amador P (2004) Standard molar enthalpies of formation of crystalline stereoisomers of Aldo-1, 4-lactones. J Chem Thermodyn 36:1019–1024
Verevkin SP (1998) Thermochemistry of amines: strain in six-membered rings from experimental standard molar enthalpies of formation of Morpholines and Piperazines. J Chem Thermodyn 30:1069–1079
Mentado J, Flores H, Amador P (2008) Combustion energies and formation enthalpies of 2-SH-benzazoles. J Chem Thermodyn 40:1106–1109
Wadsö I (1966) Calculation methods in reaction calorimetry. Sciencie Tools 13:33–39
Santos LMNBF, Silva MT, Schröder B, Gomes L (2007) Labtermo: methodologies for the calculation of the corrected temperature rise in Isoperibol calorimetry. J Therm Anal Cal 89:175–180
Knudsen M (1909) Experimental determination of vapor pressure of mercury at 0° and higher temperatures. Ann Phys 29:179–193
García-Castro M, Amador P, Hernández-Pérez JM, Medina-Favela AE, Flores H (2014) Experimental and computational thermochemistry of 3- and 4-Nitrophtalic anhydride. J Phys Chem A 118:3820–3826
Sabbah R, Xu-wu A, Chickos J, Planas M, Roux M, Torres L (1999) Reference materials for calorimetry and differential thermal analysis, Thermochim. Acta 331:93–204
Knudsen M (1909) The law of the molecular flow and viscosity of gases moving through tubes. Ann Phys 28:75–130
Knudsen M (1909) Effusion and the molecular flow of gases through openings. Ann Phys 28:999–1016
Clausing P (1932) Über die Strömung sehr Verdünnter Gase durch Röhren von Beliebiger Länge. Ann Phys 12:961–989
Ribeiro da Silva MAV, Monte MJS, Santos LMNFB (2006) The design, construction, and testing of a new Knudsen effusion apparatus. J Chem Thermodyn 38:778–787
Frisch, M.J., Trucks, G.W., Schlegel, H.B., Scuseria, G.E., Robb, M.A., Cheeseman, J.R., Scalmani, G., Barone, V., Mennucci, B., Petersson, G.A., Nakatsuji, H., Caricato, M., Li, X., Hratchian, H.P., Izmaylov, A.F., Bloino, J., Zheng, G., Sonnenberg, J.L., Hada, M., Ehara, M., Toyota, K., Fukuda, R., Hasegawa, J., Ishida, M., Nakajima, T., Honda, Y., Kitao, O., Nakai, H., Vreven, T., Montgomery, Jr., J.A., Peralta, J.E., Ogliaro, F., Bearpark, M., Heyd, J.J., Brothers, E., Kudin, K.N., Staroverov, V.N., Kobayashi, R., Normand, J., Raghavachari, K., Rendell, A., Burant, J.C., Iyengar, S.S., Tomasi, J., Cossi, M., Rega, N., Millam, J.M., Klene, M., Knox, J.E., Cross, J.B., Bakken, V., Adamo, C., Jaramillo, J., Gomperts, R., Stratmann, R.E., Yazyev, O., Austin, A.J., Cammi, R., Pomelli, C., Ochterski, J.W., Martin, R.L., Morokuma, K., Zakrzewski, V.G., Voth, G.A., Salvador, P., Dannenberg, J.J., Dapprich, S., Daniels, A.D., Farkas, Ö., Foresman, J.B., Ortiz, J.V., Cioslowski, J., Fox, D.J. (2009). Gaussian 09, Revision A.02, Gaussian, Inc. Wallingford, CT
Curtis LA, Raghavachari K, Redfern PC, Rassolov V, Pople JA (1998) Gaussian-3 (G3) theory for molecules containing first and second-row atoms. J Chem Phys 109:7764–7776
Curtiss LA, Redfern PC, Raghavachari K (2007) Gaussian-4 theory. J Chem Phys 126:084108
Kochetkov NK, Sokolov SD, Vagurtova NM, Nifant’ev EE (1960) Organomagnesium compounds of isoxazoles series. Dokl Akad Nauk SSSR 133:598–601
Shaw, G., Sugowdz, G. (1954) Isoxazolinones. VI. The hydrogenation of 5-amino-isoxazoles. A new synthesis of pyrimidines, Journal of the Chemical Society, 665–668
Kochetkov NK, Sokolov SD (1963) Isoxazole series. XV. Organomagnesium synthesis in the isoxazoles series. Zhurnal Obshchei Khimii 33:1196–1199
Kano H, Ogata K, Nishimura H, Nakajima K (1959) 3-sulfanilamido-5-methylisoxazole, Japan JP 34005566
Iwai I, Nakamura N (1966) Acetylenic compounds. XLIV. Synthesis of 3-aminoisoxazoles and 3-hydroxyisoxazoles (3-isoxazolones). Chem Pharm Bull 14:1277–1286
Grob CA (1950) Structure of ethyl β-acetamido crotonate. Helvetica Chimica Acta 33:1787–1796
Kurtz P, Gold H, Disselnkotter H (1959) Nitrile formation. III. 1-cyano-2-alkynes and 1-cyano-1,2-alkadienes. Justus Liebigs Annalen der Chemie 624:1–25
Hubbard WN, Scott DW, Waddington G, Rossini FD (Ed.) (1956) Experimental Thermochemistry, Interscience, New York
Cox JD, Wagman DD, Medvedev VA (eds) (1987) CODATA key values for thermodynamics. Hemisphere, New York
Chirico RD, Frenkel M, Diky VV (2003) ThermolML – an XML-based approach for storage and exchange of experimental and critically evaluated Thermophysical and thermochemical property data. 2. Uncertainties. J Chem Eng Data 48:1344–1359
Sunner S, Olofsson G, Mansson M (1979) Combustion Calorimetry, Eds., Pergamon Press, Oxford.
Hamilton WS, Ayers DA (1973) Heat of combustion of 3-amino-5-methylisoxazole. J Chem Eng Data 18:366–367
Merrick JP, Moran D, Radom L (2007) An evaluation of harmonic vibrational frequency scale factors. J Phys Chem A 111:11683–11700
Notario R, Castaño O, Abboud JL, Gomperts R, Frutos LM, Palmeiro R (1999) Organic thermochemistry at high ab initio levels. 1. A G2 (MP2) and G2 study of cyclic saturated and unsaturated hydrocarbons (including aromatics). J. Org. Chem. 64:9011–9014
Notario R, Castaño O, Gomperts R, Frutos LM, Palmeiro R (2000) Organic thermochemistry at high ab initio levels. 3. A G3 study of cyclic saturated and unsaturated hydrocarbons (including aromatics). J Org Chem 65:4298–4302
Glukhovtsev MN, Laiter S (1995) High level ab Initio stabilization energies of benzene. Theor Chim Acta 92:327–332
Minkin VI (1999) Glossary of terms used in theoretical chemistry. Pure Appl Chem 71:1919–1981
Raghavachari K, Stefanov BB, Curtiss LA (1997) Accurate thermochemistry for larger molecules: Gaussian-2 theory with bond separation energies. J Chem Phys 106:6764–6767
Chase Jr MW (1998) NIST-JANAF thermochemical tables. J Phys Chem Ref Data Monograph 9:1–1951
Hamilton WS, Benton S, French J, McCormick D, Pustejovsky S, Thompson P (1978) Enthalpies of combustion and formation of 3-methylisoxazole and 5-methylisoxazole. J Chem Eng Data 23:201–203
Ramos F, Flores H, Rojas A, Hernándes-Pérez JM, Camarillo EA, Amador MP (2016) Experimental and computational thermochemical study of benzofuran, benzothiophene and indole derivatives. J Chem Thermodyn 97:297–306
Camarillo EA, Flores H, Perdomo G (2014) Determination of energetic content for 3,5- dimethylisoxazole like Antilipolityc drug. J Chem Bio Phy Sci Sec C 4:88
Acknowledgements
Thanks are due to VIEP-BUAP for financial support through FLSH-NAT15-G project. G. P. thanks CONACYT for his grant (321454).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
G3- and G4-calculated energies, at T = 0 K, E 0, and enthalpies at T = 298 K, H 298, in Hartrees; values of the combustion experiments of benzoic acid to T = 298.15 K, p° = 0.1 MPa; typical values of the combustion experiments for 35dmi4c to T = 298.15 K, p° = 0.1 MPa; typical values of the combustion experiments for 5mi3c to T = 298.15 K, p° = 0.1 MPa; typical values of the combustion experiments for 5a3mi to T = 298.15 K, p° = 0.1 MPa; typical values of the combustion experiments for 3a5mi at T = 298.15 K, p° = 0.1 MPa; effusion experimental results of 35dmi4c at T med = 343.7 K; experimental effusion results of 5mi3c at T med = 343.7 K; experimental effusion results of 3a5mi at T med = 297.3 K; experimental effusion results of 5a3mi at T med = 313.7 K; experimental values of temperature, intercept, slope, and enthalpy of sublimation at T med for compounds studied; experimental values of vapor pressure for the compounds studied; areas and clausing factors of the cell effusion holes; G3- and G4-calculated enthalpies at T = 298 K, and experimental enthalpies of formation in the gas phase for the molecules used as references in isodesmic reactions; formation enthalpies in gas phase of each isodesmic reaction for each compound studied. The values were obtained using Gaussian-n theories at G3 and G4 levels; the absolute entropies of each element used to calculate the variation of entropy of each compound; Cartesian coordinates of the compounds studied.
.
ESM 1
(DOCX 440 kb)
Rights and permissions
About this article
Cite this article
Perdomo, G., Flores, H., Notario, R. et al. Enthalpies of formation of four isoxazole derivatives in the solid and gas phases: application to the study of chemical equilibria. Struct Chem 28, 1111–1123 (2017). https://doi.org/10.1007/s11224-017-0923-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-017-0923-1