Abstract
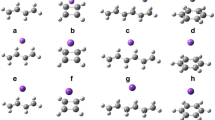
By doping π-π systems with Li atom, a series of Li@sandwich configuration and Li@T-shaped configuration compounds have been theoretically designed and investigated using density functional theory. It is revealed that energy gaps (E gap) between highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) of all compounds are in a range of 0.4–0.9 ev. When Li atom is introduced into different sandwich configuration π-π systems (C60-toluene, C60-fluorobenzene, C60-phenol, C60-benzonitrile), Li@C60-benzonitrile exhibits considerable first hyperpolarizability as large as 19,759 au, which is larger by about 18,372–18,664 au than those of other compounds. When Li atom is introduced into different T-shaped configuration π-π systems (C60-pyridine, C60-pyrazine, C60-1, 3, 5-triazine, C60-pyridazine), Li@C60-pyridazine is found to present largest first hyperpolarizability up to 67,945 au in all compounds. All compounds are transparency in the deep ultraviolet spectrum range. We hope that this study could provide a new idea for designing nonlinear optical materials using π-π systems as building blocks.






Similar content being viewed by others
References
Sherrill CD (2013) Acc Chem Res 46:1020–1028
Mignon P, Loverix S, Geerlings P (2005) Chem Phys Lett 401:40–46
Ercolani G, Mencarelli P (2003) J Organomet Chem 68:6470–6473
Mishra BK, Sathyamurthy N (2005) J Phys Chem A 109:6–8
Grimme S (2008) Angew Chem Int Ed 47:3430–3434
Sinnokrot MO, Sherrill CD (2004) J Phys Chem A 108:10200–10207
Meyer EA, Castellano RK, Diederich F (2003) Angew Chem Int Ed 42:210–1250
Burley SK, Petsko GA (1985) Science 23:229
Mulliken RS (1952) J Am Chem Soc 74:811–824
Mcneil AJ, Muller P, Whitten JE, Swager TM (2006) J Am Chem Soc 128:12426–12427
Hunter CA, Meah MN, Sanders JKM (1990) J Am Chem Soc 112:5773–5780
Philp D, Stoddart JF (1996) Angew Chem, Int Ed Engl 35:1154–1196
Lerman LS (1961) J Mol Biol 3(1):18IN13–30IN14
Saenger W (1984) Principles of nucleic acid structure. Springer, New York
Hunter CA, Sanders JKM (1990) J Am Chem Soc 112:5525–5534
Arunan E, Gutowsky HS (1993) J Chem Phys 98:4294–4296
Sinnokrot MO, Valeev EF, Sherrill CDI (2002) J Am Chem Soc 124:10887–10893
Tsuzuki S, Honda K, Uchimaru T, Mikami M, Tanabe K (2002) J Am Chem Soc 124:104–112
Sinnokrot MO, Sherrill CD (2004) J Am Chem Soc 126:7690–7697
Hohenstein EG, Sherrill CD (2009) J Phys Chem A 113:878–886
Nakano M, Fujita H, Takahata M, Yamaguchi K (2002) J Am Chem Soc 124:9648–9655
Geskin VM, Lambert C, Brédas JL (2003) J Am Chem Soc 125:15651–15658
Ostroverkhova O, Moemer WE (2004) Chem Rev 104:3267–3314
Coe BJ (2006) Acc Chem Res 39:383–393
Xu HL, Li ZR, Wu D, Ma F, Li ZJ, Gu FL (2009) J Phys Chem C 113:4984–4986
Hu YY, Sun SL, Muhammad S, Xu HL, Su ZM (2010) J Phys Chem C 114:19792–19798
Xu HL, Li ZR, Wu D, Wang BQ, Li Y, Gu FL, Aoki Y (2007) J Am Chem Soc 129:2967–2970
Wu HQ, Zhong RL, Sun SL, Xu HL, Su ZM (2014) J Phys Chem C 118:6952–6958
Wang SJ, Li Y, Wang YF, Wu D, Li ZR (2013) Phys Chem Chem Phys 15:12903–12910
Xu HL, Zhang CC, Sun SL, Su ZM (2012) Organometallics 31:4409–4414
Shelton DP, Rice JE (1994) Chem Rev 94:3–29
Willets A, Rice JE, Burland DM, Shelton DP (1992) J Chem Phys 97:7590–7599
Kanis DR, Ratner MA, Marks TJ (1994) Chem Rev 94:195–242
Huang W, Sergeeva AP, Zhai HJ, Averkiev BB, Wang LS, Boldyrev AI (2010) Nat Chem 2:202–206
Jimenez-Halla JOC, Islas R, Heine T, Merino G (2010) Angew Chem Int Ed 49:5668–5671
Uchino T, Kurumoto N, Natsuko S (2006) Phys Rev B 73:233203
Fazio G, Ferrighi L, Valentin CD (2014) J Catal 318:203–210
Zaboli M, Raissi H (2010) Struct Chem 26:1059–1075
Champagne B, Botek E, Nakano M, Nitta T, Yamaguchi K (2005) J Chem Phys 122:114315
Zhang CC, Xu HL, Hu YY, Sun SL, Su ZM (2011) J Phys Chem A 115:2035–2040
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery Jr JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2013) Gaussian 09. Gaussian Inc., Wallingford
Dennington R, Keith T, Millam JGV (2009) GaussView, version 5. Semichem, Shawnee Mission, KS
Lu T, Chen FW (2012) J Comput Chem 33:580–592
Chen W, Li ZR, Wu D, Li Y, Sun CC, Gu FL, Aoki Y (2006) J Am Chem Soc 128:1072–1073
Oudar JL, Chemla DS (1977) J Chem Phys 66:2664–2668
Oudar JL (1977) J Chem Phys 67:446–457
Datta A, Pati SK (2006) Chem Soc Rev 35:1305–1323
Acknowledgements
The authors gratefully acknowledge financial support from the Fujian University of Technology (GY-Z13109), the Education Department of Fujian Province (JB14075), and the Development Fund of Fujian University of Technology (GY-Z160127).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Song, YD., Wang, L. & Wu, LM. Constructing a novel nonlinear optical materials: substituents and heteroatoms in π-π systems effect on the first hyperpolarizability. Struct Chem 28, 1623–1630 (2017). https://doi.org/10.1007/s11224-017-0918-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-017-0918-y