Abstract
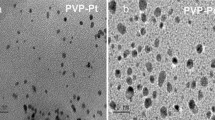
An efficient and stable nanocatalyst for selective oxidation of alcohols was developed. It contains palladium nanoparticles, which are well distributed throughout the network of a copolymer based on an ionic liquid. The synthesized nanomaterials were characterized by various techniques such as nitrogen adsorption—desorption analysis, thermal gravimetric analysis, TEM, and FTIR spectroscopy. A high surface area and the appropriate pore size of the nanocatalyst make active metal sites accessible to reagents, whereas the presence of an ionic liquid in the network of the polymer structure creates a good environment for the leaching protection and stabilization of extremely dispersed palladium nanoparticles. The availability and abundance of active sites of highly dispersed palladium nanoparticles make the synthesized nanocatalyst very promising for oxidation of alcohols. The nanocatalyst has a number of features such as a high surface area, an appropriate size of pores with high catalytic activity, high thermal stability of the nanostructures, and low amounts of the ionic liquid needed for its synthesis. Using this nanocatalyst, carbonyl compounds were prepared from the corresponding alcohols in high yields. Additionally, the prepared nanocatalyst can easily be recovered by centrifugation after completion of the reactions and was reused five times without a significant loss in its catalytic activity.
Similar content being viewed by others
References
G. Urgoitia, G. Galdón, F. Churruca, R. SanMartin, M. T. Herrero, E. Domínguez, Environ. Chem. Lett., 2018, 16, 1101; DOI: https://doi.org/10.1007/s10311-018-0730-y.
I. Ibrahem, M. N. Iqbal, O. Verho, A. Eivazihollagh, P. Olsén, H. Edlund, C. W. Tai, M. Norgren, E. V. Johnston, Chem. Nano Mater., 2018, 4, 71; DOI: https://doi.org/10.1002/cnma.201700309.
G. Marcì, E. García-López, L. Palmisano, Catal. Today, 2018, 315, 126; DOI: https://doi.org/10.1016/j.cattod.2018.03.038.
Y. Han, M. Zhang, Y.-Q. Zhang, Z.-H. Zhang, Green Chem., 2018, 20, 4891; DOI: https://doi.org/10.1039/C8GC02611D.
T. Punniyamurthy, S. Velusamy, J. Iqbal, Chem. Rev., 2005, 105, 2329; DOI: https://doi.org/10.1021/cr050523v.
E. Gopi, E. Gravel, E. Doris, Nanoscale Adv., 2019, 1, 1181; DOI: https://doi.org/10.1039/C8NA00192H.
G. Csjernyik, A. H. Éll, L. Fadini, B. Pugin, J. E. Bäckvall, J. Org. Chem., 2002, 67, 1657; DOI: https://doi.org/10.1021/jo0163750.
Z. Ma, Q. Wang, E. CBA Alegria, C. G. da Silva, M. Fátima, L. MDRS Martins, J. P. Telo, I. Correia, A. JL Pombeiro, Catalysis, 2019, 9, 424; DOI: https://doi.org/10.3390/catal9050424.
B. Heidari, M. M. Heravi, M. R. Nabid, R. Sedghi, Appl. Organomet. Chem., 2019, 33, 4934; DOI: https://doi.org/10.1002/aoc.4934.
A. Ghorbani-Choghamarani, B. Tahmasbi, R. H. Hudson, A. Heidari, Micropor. Mesopor. Mater., 2019, 248, 366; DOI: https://doi.org/10.1016/j.micromeso.2019.04.061.
J. Jiang, G. H. Gunasekar, S. Park, S.-H. Kim, S. Yoon, L. Piao, Mater. Res. Bull., 2018, 100, 184; DOI: https://doi.org/10.1016/j.materresbull.2017.12.018.
R. P. Bagwe, L. R. Hilliard, W. Tan, Langmuir, 2006, 22, 4357; DOI: https://doi.org/10.1021/la052797j.
J. R. Conway, A. S. Adeleye, J. Gardea-Torresdey, A. A. Keller, Environ. Sci. Technol., 2015, 49, 2749; DOI: https://doi.org/10.1021/es504918q.
Y. Qie, H. Yuan, C. A. Von Roemeling, Y. Chen, X. Liu, K. D. Shih, J. A. Knight, H. W. Tun, R. E. Wharen, W. Jiang, Sci. Rep., 2016, 6, 26269; DOI: https://doi.org/10.1038/srep30663.
Y. Chen, Y. Xianyu, X. Jiang, Acc. Chem. Res., 2017, 50, 310; DOI: https://doi.org/10.1021/acs.accounts.6b00506.
B. A. Moser, R. C. Steinhardt, A. P. Esser-Kahn, ACS Biomater. Sci. Eng., 2016, 3, 206; DOI: https://doi.org/10.1021/acsbiomaterials.6b00473.
G. Cui, J. Wang, S. Zhang, Chem. Soc. Rev., 2016, 45, 4307; DOI: https://doi.org/10.1039/C5CS00462D.
G. V. Lisichkin, A. Yu. Olenin, Russ. Chem. Bull., 2018, 67, 949; DOI: https://doi.org/10.1007/s11172-018-2163-y.
F. Parveen, T. Patra, S. Upadhyayula, Carbohydr. Polym., 2016, 135, 280; DOI: https://doi.org/10.1039/C7NJ03146G.
Nanocatalysis in Ionic Liquids, Ed. M. H. G. Prechtl, John Wiley & Sons, Weinheim, Germany, 2017, pp. 83.
M. M. Abolghasemi, V. Yousefi, M. Piryaei, Microchim. Acta, 2015, 182, 2155; DOI: https://doi.org/10.1007/s00604-015-1553-1.
Z. Wu, C. Chen, Q. Guo, B. Li, Y. Que, L. Wang, H. Wan, G. Guan, Fuel, 2016, 184, 128; DOI: https://doi.org/10.1016/j.fuel.2016.07.004.
Z. Wu, C. Chen, L. Wang, H. Wan, G. Guan, Ind. Eng. Chem. Res., 2016, 55, 1833; DOI: https://doi.org/10.1021/acs.iecr.5b02906.
B. Karimi, M. R. Marefat, M. Hasannia, P. F. Akhavan, F. Mansouri, Z. Artelli, F. Mohammadi, H. Vali, Chem. Cat. Chem., 2016, 8, 2508; DOI: https://doi.org/10.1002/cctc.201600630.
R. Yang, X. Wang, Y. Zhang, H. Mao, P. Lan, D. Zhou, BioResources, 2019, 14, 87; DOI: https://doi.org/10.15376/biores.14.1.87-98.
B. Karimi, Z. Naderi, M. Khorasani, H. M. Mirzaei, H. Vali, ChemCatChem., 2016, 8, 906; DOI: https://doi.org/10.1002/cctc.201501229.
Q. Wang, W. Hou, Sh. Li, J. Xie, J. Li, Y. Zhou, J. Wang, Green Chem., 2017, 19, 3820; DOI: https://doi.org/10.1039/C7GC01116D.
K. Lagerblom, P. Wrigstedt, J. Keskiväli, A. Parviainen, T. Repo, Chempluschem., 2016, 81, 1160; DOI: https://doi.org/10.1002/cplu.201600240.
B. L. Ryland, S. S. Stahl, Angew. Chem., Int. Ed., 2014, 53, 8824; DOI: https://doi.org/10.1002/anie.201403110.
H. Wang, Y. Shi, M. Haruta, J. Huang, Appl. Catal., Part A, 2017, 536, 27; DOI: https://doi.org/10.1016/j.apcata.2017.02.015.
A. Vasseur, R. Membrat, D. Gatineau, A. Tenaglia, D. Nuel, L. Giordano, ChemCatChem., 2017, 9, 728; DOI: https://doi.org/10.1002/cctc.201601261.
K. Ando, J. Nakazawa, S. Hikichi, Eur. J. Inorg. Chem., 2016, 2603; DOI: https://doi.org/10.1002/ejic.201600206.
N. Armenise, N. Tahiri, N.N.H.M. Eisink, M. Denis, M. Jäger, J. G. De Vries, M. D. Witte, A. J. Minnaard, Chem. Commun., 2016, 52, 2189; DOI: https://doi.org/10.1039/C5CC08588H.
L. M. Dornan, M. J. Muldoon, Catal. Sci. Technol., 2015, 5, 1428; DOI: https://doi.org/10.1039/C4CY01632G.
K. Karami, N.H. Naeini, V. Eigner, M. Dusek, J. Lipkowski, P. Hervés, H. Tavakol, RSC Adv., 2015, 5, 102424; DOI: https://doi.org/10.1039/C5RA17249G.
S. Gowrisankar, H. Neumann, D. Gördes, K. Thurow, H. Jiao, M. Beller, Chem.—Eur. J., 2013, 19, 15979; DOI: https://doi.org/10.1002/chem.201302526.
B. Karimi, D. Elhamifar, J. H. Clark, A. J. Hunt, Org. Biomol. Chem., 2011, 9, 7420.
X. Sun, Y. Zheng, L. Sun, Q. Lin, H. Su, C. Qi, Nano-Struct. Nano-Object., 2016, 5, 7; DOI: https://doi.org/10.1039/C1OB05752A.
J. Li, T. Qi, L. Wang, Ch. Liu, Y. Zhang, Mater. Lett., 2007, 61, 3197; DOI: https://doi.org/10.1016/j.matlet.2006.11.079.
J. Azizian, H. Tahermansouri, E. Biazar, S. Heidari, D. Chobfrosh Khoei, Int. J. Nanomed., 2010, 5, 907; DOI: https://doi.org/10.2147/IJN.S13629.
A. Wolfson, S. Wuyts, D. E. De Vos, I. F. J. Vankelecom, P. A. Jacobs, Tetrahedron Lett., 2002, 43, 8107; DOI: https://doi.org/10.1016/S0040-4039(02)01921-4.
A. Dijksman, I. W. C. E. Arends, R. A. Sheldon, Chem. Commun., 1999, 1591; DOI: https://doi.org/10.1039/A902594D.
J. Muzart, Tetrahedron, 2003, 49, 5789; DOI: https://doi.org/10.1016/j.tet.2005.04.002.
M. Nasrollahzadeh, M. Sajjadi, M. Shokouhimehr, R. S. Varma, Coord. Chem. Rev., 2019, 397, 54; DOI: https://doi.org/10.1016/j.ccr.2019.06.010.
Sh. S. Stahl, Angew. Chem., Int. Ed., 2004, 43, 3400; DOI: https://doi.org/10.1002/anie.200300630.
B. A. Steinhoff, Sh. R. Fix, Sh. S. Stahl, J. Am. Chem. Soc., 2002, 124, 766; DOI: https://doi.org/10.1021/ja016806w.
F. Zamani, S. M. Hosseini, Catal. Commun., 2014, 43, 164; DOI: https://doi.org/10.1016/j.catcom.2013.09.029.
L. Zhang, P. Li, J. Yang, M. Wang, L. Wang, Chempluschem., 2014, 79, 217; DOI: https://doi.org/10.1002/cplu.201300353.
D. Wang, C. Deraedt, L. Salmon, C. Labrugère, L. Etienne, J. Ruiz, D. Astruc, Chem.—Eur. J., 2015, 21, 6501; DOI: https://doi.org/10.1002/chem.201500122.
H. Veisi, S. Hemmati, M., Tetrahedron Lett., 2017, 58, 4191; DOI: https://doi.org/10.1016/j.tetlet.2017.09.057.
P. Sangtrirutnugul, T. Chaiprasert, W. Hunsiri, T. Jitjaroendee, P. Songkhum, K. Laohhasurayotin, T. Osotchan, V. Ervithayasuporn, ACS Appl. Mater. Interfaces, 2017, 9, 12812; DOI: https://doi.org/10.1021/acsami.7b03910.
Q. Wang, X. Cai, Y. Liu, J. Xie, Y. Zhou, J. Wang, Appl. Catal., Part B, 2016, 189, 242; DOI: https://doi.org/10.1016/j.apcatb.2016.02.067.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1194–1203, June, 2022.
Rights and permissions
About this article
Cite this article
Karimi, Z., Hassanpour, A., Kangari, S. et al. Highly dispersed palladium nanoparticles supported on an imidazolium-based ionic liquid polymer: an efficient catalyst for oxidation of alcohols. Russ Chem Bull 71, 1194–1203 (2022). https://doi.org/10.1007/s11172-022-3520-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-022-3520-4